With expert commentary by
Alison Duffy, PharmD, BCOP
Made possible by funding from



The endorsement mark certifies that the information presented in educational seminars, publications, or other resources is reliable and credible.
The most common adult leukemia is chronic lymphocytic leukemia (CLL), with approximately 21,250 new cases and 4320 attributed deaths in 2021.1 In CLL, there is a higher incidence in men compared with women (6.7 vs 3.5 cases per 100,000/year) and in white patients (7.3 [men] and 3.9 [women] cases per 100,000/year) compared with black patients (4.7 [men] and 2.2 [women] cases per 100,000/year).2
A stable course may be seen in patients with CLL, but the disease can become aggressive with frequent relapse episodes, or it may transform into an aggressive lymphoma type, such as diffuse large B-cell lymphoma, known as Richter transformation.3 CLL has a variable natural history, where patients are estimated to survive from approximately 2 to 20 years.4 Most patients with CLL follow a “watch and wait” approach, which involves determining the disease progression rate followed by periodic symptom development assessment.5
Commentary by Alison Duffy, PharmD, BCOP: As oral chemotherapy has taken an exciting, front row seat in the CLL treatment landscape, oncology pharmacists are increasingly involved in the care of patients with CLL, both within the ambulatory clinic space as well as at the dispensing pharmacies. Pharmacists play a key role in facilitating affordable and timely medication access, providing initial oral chemotherapy education prior to patients starting therapy, assessing education comprehension/retention and medication access soon after oral chemotherapy is started, as well as conducting adherence/tolerability follow-up assessments that can be conducted over the phone or in person, and complement those “check-ins” from other vital healthcare team members.
Shared Decision-Making
Initiation of Therapy
Pharmacists and other healthcare professionals apart from the core research team have been shown to be effective for recruiting patients to participate in clinical trials due to established relationships with patients who may be potential trial participants.6,7 Pharmacists are thought to be particularly helpful for recruiting to community-based trials or trials focusing on adherence or medications.
Commentary by Alison Duffy, PharmD, BCOP: Pharmacists greatly assist in screening for concomitant prescription and nonprescription medications that may interact with the patient’s investigational medication or CLL itself. All healthcare providers bring different perspectives, experience, and backgrounds to the table. I have observed that pharmacists can really play a key role in educating about medication administration (eg, in relation to taking with or without food), safely disposing previous therapies and safely handling oral chemotherapy (especially for caregivers), as well as discussing how to monitor for known clinically significant drug interactions that might not be able to be avoided.
Pharmacists can also contribute to improved health outcomes and patient satisfaction through services such as patient education, comprehensive medication management, and medication access coordination (Figure 1).8-12 An integral component of successful therapy, patient education and adherence is a collaborative effort of patients, family members, healthcare providers, and pharmacists.13 Successful counseling of patients should include treatment indication, dosing regimen, adverse effects, storage and administration, supportive care, and medication reconciliation.14 Results from an observational study demonstrated that 86% of patients believed that meeting with a pharmacist prior to initiating chemotherapy was absolutely necessary, and 83% of patients noted that they would be willing to pay for this service.9
Commentary by Alison Duffy, PharmD, BCOP: Asking how patients recognize and keep track of their current medications and the role caregivers have in daily medication administration as well as any past adherence barriers or strategies that have helped or hindered adherence is key before educating the patient on oral chemotherapy. Developing SMART (Smart, Measurable, Achievable, Relevant, Time-bound) goals and a customized medication schedule that fits into the patient’s daily routine is critical and really helps develop rapport with patients and their loved ones from the start. It can be helpful to identify when patients are most successful at taking other medications and considering adding a new medication to that routine (if that is appropriate based on how the medication should be administered). If a new medication must be taken at a different time or a patient has not routinely taken medications before, it can be helpful to identify another type of activity that happens regularly and add to that routine (keep next to bed and take after brushing teeth before bedtime).
