Isavuconazonium sulfate is a prodrug of the triazole antifungal, isavuconazole, and has been used for the treatment of invasive fungal disease.1 Isavuconazonium sulfate is approved by the US Food and Drug Administration for the treatment of invasive aspergillosis and mucormycosis in patients aged ≥18 years.1 Although isavuconazonium sulfate is generally well-tolerated, it can cause significant drug–drug interactions, given its metabolism via the cytochrome (CY)P450 pathway. Specifically, isavuconazonium sulfate is a major substrate for CYP3A4 and CYP3A5; an inhibitor of CYP3A4, CYP2C8, CYP2C9, CYP2C19, and CYP2D6; and a weak inducer of CYP2B6, CYP3A4, CYP2C8, and CYP2C9.1
Triazole antifungals often require therapeutic drug monitoring for the evaluation of their safety and efficacy. However, therapeutic drug monitoring of isavuconazonium sulfate in adults is not routinely performed, as a result of a phase 3 clinical trial of isavuconazonium sulfate demonstrating linear pharmacokinetics and only minimal interpatient variability in isavuconazole concentrations.2
Decembrino and colleagues evaluated the pharmacokinetics of isavuconazonium sulfate in a case series of children, and concluded that contrary to what has been seen in adults, therapeutic drug monitoring was warranted in younger patients, for whom there is currently no approved dosing strategy of this drug.3
In a 2020 clinical trial, Borman and colleagues completed subgroup analyses based on patient age, showing that patients aged <18 years had greater interpatient variability in isavuconazole serum concentrations than patients aged >18 years.4 They further noted that the patients who receive prolonged isavuconazonium sulfate therapy may over time have an accumulation of isavuconazonium sulfate and subsequent gradual elevation of serum isavuconazole concentrations.4
A phase 1 clinical trial was recently published that was focused on isavuconazonium sulfate pharmacokinetics in children,5 but research is still ongoing to determine the most appropriate dosing strategy in children, and if therapeutic drug monitoring is warranted.6 Currently, there are limited data on the dosing of isavuconazonium sulfate in the pediatric population.
Previous small studies have used isavuconazonium sulfate in the management of pediatric mucormycosis.7-9 Muggeo and colleagues successfully used isavuconazonium sulfate in 2 pediatric patients with mucormycosis and did not follow the therapeutic drug concentrations.7 Cornu and colleagues successfully used isavuconazonium sulfate with dosing guided by therapeutic drug monitoring for compassionate off-label use in a pediatric patient aged 3 years with disseminated mucormycosis.8 The researchers aimed for a serum isavuconazole concentration of 2 mcg/mL to 4 mcg/mL after initiating maintenance treatment with isavuconazonium sulfate at 70 mg daily.8 Finally, Pomorska and colleagues successfully used isavuconazonium sulfate for the treatment of disseminated mucormycosis in a pediatric patient aged 7 years.9 The isavuconazonium sulfate dose was started at 200 mg once daily, which was adjusted based on therapeutic drug monitoring, with a target serum isavuconazole concentration of 3 mcg/mL to 4 mcg/mL.9
We conducted a study to describe isavuconazonium sulfate dosing and therapeutic drug monitoring at a single, pediatric academic medical center, Children’s Hospital Colorado in Aurora. We included patients who received isavuconazonium sulfate for invasive fungal disease and had therapeutic drug monitoring between January 2017 and September 2020.
The demographic and clinical data were extracted from our electronic health record (EHR) system (Epic; Verona, WI) and included isavuconazonium sulfate doses, isavuconazole serum concentrations, concomitant medications, and serum transaminase and bilirubin levels.
