Acute myeloid leukemia (AML) is the most common form of acute leukemia in adults, with a median age of approximately 65 years at diagnosis, and an increasing incidence with age.1 AML has been associated with environmental factors, inherited genetic abnormalities, and heterogeneous groups of gene mutations that often have overlapping incidences and prognostic significance.2-4
Different cytogenetic abnormalities and gene mutations influence survival in older patients5; however, few of these abnormalities are targetable, such as FMS-like tyrosine kinase 3 (FLT3) and isocitrate dehydrogenase 1 and 2 (IDH1/IDH2). Novel and efficient epigenetic-targeted therapeutics are the new approach to the management of AML and have shown significant improvement in patient survival.6-8 This article is focused on IDH1/IDH2-targeting agents, specifically, the IDH2 inhibitor, enasidenib.9
IDH enzymes catalyze the conversion of isocitrate to alpha-ketoglutarate.10 IDH2-mutated enzymes generate the “oncometabolite” 2-hydroxyglutarate (2-HG),10 which inhibits the TET2 function,9 leading to disordered hematopoietic stem-cell homeostasis and impaired myeloid differentiation precipitating a pro-leukemogenic effect that predispose individuals to hematologic malignancies, including AML.11
IDH2 mutations have been reported in 8% to 28% of patients with AML, and predominantly present as alterations in IDH2-R140 rather than IDH2-R172.12,13 Inhibition of the IDH2 mutation has led to decreased 2-HG levels and induced myeloid differentiation in patients with AML and IDH2 mutation.14,15
Enasidenib, a small-molecule inhibitor of IDH2 mutation, was approved in August 2017,16 based on results of a phase 1/2 clinical trial that showed an overall response rate of 40.3% in relapsed or refractory disease, with 19.3% of the patients achieving complete remission.17 At an approved dose of 100 mg orally daily, enasidenib targets the IDH2 mutation variants R140Q, R172S, and R172K.18
During the dose-escalation portion of the study that led to its approval, enasidenib was administered with a starting dose of 50 mg and up to a maximum dose of 650 mg once daily, in continuous 28-day cycles.15 The median 2-HG suppression from baseline was 92.4%, 90.4%, and 93.1% for patients receiving enasidenib doses of <100 mg, 100 mg, and >100 mg, respectively. Steady-state plasma levels were reached within 29 days of once-daily dosing. Clinical activity was observed at all enasidenib doses (50 mg-650 mg daily). The 100-mg daily dose was chosen as a result of good steady-state drug concentration. No dose-dependent 2-HG inhibition was observed among doses of <100 mg, 100 mg, or >100 mg in patients using enasidenib.15
Enasidenib inhibits the IDH2 mutation enzyme and suppresses 2-HG; however, suppression of 2-HG alone did not predict clinical response.19 The extent of 2-HG suppression in patients with R172 and IDH2 mutation was less than in patients with R140 and IDH2 mutation, but clinical responses were equivalent in both mutation subtypes.19 Therefore, 2-HG suppression is not useful as a predictor of response,20 which is believed to be affected by differential production of 2-HG by various cell populations during enasidenib-induced differentiation, specific co-mutational patterns, or alterations in AML cell metabolism.19
Promoting leukemic-cell differentiation by inhibition of IDH2 mutation is associated with clinical response that aimed to achieve near normalization of the immature-to-mature cell population.19 This could be enhanced by increasing the area under the curve (AUC) of enasidenib. The AUC of enasidenib increases in an approximately dose-escalation manner from a 50-mg (0.5 times the approved recommended dosage) to 450-mg (4.5 times the approved recommended dosage) daily dose.21
At higher doses (any dose >100 mg), enasidenib was less tolerated, but without increased incidence of nausea, vomiting, fatigue, hyperbilirubinemia, and diarrhea.15 Few patients received dose modification in the 650-mg daily dose group.15,16
Increasing the enasidenib dose could potentially increase the AUC, which could enhance leukemic-cell differentiation that would affect the clinical response and overcome an initial lack of response, with a risk for increased adverse events, as well. Although the treatment approach to AML is evolving, with several new therapies in the pipeline, the management of AML in the relapsed or refractory setting is challenging, because of patient tolerability issues and limited treatment options. Hence, investigating new approaches to optimizing the response to enasidenib for responding patients, with the aim of prolonging the time to the next line of therapy, is crucial at this stage of the disease.