A patient education checklist is shown in Figure 2.15,16 Medication-related variables to evaluate include the treatment’s place in therapy, targets, metabolism/transport, drug–food/drug–drug (including herbal medication and medical cannabis) interactions (if there are drug interactions, it is important to discuss how clinically significant they are and to make a new action plan for/modify the patient’s oral chemotherapy dose, modify the dose of the offending agent, and monitor more closely using labs and/or symptoms); adverse effects (prevention, monitoring, management, and past supportive care medications that were effective/ineffective and tolerated/not tolerated); contraindications (based on the patient’s comorbidities and past intolerances); dosing/administration (dose modifications needed, pill burden, frequency, administration with regard to food, impact of food on drug absorption); and home/type of hospital setting for initiation based on patient- and agent-related factors (eg, venetoclax based on tumor burden may need to be given inpatient initially). Patient-related variables to evaluate include comorbidities, age, frailty, performance status, ability to adhere (to oral chemotherapy/infusion appointments), and patient preference (time-directed vs continuous therapy options).
Commentary by Alison Duffy, PharmD, BCOP: When discussing new oral chemotherapy, I like to provide the patient with a handout from oral chemoedsheets.com and encourage the patient and caregiver to take notes that we can discuss later. If I have to complete education over the phone, I like to refer the patient to this website in advance so they can take a look and brainstorm any questions to make our education sessions even more successful.
Commentary by Alison Duffy, PharmD, BCOP: Educating on new therapies can be quite the “information overload” so it is important to use open-ended teach-back questions to gauge the patient and caregivers’ understanding. I usually preface my education sessions by stating that I will ask the patient some questions throughout the session to make sure that I have been clear and to make sure they are confident understanding about the new medication before starting. That helps tremendously with engagement during the session and helps to prevent the patient and caregiver from feeling like they are being quizzed at the end of the session.
Pharmacists completing a comprehensive medication review perform a detailed medication reconciliation and have the opportunity to identify any medication-related problems and access issues.13 Results from a study evaluating the impact of pharmacy student–driven medication reconciliation revealed that 88% of patients had ≥1 discrepancies on their medication list and 11.4% of patients had a problem related to medication.17 The small-molecule oral chemotherapeutic agents acalabrutinib, ibrutinib, and venetoclax are preferred first-line therapies for patients with CLL. These agents are metabolized by the cytochrome P450 3A enzyme, which leads to the requirement for dose adjustments in the context of concomitant administration with 3A inhibitors and inducers.18-20 Excessive toxicity or loss of efficacy may occur upon administration of these agents with interacting medications, which may lead to a negative impact on patient outcomes. Drug–drug interactions are relevant in patients with CLL because they are often diagnosed later in life when they are managing other medical comorbidities. Each agent has specific requirements for administration (eg, taking venetoclax with food to optimize absorption, separating acalabrutinib and acid-suppressing agents) that may lead to complications for appropriate medication adherence in patients taking several other medications.18,20
A retrospective analysis of patients with CLL taking ibrutinib demonstrated that medication adherence <80% was associated with worse progression-free survival.21 It has been demonstrated that missing 8 consecutive doses of ibrutinib predicts worse progression-free survival, highlighting the importance of monitoring follow-up and continuity of care.22 Therefore, ensuring adherence to oral therapies for patients with CLL is an opportunity for pharmacists to improve clinical outcomes.
Commentary by Alison Duffy, PharmD, BCOP: There are different ways to ask patients about missing doses, but the key is to stress that they aren’t in trouble and that honesty is key so that we can help them and work together to set them up for success. At my institution, we typically ask patients to reflect on if doses were missed or taken differently in the past 2-week window. We think of developing new strategies to help with adherence, if 2 or more doses are missed within a 2-week window. Patients and caregivers are increasingly engaged in their care if they understand the importance of adherence as it relates to treating their CLL upfront. It is also helpful to let patients know in advance how to keep track of this, to notify us of issues, and that we will ask routinely during follow-up visits. This gives the patient time to reflect on what is working for them and helps us brainstorm strategies together to improve adherence earlier on.