The dosing regimens of isavuconazonium sulfate changed over time at our institution as new data were published, although the standard regimen in our institution was based on pharmacokinetic modeling in pediatric patients that during the writing of this article is still undergoing further testing in clinical trials.5,6 The doses of isavuconazonium sulfate were 10 mg/kg once daily for patients aged 1 to 18 years and 6 mg/kg once daily for those aged 6 months to 1 year, with a maximum dose of 372 mg of isavuconazonium sulfate daily.5,6
In our study, we used intravenous (IV) and oral formulations of isavuconazonium sulfate with identical dosing. Loading doses before the maintenance dosing were used in some (but not all) patients to achieve steady-state concentrations more quickly. For therapeutic drug monitoring, isavuconazole concentrations were drawn at least 7 days after the initiation of therapy or a change in dose. For all patients with a documented serum isavuconazole goal range, the target isavuconazole trough concentration was 2 mcg/mL to 4 mcg/mL based on a study in the adult population that demonstrated this range to be adequate for the management of invasive fungal disease.10
We measured isavuconazole serum concentrations at a commercial laboratory. Statistical analyses were completed using descriptive statistics. Our study was approved by the Colorado Multiple Institutional Review Board.
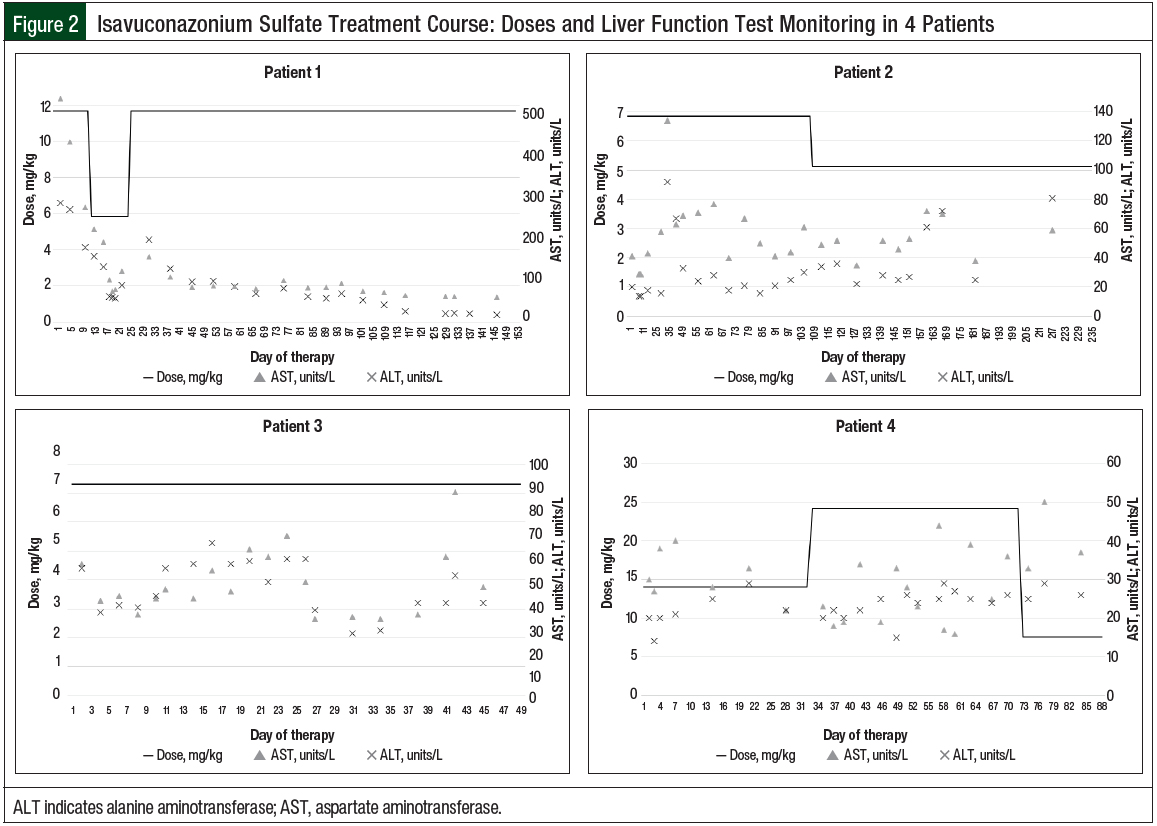
In this article, we report our institution’s use of therapeutic drug monitoring to guide the dosing of isavuconazonium sulfate in 4 patients who were receiving treatment for cancer or were undergoing hematopoietic stem-cell transplant (HSCT) at the hospital’s center for oncology and blood disorders; they also received isavuconazonium sulfate, as salvage therapy for invasive fungal disease, with therapeutic drug monitoring. All 4 patients had received posaconazole treatment before switching to treatment with isavuconazonium sulfate (Table).