Our article is based on retrospective observations of patients who were diagnosed with AML and IDH2 mutation between January 2017 and March 2019 at the Stephenson Cancer Center, University of Oklahoma, and received treatment with enasidenib after first relapse. The patients were followed until July 2020, with the first patient in our institution having started enasidenib therapy for relapsed or refractory AML and IDH2 mutation in March 2018.
Two patients received an escalated enasidenib dose of 200 mg daily. Before increasing the enasidenib dose, the experimental treatment plan, potential benefits, and adverse events were discussed at length with the patients and their families. The patients provided informed consent, understanding all risks, benefits, and alternatives for the proposed plan. Health insurance providers gave prior authorization to supply the increased dose.
We included patients with intermediate- to poor-risk AML who received at least 1 previous line of therapy, had started treatment with standard-dose enasidenib (100 mg daily) for a minimum of 3 months, and were followed until disease relapse. We excluded patients who were primarily resistant or intolerant to standard-dose enasidenib.
AML risk stratification and prognostication were evaluated after the 2017 European LeukemiaNet (2017 ELN) classification by gene expression and molecular profiling.22,23 We evaluated the response using 2017 ELN recommendations from an international expert panel at 3-month intervals from starting enasidenib until relapse.24
IDH2 mutations were identified by a local diagnostic laboratory that is regulated under the Clinical Laboratory Improvement Amendments through polymerase chain reaction (PCR), where DNA is isolated from the patient’s tumor, with or without previous microdissection, then amplified in 2 separate PCRs and sequenced using separate pyrosequencing reactions to determine the presence or absence of mutations in codons 140 and 172 of the IDH2 gene. The assay is validated to detect ≥10% mutated allele frequency.
To quantify IDH2 variant frequency, confirmatory testing was performed via the Associated Regional and University Pathologists next-generation sequencing panel.
Treatment-emergent adverse events, defined as events that began or worsened between the first enasidenib dose and 28 days after the last dose, were graded by using Common Terminology Criteria for Adverse Events, version 4.0.25 Serious treatment-emergent adverse events were those that were life-threatening, resulted in death, required hospitalization, or caused significant incapacity.
We report the outcomes for 2 patients who received escalated-dose enasidenib (200 mg orally daily) for relapsed or refractory AML and an IDH2 mutation.
Patient Case 1
A 65-year-old female was diagnosed with poor-risk AML in the setting of pancytopenia, with bone marrow evaluation consistent with the background of erythroid and granulocytic dysplasia, with a normal karyotype analysis. The patient received induction chemotherapy and 2 cycles of consolidation chemotherapy, which was complicated by bacterial infections, prolonged hospitalization, and profound deconditioning that ultimately led to stopping consolidation therapy.
After 18 months of surveillance, repeated bone marrow evaluation for new-onset pancytopenia revealed relapsed AML with evidence of IDH2 mutation. Next-generation sequencing confirmed the presence of a mutation at codon 140 of the IDH2 gene, with a variant frequency of 36.9%. The patient started treatment with an oral daily dose of enasidenib 100 mg orally. After 4 months of therapy, the patient continued to have cytopenia, and a repeated bone marrow evaluation showed residual AML with IDH2 variant frequency of 27.6%.