Commentary by Alison Duffy, PharmD, BCOP: Asking how patients recognize and keep track of their current medications and the role caregivers have in daily medication administration as well as any past adherence barriers or strategies that have helped or hindered adherence is key before educating the patient on oral chemotherapy. Developing SMART goals and a customized medication schedule that fits into the patient’s daily routine is critical and really helps develop rapport with patients and their loved ones from the start. It can be helpful to identify when patients are most successful at taking other medications and considering adding a new medication to that routine (if that is appropriate based on how the medication should be administered). If a new medication has to be taken at a different time or a patient has not routinely taken medications before, it can be helpful to identify another type of activity that happens regularly and add to that routine (keep next to bed and take after brushing teeth before bedtime). Although most patients would likely benefit from an oral chemotherapy diary and side-effect tracking journal, some patients may be more successful doing so via a side-effect monitoring app (eg, chemoWave), while some patients may prefer a handwritten diary to keep on their fridge if they take their medications around a mealtime at home versus tracking on a medication app (eg, Medisafe). Patients with past adherence challenges such as busy work schedules may benefit from a travel pillbox to remember to take their medications at work with them. Some of my favorite medication apps include Medisafe, MyMeds, CareZone, and Mango Health. Some of my favorite chemotherapy toxicity journaling apps include chemoWave, Cancer.net, CaringBridge, and LivingWith™: Cancer support.
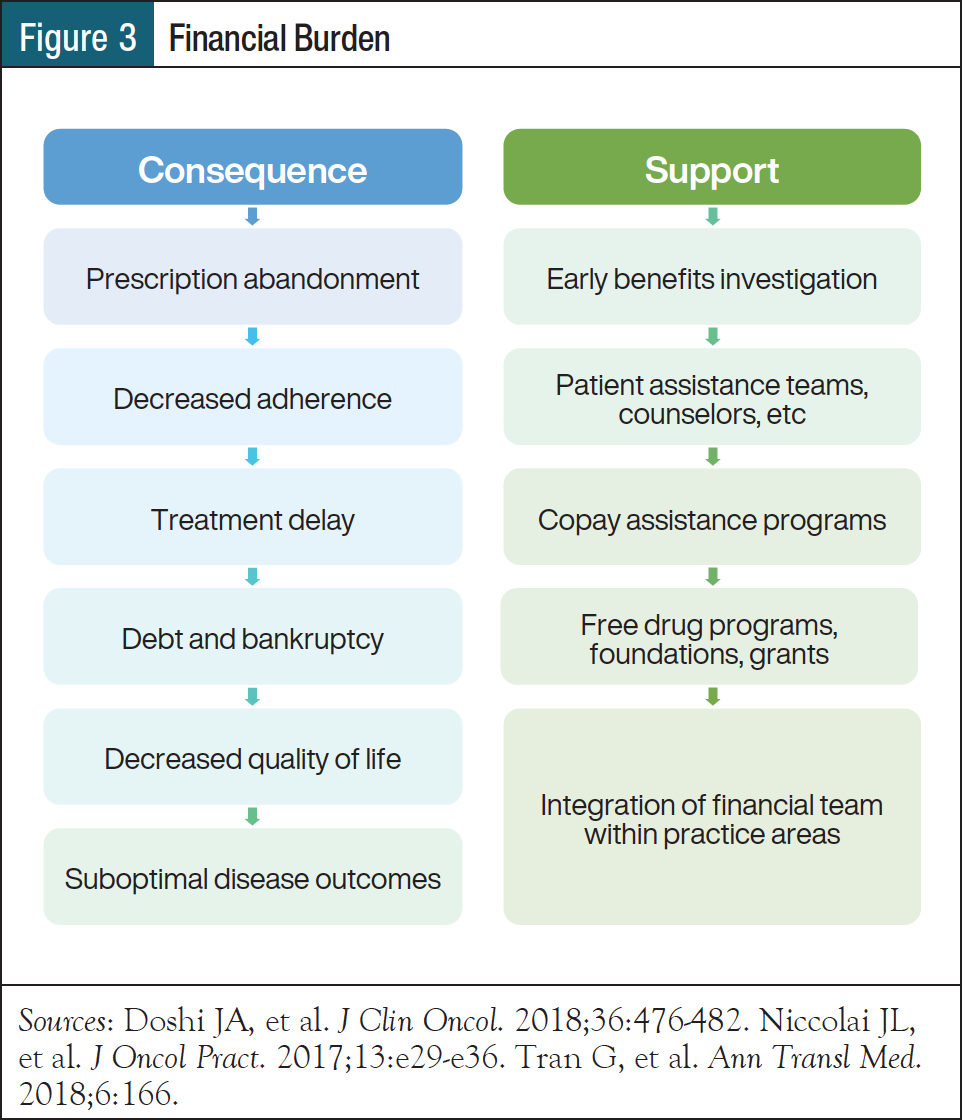