The 4 patients combined received a total of 524 days of therapy with isavuconazonium sulfate, and 28 serum isavuconazole concentrations were obtained. Among the 4 patients, 3 required a dose adjustment of isavuconazonium sulfate based on therapeutic drug monitoring. None of the 4 patients had hepatotoxicity, which was defined by increased transaminase and/or bilirubin levels of >5 times the upper limit of normal (ULN) and would warrant the discontinuation of isavuconazonium sulfate.
Patient 1
An 11-year-old girl weighing 31.8 kg was receiving treatment in the hospital’s center for oncology and blood disorders for metastatic osteosarcoma and was transitioned from posaconazole to oral isavuconazonium sulfate therapy. She started treatment with isavuconazonium sulfate 372 mg (11.7 mg/kg) once daily, with a serum concentration goal of 2 mcg/mL to 4 mcg/mL, for the management of an infection caused by the Rhizopus species.
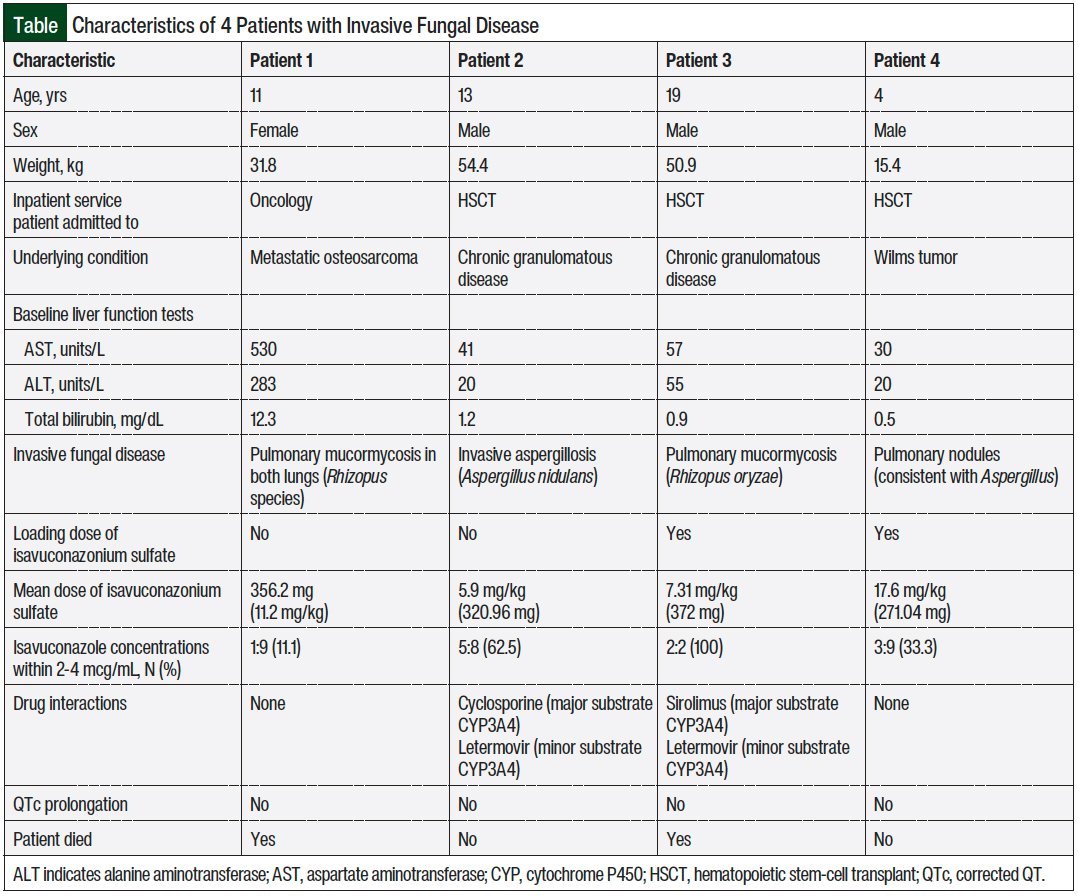
The doses of isavuconazonium sulfate were subsequently reduced by approximately 50% for a supratherapeutic serum isavuconazole concentration of 4.6 mcg/mL. After this dose change, the serum concentrations were considered low, at 2.1 mcg/mL and 1.1 mcg/mL; therefore, the dose was increased back to 372 mg daily. The serum isavuconazole concentrations ranged from 1.4 mcg/mL to 6.4 mcg/mL with this final regimen (Figure 1).
