Medicare prescription drug coverage is an optional benefit offered to everyone who has Medicare. There are 2 ways for eligible patients to enroll—through Medicare Advantage or through Medicare Part D. Medicare Advantage plans (also known as Part C) offer Medicare prescription drug coverage along with medical coverage and other benefits. Since 2006, Medicare beneficiaries have had access to prescription drug coverage through Part D, where private plan sponsors contract with Medicare to provide drug benefits only. Part D benefits can be purchased in addition to Original (or fee-for-service) Medicare and supplemental, or Medigap coverage, neither of which covers prescription drugs. Medicare Part D plans cover brand-name and generic prescription drugs.
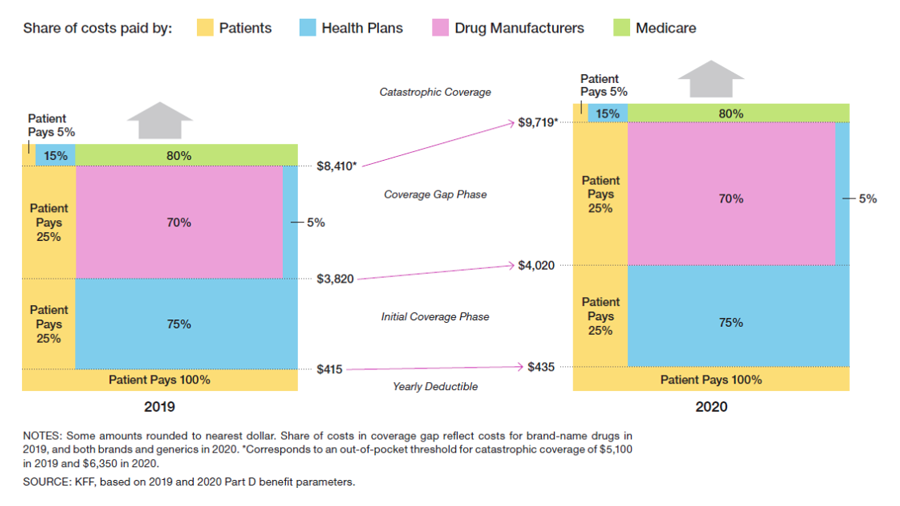
The standard Medicare Part D benefit has changed for 2020, as shown below.
The Medicare Part D yearly deductible increases to $435 in 2020, up from $415 in 2019. Patients must pay 100% of the cost of their prescription drugs until the cap of $435 is reached. After the yearly deductible is met, patients move through the initial coverage phase and are responsible for 25% of brand-name drug costs until a total of $4020 is spent. Subsequently, they move through the coverage gap phase (formerly the donut hole, which no longer exists) until a total of $9719 is spent. During the initial coverage and coverage gap phases, the remaining 75% is paid by health insurers and drug companies. The cap was increased substantially between 2019 and 2020, from $8410 to $9719. As a result, many Medicare Part D enrollees will face a substantial increase in out-of-pocket drug costs before they qualify for catastrophic coverage in 2020.
After reaching the catastrophic phase of coverage, beneficiaries are responsible for 5% of the cost of prescription drugs for the remainder of the calendar year. However, many Medicare Part D plans have no out-of-pocket spending cap. Even with 5% cost-sharing, this can have important financial implications for some patients throughout the calendar year.