Traditionally, pharmacists have played a key role in the acquisition of costly medications (Figure 3).13,23-25 Embedded medication assistance program pharmacy technicians work with clinical pharmacists to reduce treatment delays through the assistance of uninsured and low-income patients in need of costly medications. The use of this multidisciplinary team–based approach helps to ensure that manufacturer assistance, grant applications, and prior authorizations are quickly and efficiently navigated to reduce out-of-pocket costs and minimize financial toxicity (Figure 4).9,26-32
Patient activation and communication
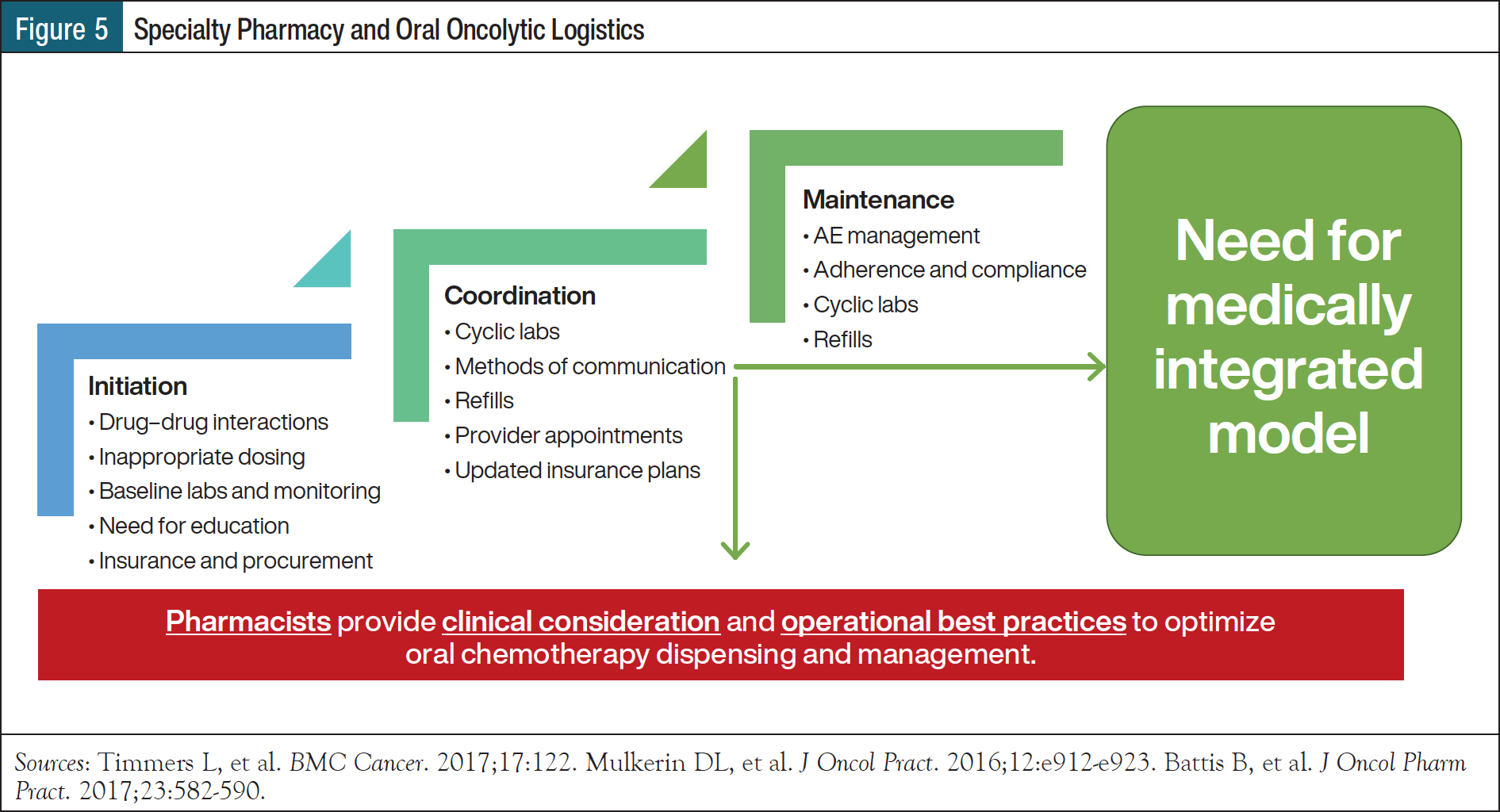
Pharmacists should be involved throughout the continuum of disease, through initiation, coordination, and maintenance (Figure 5).33-35 During the initial patient assessment, pharmacists should discuss several topics with patients/caregivers, and other healthcare providers may provide input as appropriate. These topics include past challenges with oral chemotherapy (access; adherence; if adherence challenges, identify barriers; past adherence strategies or tools that were/were not helpful; tolerability); the patient’s readiness for an education session (barriers to information comprehension on a given day); ability to swallow medicines (and if so, whether the medicine can be compounded safely without impacting efficacy); ability to read prescription labels due to eyesight/cognitive impairment; language barriers; support for managing medications and CLL; and type of learner (eg, visual, auditory) to tailor the education session.
Commentary by Alison Duffy, PharmD, BCOP: All healthcare providers bring different perspectives, experience, and backgrounds to the table. I rely heavily on my nursing and physician colleagues in terms of guiding patients in side-effect monitoring. I find that I am especially helpful when it comes to guiding the patient and healthcare team on how to use supportive care medications, such as antiemetics, to prevent and treat different side effects and to guide the patient on how to monitor to see if these supportive care medications are working and tolerated well.