Of 9 isavuconazole concentrations, 6 (67%) were collected in the outpatient setting. This patient was not receiving any medication that could have increased or decreased her exposure to isavuconazonium sulfate, and she was not receiving concomitant immunosuppression therapy. On the day that this patient was switched from posaconazole to isavuconazonium sulfate therapy, her aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels were elevated by approximately 5 times the ULN (AST, 530 units/L; ALT, 283 units/L), and her total bilirubin was 12.3 mg/dL.
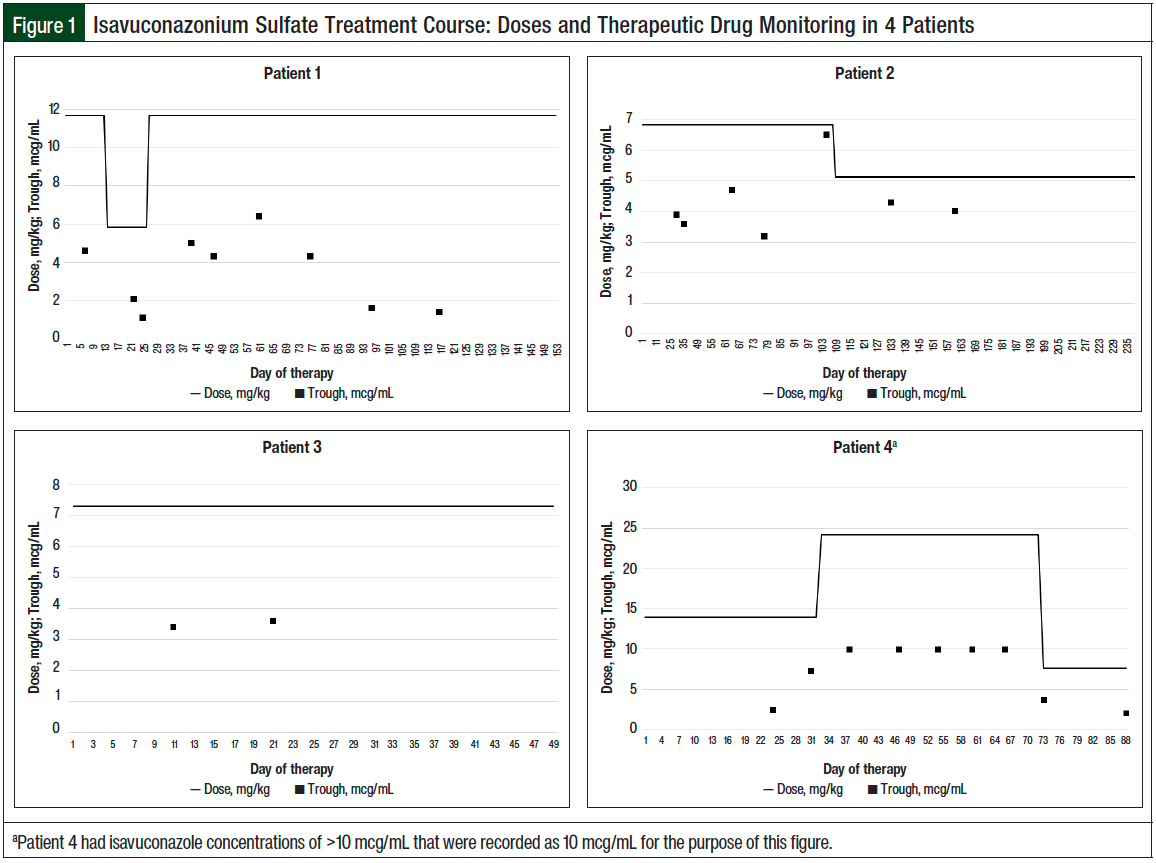
The patient’s AST, ALT, and bilirubin levels decreased after initiating treatment with isavuconazonium sulfate and were normalized within 3 weeks (Figure 2). The patient continued to receive isavuconazonium sulfate through the duration of our study, but she later died from her primary diagnosis of metastatic osteosarcoma.