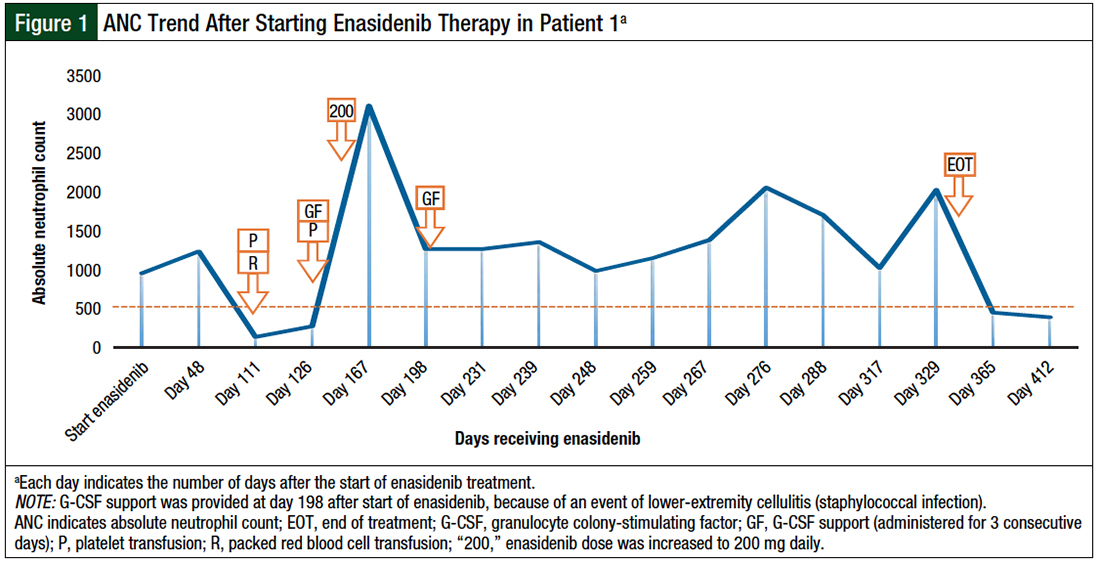
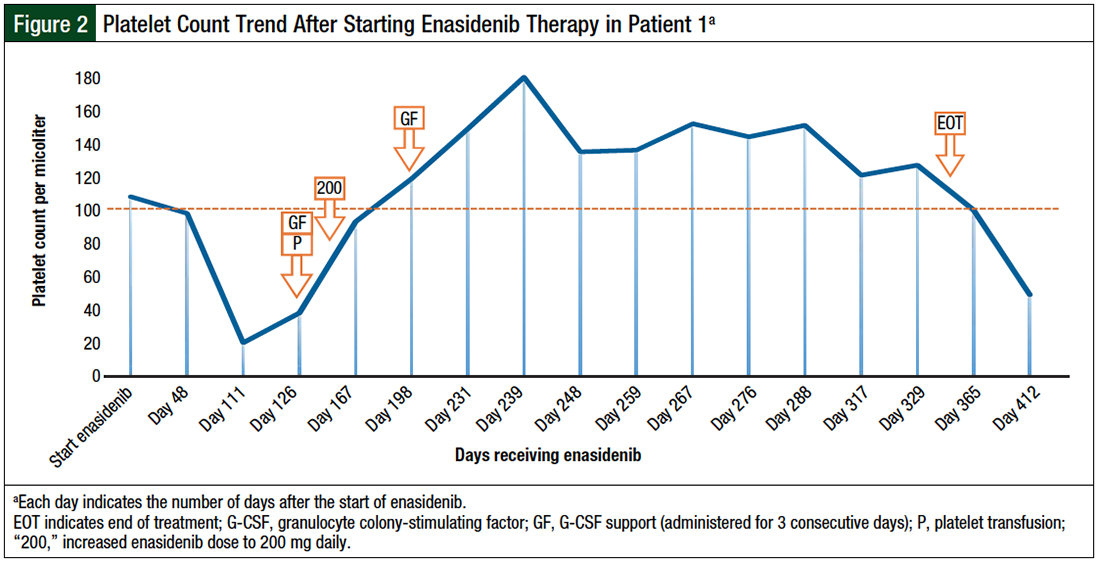
Based on an educated assumption that increasing the enasidenib dose will proportionately increase the AUC and enhance leukemic-cell differentiation to achieve near normalization of the immature-to-mature cell population, the decision was made to increase the enasidenib dose to 200 mg daily. With follow-up at 3 months after the initiation of the escalated dose, the patient achieved hematologic complete response, with normalization of peripheral cytopenia independent of growth factor support (Figure 1 and Figure 2).
The patient maintained hematologic complete response for 7 months while receiving 200 mg enasidenib daily, until she was diagnosed with COVID-19. The treatment was held, and she was switched to hospice care because of worsening respiratory failure.
The noted adverse events in this case were limited to fatigue, joint pains, lower-extremity pitting edema, and skin dryness and itchiness, which was complicated by staphylococcal infection.