After the initial patient assessment, pharmacists should discuss drug name(s) and how to identify medications (eg, pill color, shape); goals of therapy (and how to monitor from an efficacy perspective if not covered by the provider); medication administration and dosing (including what to do if missed/extra doses/taking at different time than customized administration schedule developed); and adherence (discuss importance for CLL and tie to efficacy outcomes, tools to use, develop customized administration schedule to incorporate into patient’s routine). It is also important to set a goal with the patient to be assessed at the next encounter (eg, related to adherence/tolerability and symptom management), and to discuss adverse effects (how to prevent, manage, monitor, and when to call for help), safe handling and disposal, and access (how to obtain medication).
To address financial barriers and issues related to medication access, pharmacists may be involved with discussions related to insurance procurement and coverage (what pharmacy will be used, how and when to receive medication, patient copay, need and co-facilitation of financial assistance program enrollment). Clinical pharmacists may also serve as a facilitator among specialty pharmacies, nursing navigators, social work and financial coordinators and advocates, patient assistance programs, and patient/caregiver.
Throughout the treatment process, pharmacists should remain in contact with patients who have CLL to facilitate coordination of care. Considerations include method of communication refills, appointments with oncologists and other healthcare providers, calls, updated insurance information, and labs. Best practices for follow-up calls/encounters after initial education include verification of information retention and comprehension, medication access, and early toxicities. During the maintenance phase, pharmacists should remain involved in adverse event management, adherence/compliance assessments, cyclic labs, and refills.