Patient 2
A 13-year-old male weighing 54.4 kg with chronic granulomatous disease discontinued treatment with posaconazole for the treatment of invasive aspergillosis and was initiated treatment with oral isavuconazonium sulfate at the recommended adult dose (ie, 372 mg daily) after undergoing an allogeneic HSCT. The serum concentration goal for isavuconazole was 2 mcg/mL to 4 mcg/mL, with an ideal target closer to 3 mcg/mL, based on the minimum inhibitory concentration of his Aspergillus nidulans isolate, which was 0.25 mcg/mL.
The IV formulation was given for 9 days during the first month of therapy, when the patient was unable to take oral medications. His initial isavuconazole concentrations were appropriate, with serum concentrations ranging from 2 mcg/mL to 4.7 mcg/mL. After approximately 3 months of therapy without a change in dose, the patient had an elevated isavuconazole concentration of 6.5 mcg/mL. The isavuconazonium sulfate dose was then lowered by 25%, to 372 mg every other day, alternating with 186 mg every other day. His follow-up isavuconazole concentrations while receiving this regimen ranged from 4 mcg/mL to 4.3 mcg/mL (Figure 1). A total of 8 isavuconazole concentrations were collected, and 5 (63%) of these were collected in the outpatient setting.
While receiving isavuconazonium sulfate, the patient was concomitantly receiving letermovir, a moderate CYP3A4 inhibitor that can increase isavuconazole concentrations. Letermovir was initiated at the beginning of the patient’s course of isavuconazonium sulfate, and he continued to receive the same regimen for the duration of the isavuconazonium sulfate treatment. In addition, he was receiving immunosuppressive therapy with cyclosporine, an inhibitor of and substrate for CYP3A4, which was gradually tapered down and was discontinued during the last few months of isavuconazonium sulfate therapy.
The patient’s cyclosporine levels were not significantly affected during his course of isavuconazonium sulfate therapy, nor did the discontinuation of cyclosporine affect his serum isavuconazole concentrations. The patient’s transaminase levels were normal at the initiation of the isavuconazonium sulfate therapy, then became mildly elevated while receiving therapy (remaining <5 times the ULN), and later normalized while still receiving isavuconazonium sulfate therapy (Figure 2).
Patient 2 continued to receive isavuconazonium sulfate therapy for the duration of our study and had adequate antifungal treatment while receiving this regimen, because his infection did not progress.
Patient 3
A 19-year-old male weighing 50.9 kg with chronic granulomatous disease was transitioned from posaconazole therapy to isavuconazonium sulfate for the treatment of pulmonary mucormycosis after undergoing allogeneic HSCT. He received an oral loading dose of isavuconazonium sulfate 372 mg every 8 hours for 2 days (ie, 6 doses), followed by 372 mg once daily orally. The concentrations goal for isavuconazole were 2 mcg/mL to 4 mcg/mL to manage adequately his pulmonary mucormycosis with Rhizopus oryzae. Two isavuconazole concentrations were collected, ranging from 3.4 mcg/mL to 3.6 mcg/mL. No changes were made to the isavuconazonium sulfate dose throughout the course of therapy (Figure 1).
In the last 2 days of isavuconazonium sulfate treatment, the patient was switched to IV therapy, because of his inability to use oral medications. All the isavuconazole concentrations were collected in the inpatient setting. During this patient’s isavuconazonium sulfate treatment, he concomitantly received letermovir prophylaxis and sirolimus, a substrate of CYP3A4, for immunosuppression. He did not have any changes in sirolimus concentrations while also receiving isavuconazonium sulfate therapy. The patient’s transaminases began to rise near the end of the isavuconazonium sulfate course; however, these levels never exceeded 5 times the ULN (Figure 2). He continued to receive isavuconazonium sulfate treatment for invasive fungal disease, but eventually died from his underlying chronic granulomatous disease.
Patient 4
A 4-year-old boy weighing 15.4 kg with recurrent Wilms tumor started isavuconazonium sulfate therapy shortly after its approval for treatment in adults, but before the publication of any data about its efficacy in pediatric patients. The patient transitioned from posaconazole to isavuconazonium sulfate treatment after having an autologous HSCT for his cancer.
Antifungals were initiated because of concerning pulmonary nodules that were consistent with Aspergillus species infection. The patient was switched from posaconazole treatment to a loading dose of IV isavuconazonium sulfate 215 mg (14 mg/kg) every 8 hours for 2 days, followed by 215 mg daily for the treatment of pulmonary nodules consistent with Aspergillus infection. All his isavuconazonium sulfate doses were the IV formulation. The patient’s EHR report did not include any documented goal serum isavuconazole concentration range. He received approximately 3 weeks of isavuconazonium sulfate therapy before obtaining the first serum concentration of 2.5 mcg/mL, and no dose adjustments were reported.
After an additional week of therapy and a serum isavuconazole concentration of 7.3 mcg/mL, the isavuconazonium sulfate dose was increased to 372 mg (24.2 mg/kg), and all follow-up isavuconazole concentrations were >10 mcg/mL. The isavuconazonium sulfate dose was held for 2 days, and was then reduced to 117 mg (7.6 mg/kg) once daily, with a follow-up concentration of 2.1 mcg/mL while receiving the new regimen (Figure 1). All 9 isavuconazole concentrations were collected in the outpatient setting.
The patient did not receive any medications that would have been expected to increase or decrease isavuconazole concentrations, and he did not receive concomitant immunosuppression. A slight rise in the patient’s ALT level approximately 2 weeks after the initiation of therapy subsequently resolved, without any elevations in AST or bilirubin levels (Figure 2).
The patient’s isavuconazonium sulfate treatment was eventually changed back to posaconazole, because of concern by the clinical team that it could be contributing to his worsening neuropathy. After additional workup and discontinuation of isavuconazonium sulfate, consulting services agreed that the neuropathy was likely a Guillain-Barré syndrome variant that was associated with a recent viral illness.
Discussion
The findings from these 4 pediatric patients demonstrate interpatient and intrapatient variabilities in isavuconazole concentrations, suggesting the benefit of therapeutic drug monitoring in this younger-age population. In contrast to a published pharmacokinetic study in adults suggesting that isavuconazole therapeutic drug monitoring is not required,2 our findings are similar to the findings by Decembrino and colleagues of previous pediatric case reports.3
Decembrino and colleagues used a dosing regimen of 372 mg isavuconazonium sulfate for patients weighing >30 kg and a regimen of 186 mg isavuconazonium sulfate for those weighing <30 kg. In the <30-kg cohort of pediatric patients, the median isavuconazole concentrations was 1.1 mcg/mL, which is below the proposed target of 2 mcg/mL to 4 mcg/mL.3 In addition, in their phase 1 clinical trial, Arrieta and colleagues showed that a daily dose of oral or IV isavuconazonium sulfate at 10 mg/kg was well-tolerated, with similar exposure in adults and in children.5 The investigators noted that in certain patients it may be helpful to complete therapeutic drug monitoring to ensure therapeutic efficacy, because a majority of patients who did not have isavuconazole levels within the predicted drug exposure range were below the lower target of isavuconazole concentration threshold.5
Among our 4 young patients, the 1 adult (ie, the patient aged 19 years) was the only patient who did not require dose adjustment during his isavuconazonium sulfate therapy. The 3 patients aged <18 years required isavuconazonium sulfate dose adjustments to meet the target isavuconazole concentrations.
Of our 4 patients, 2 received an isavuconazonium sulfate loading dose before starting the maintenance dosing, which had no apparent correlation to serum isavuconazole ranges. Patient 1 had supratherapeutic and subtherapeutic concentrations while receiving the same regimen (11.7 mg/kg) at different time points, and patient 2 had supratherapeutic concentrations after 3 months of isavuconazonium sulfate exposure. Patient 4 had supratherapeutic concentrations while receiving relatively large doses of isavuconazonium sulfate that required withholding therapy and reducing the dose to normalize the isavuconazole concentrations.