Recommended Resources
Additional resources related to education and toxicity monitoring are available for healthcare practitioners and for patients/caregivers:
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma
www.nccn.org/guidelines/guidelines-detail?category=1&id=1478 - Leukemia & Lymphoma Society®: Chronic Lymphocytic Leukemia
www.lls.org/leukemia/chronic-lymphocytic-leukemia?src1=20032&src2 - National Cancer Institute: Chronic Lymphocytic Leukemia Treatment (PDQ®)–Health Professional Version
www.cancer.gov/types/leukemia/hp/cll-treatment-pdq - CLL Society
https://cllsociety.org - Hematology/Oncology Pharmacy Association: Oral Chemotherapy Resources
www.hoparx.org/images/hopa/resource-library/guidelines-standards/Oral_Chemo_Resources_Web_Outline.pdf - Oncology Nursing Society Oral Adherence Toolkit
www.ons.org/sites/default/files/ONS_Toolkit_ONLINE.pdf - Oral Chemotherapy Education
www.oralchemoedsheets.com/index.php - Chemocare.com
https://chemocare.com - Academy of Oncology Nurse & Patient Navigators (AONN+): Education & Resources
https://aonnonline.org/education - CONQUER 2022 Patient Guide to Cancer Support Services
https://conquer-magazine.com/pssguide2022
Method
Pharmacists have an essential role in the management of patients with CLL. Pharmacists have medication-related expertise and a unique perspective that contributes value throughout the disease continuum. Pharmacists may play a leading role in the care of patients with CLL, from providing education and medication management to completing follow-up clinic visits. A multidisciplinary care model whereby pharmacists work alongside physicians, nurses, navigators, advanced practice providers, and other support staff allows pharmacists to use the extent of their training and practice at a high level. Due to the complexity of the treatment regimens, numerous toxicities and drug–drug interactions, and comorbidity management, CLL requires pharmacist collaboration, which is only likely to become more paramount as the treatment landscape expands.
References
- American Cancer Society. Key statistics for chronic lymphocytic leukemia. Revised January 12, 2021. www.cancer.org/cancer/chronic-lymphocytic-leukemia/about/key-statistics.html. Accessed March 8, 2021.
- National Cancer Institute Surveillance, Epidemiology, and End Results Program. SEER*Explorer: an interactive website for SEER cancer statistics. Surveillance Research Program NCIAM, 2022. https://seer.cancer.gov/explorer. Accessed March 15, 2022.
- Delgado J, Nadeu F, Colomer D, Campo E. Chronic lymphocytic leukemia: from molecular pathogenesis to novel therapeutic strategies. Haematologica. 2020;105:2205-2217.
- Scarfò L FA, Ghia P. Chronic lymphocytic leukaemia. Crit Rev Oncol Hematol. 2016;104:169-182.
- JG G. How I treat CLL up front. Blood. 2010;115:187-197.
- Tsuyuki RT, Al Hamarneh YN, Jones CA, Hemmelgarn BR. The effectiveness of pharmacist interventions on cardiovascular risk: the multicenter randomized controlled RxEACH trial. J Am Coll Cardiol. 2016;67:2846-2854.
- Peytremann-Bridevaux I, Bordet J, Santschi V, et al. Community-based pharmacies: an opportunity to recruit patients? Int J Public Health. 2013;58:319-322.
- McKee M, Frei BL, Garcia A, et al. Impact of clinical pharmacy services on patients in an outpatient chemotherapy academic clinic. J Oncol Pharm Pract. 2011;17:387-394.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association best practices for the management of oral oncolytic therapy: pharmacy practice standard. J Oncol Pract. 2019;15:e346-e355.
- Hibbard JH, Greene J. What the evidence shows about patient activation: better health outcomes and care experiences; fewer data on costs. Health Aff (Millwood). 2013;32:207-214.
- Hibbard JH, Greene J, Overton V. Patients with lower activation associated with higher costs; delivery systems should know their patients’ ‘scores.’ Health Aff (Millwood). 2013;32:216-222.
- Carman KL, Dardess P, Maurer M, et al. Patient and family engagement: a framework for understanding the elements and developing interventions and policies. Health Aff (Millwood). 2013;32:223-231.
- Chen KY, Brunk KM, Patel BA, et al. Pharmacists’ role in managing patients with chronic lymphocytic leukemia. Pharmacy (Basel). 2020;8:52.
- Felton MA, van Londen GJ, Marcum ZA. Medication adherence to oral cancer therapy: the promising role of the pharmacist. J Oncol Pharm Pract. 2016;22:378-381.
- HOPA Oral Chemotherapy Resources. Checklist for a new start oral chemotherapy. hoparx.org/images/hopa/membership/Initiation-Checklist-for-a-New-Start-Oral-Chemotherapy.pdf. Accessed April 1, 2022.