Previous studies support the phenomenon of isavuconazonium sulfate accumulation, showing a linear increase over time in serum isavuconazole concentrations in patients who received prolonged isavuconazonium sulfate therapy.4,10 Although this was true for our patient 2, our patient 1 had decreasing and increasing isavuconazole concentrations while receiving the same dose over time, which further supports the significant variability in isavuconazole concentrations among individual children. Although 2 of our patients died secondary to their underlying malignancy, isavuconazonium sulfate was effective at preventing the progression of their invasive fungal disease.
All 4 of our patients tolerated isavuconazonium sulfate treatment well without apparent adverse events, but this drug has safety concerns.11 Although isavuconazonium sulfate treatment has fewer adverse events than other triazole antifungals, including a shortened corrected QT (QTc), a well-defined threshold for adverse events has yet to be established.12 Some data suggest that increased gastrointestinal side effects correlate with concentrations of isavuconazole between 4.87 mcg/mL and 5.13 mcg/mL and with more prolonged exposure to the drug.10 Unlike other triazoles, no clear association between isavuconazonium sulfate exposure and hepatotoxicity has been established.10
In patients who received posaconazole therapy and subsequently converted to treatment with isavuconazonium sulfate, the total bilirubin, alkaline phosphatase, ALT, and AST levels, as well as QTc interval, decreased and/or improved.13,14 These objective measures also improved in our 4 patients, of whom switched from posaconazole to isavuconazonium sulfate treatment. One patient had elevated AST, ALT, and total bilirubin levels >5 times the ULN before starting isavuconazonium sulfate therapy. The other 3 patients had slightly elevated transaminase levels before switching treatment, but all 4 patients had improved transaminases after changing from posaconazole to isavuconazonium sulfate treatment.
Isavuconazonium sulfate is a substrate, inhibitor, and inducer of major CYP450 enzymes that can produce significant drug–drug interactions, specifically in patients who receive certain antivirals and immunosuppressive therapy, such as tacrolimus, sirolimus, cyclosporine, and mycophenolate mofetil.15
In patients who receive concomitant isavuconazonium sulfate and immunosuppression therapy, immunosuppressive medications required a dose decrease by as much as 34.3% to maintain appropriate serum concentrations of the immunosuppressive medications.16
Two of our patients received isavuconazonium sulfate concomitantly with immunosuppressive therapy and did not have significant elevations in the immunosuppression drug concentrations. Furthermore, none of our patients received strong CYP3A4 inhibitors or inducers that affected the serum isavuconazole concentrations. Pediatric patients receiving isavuconazonium sulfate frequently have other medical problems and likely are receiving other medications that may influence metabolism and drug levels. This further supports the need for monitoring therapeutic drugs in children who are receiving isavuconazonium sulfate.
The small sample size of only 4 patients significantly limits the generalizability of our findings. A total of 2 patients were receiving concomitant medications that are also metabolized by CYP450 enzymes, and we cannot definitively know the influence this had on any patient’s individual drug exposure.
Patient 2 was receiving concomitant immunosuppressive medication with cyclosporine after HSCT that was eventually tapered down to discontinuation per HSCT protocol. It is unlikely that the treatment discontinuation influenced the serum concentrations of isavuconazole, given that cyclosporine is a weak CYP3A4 inhibitor.
In addition, although therapeutic drug monitoring can help guide therapy, difficulties in the precise timing of blood draws in the outpatient setting can lead to inaccurate or difficult-to-interpret serum concentrations. Nevertheless, we considered all obtained isavuconazole concentrations, including those obtained in the outpatient setting, because its long half-life (ie, approximately 130 hours) minimizes the impact of blood draw timing.1
Although we waited at least a week after isavuconazonium sulfate initiation or a dose change to obtain the isavuconazole concentrations, it is possible that isavuconazonium sulfate was not truly at a steady state when concentrations were collected given its long half-life.
Conclusion
As a triazole antifungal agent that is effective and well-tolerated in adults, with minimal adverse events, isavuconazonium sulfate has many potential benefits in a pediatric population of patients with invasive fungal disease. To ensure adequate isavuconazole serum concentrations, therapeutic drug monitoring can be helpful in children, including patients with cancer and invasive fungal disease, as was seen in patients presented here.
Author Disclosure Statement
The authors have no conflicts of interest to report.
References
- Cresemba (isavuconazonium sulfate) capsules/for injection, for oral use/for intravenous use [prescribing information]. Astellas US; February 2022. www.astellas.us/docs/cresemba.pdf. Accessed July 5, 2022.
- Kaindl T, Andes D, Engelhardt M, et al. Variability and exposure–response relationships of isavuconazole plasma concentrations in the phase 3 SECURE trial of patients with invasive mould diseases. J Antimicrob Chemother. 2019;74:761-767.
- Decembrino N, Perruccio K, Zecca M, et al. A case series and literature review of isavuconazole use in pediatric patients with hemato-oncologic diseases and hematopoietic stem cell transplantation. Antimicrob Agents Chemother. 2020;64:e01783-19. doi: 10.1128/AAC.01783-19.
- Borman AM, Hughes JM, Oliver D, et al. Lessons from isavuconazole therapeutic drug monitoring at a United Kingdom Reference Center. Med Mycol. 2020;58:996-999.
- Arrieta AC, Neely M, Day JC, et al. Safety, tolerability, and population pharmacokinetics of intravenous and oral isavuconazonium sulfate in pediatric patients. Antimicrob Agents Chemother. 2021;65:e00290-21. doi: 10.1128/AAC.00290-21.
- Desai AV, Kovanda LL, Lademacher C, et al. Predictions of isavuconazonium sulfate dosage in patients aged 6 months-<18 years by physiologically based pharmacokinetic modeling. Poster presented at IDWeek; October 3-7, 2018; San Francisco, CA.
- Muggeo P, Calore E, Decembrino N, et al. Invasive mucormycosis in children with cancer: a retrospective study from the Infection Working Group of Italian Pediatric Hematology Oncology Association. Mycoses. 2018;62:165-170.
- Cornu M, Bruno B, Loridant S, et al. Successful outcome of disseminated mucomycosis in a 3-year-old child suffering from acute leukemia: the role of isavuconazole? A case report. BMC Pharmacol Toxicol. 2018;19(81). doi: 10.1186/s40360-018-0273-7.
- Pomorska A, Malecka A, Jaworski R, et al. Isavuconazole in a successful combination treatment of disseminated mucormycosis in a child with acute lymphoblastic leukaemia and generalized haemochromatosis: a case report and review of the literature. Mycopathologia. 2019;184:81-88.
- Furfaro E, Signori A, Di Grazia C, et al. Serial monitoring of isavuconazole blood levels during prolonged antifungal therapy. J Antimicrob Chemother. 2019;74:2341-2346.
- Barg AA, Malkiel S, Bartuv M, et al. Successful treatment of invasive mucormycosis with isavuconazole in pediatric patients. Pediatr Blood Cancer. 2018;65:e27281. doi: 10.1002/pbc.27281.
- Keirns J, Desai A, Kowalski D, et al. QT interval shortening with isavuconazole: in vitro and in vivo effects on cardiac repolarization. Clin Pharmacol Ther. 2017;101:782-790.
- DiPippo AJ, Rausch CR, Kontoyiannis DP. Tolerability of isavuconazole after posaconazole toxicity in leukaemia patients. Mycoses. 2019;62:81-86.
- Mellinghoff SC, Bassetti M, Dörfel D, et al. Isavuconazole shortens the QTc interval. Mycoses. 2018;61:256-260.
- Pettit NN, Carver PL. Isavuconazole: a new option for the management of invasive fungal infections. Ann Pharmacother. 2015;49:825-842.
- Van Matre ET, Evans SL, Mueller SW, et al. Comparative evaluation of isavuconazonium sulfate, voriconazole, and posaconazole for the management of invasive fungal infections in an academic medical center. Ann Clin Microbiol Antimicrob. 2019;18:13. doi: 10.1186/s12941-019-0311-3.