- Oncology Nursing Society. Oral adherence toolkit. Published 2016. www.ons.org/sites/default/files/ONS_Toolkit_ONLINE.pdf. Accessed April 1, 2022.
- Ashjian E, Salamin LB, Eschenburg K, et al. Evaluation of outpatient medication reconciliation involving student pharmacists at a comprehensive cancer center. J Am Pharm Assoc (2003). 2015;55:540-545.
- AbbVie Inc. Venclexta (venetoclax) prescribing Information. Updated December 2021. www.rxabbvie.com/pdf/venclexta.pdf. Accessed March 14, 2022.
- AbbVie Inc. Imbruvica (ibrutinib) prescribing Information. Updated December 2020. https://imbruvica.com/files/prescribing-information.pdf. Accessed March 14, 2022.
- AstraZeneca. Calquence (acalabrutinib) prescribing Information. Updated November 2019. www.azpicentral.com/calquence/calquence.pdf#page=1. Accessed March 14, 2022.
- Williams AM, Baran AM, Casulo C, et al. Ibrutinib dose adherence and therapeutic efficacy in non-Hodgkin lymphoma: a single-center experience. Clin Lymphoma Myeloma Leuk. 2019;19:41-47.
- Barr PM, Brown JR, Hillmen P, et al. Impact of ibrutinib dose adherence on therapeutic efficacy in patients with previously treated CLL/SLL. Blood. 2017;129:2612-2615.
- Doshi JA, Li P, Huo H, et al. Association of patient out-of-pocket costs with prescription abandonment and delay in fills of novel oral anticancer agents. J Clin Oncol. 2018;36:476-482.
- Niccolai JL, Roman DL, Julius JM, Nadour RW. Potential obstacles in the acquisition of oral anticancer medications. J Oncol Pract. 2017;13:e29-e36.
- Tran G, Zafar SY. Financial toxicity and implications for cancer care in the era of molecular and immune therapies. Ann Transl Med. 2018;6:166.
- Hansen EA, Pietkiewicz JM, Blum BL. Evaluation of the feasibility and utility of a pharmacist-centered collaborative drug therapy management program for oncology-based symptom management. J Pharm Pract. 2016;29:206-211.
- Holle LM, Puri S, Clement JM. Physician-pharmacist collaboration for oral chemotherapy monitoring: insights from an academic genitourinary oncology practice. J Oncol Pharm Pract. 2016;22:511-516.
- Lam MS, Cheung N. Impact of oncology pharmacist-managed oral anticancer therapy in patients with chronic myelogenous leukemia. J Oncol Pharm Pract. 2016;22:741-748.
- Muluneh B, Schneider M, Faso A, et al. Improved adherence rates and clinical outcomes of an integrated, closed-loop, pharmacist-led oral chemotherapy management program. J Oncol Pract. 2018;14:e324-e334.
- Parsons LB, Edwards K, Perez A, et al. Positive outcomes associated with a pharmacist-driven oral chemotherapy program. J Hematol Oncol Pharm. 2015;5:99-108.
- Mancini R, Wilson D, Modlin J, et al. Oral chemotherapy: a focus in interventions and access. J Clin Oncol. 2012;30(suppl_34):Abstract 44.
- Koselke E, Marini B, Kraft S, et al. Implementation of and satisfaction with an outpatient oral anticancer program. J Hematol Oncol Pharm. 2015;5:62-68.
- Timmers L, Boons CCLM, Verbrugghe M, et al. Supporting adherence to oral anticancer agents: clinical practice and clues to improve care provided by physicians, nurse practitioners, nurses and pharmacists. BMC Cancer. 2017;17:122.
- Mulkerin DL, Bergsbaken JJ, Fischer JA, et al. Multidisciplinary optimization of oral chemotherapy delivery at the University of Wisconsin Carbone Cancer Center. J Oncol Pract. 2016;12:e912-e923.
- Battis B, Clifford L, Huq M, et al. The impacts of a pharmacist-managed outpatient clinic and chemotherapy-directed electronic order sets for monitoring oral chemotherapy. J Oncol Pharm Pract. 2017;23:582-590.