Patient Case 2
A 59-year-old female presented with poor-risk AML in the setting of leukocytosis. A bone marrow evaluation was consistent with the background of megakaryocytic dysplasia and karyotype analysis was positive for monosomy 7. The patient initially received induction chemotherapy. At day 14 of induction chemotherapy, repeated bone marrow evaluation showed primary refractory disease with IDH2 mutation at codon 140, with a variant allelic frequency of 40.7%. Treatment with 100 mg enasidenib was started. The patient maintained a partial response for 6 months, when peripheral blast counts started to gradually increase.
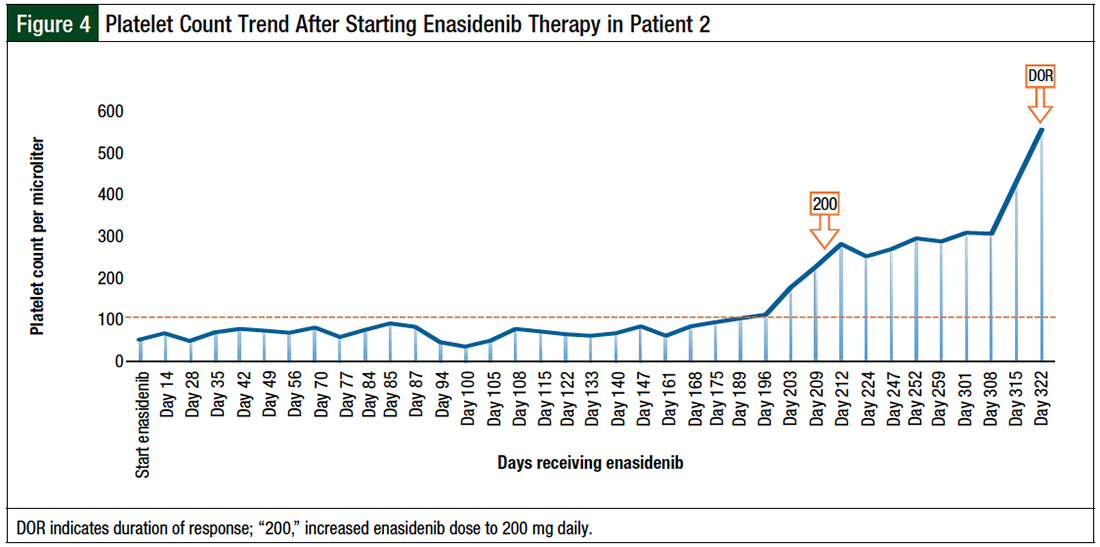
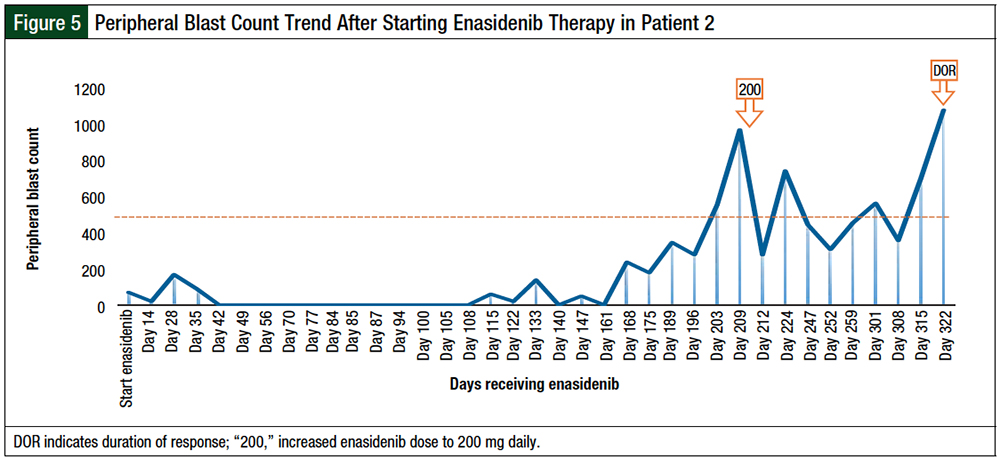
A repeated bone marrow evaluation showed partial response, with persistence IDH2 mutation by next-generation sequencing at a variant allelic frequency of >10%. The decision was made to increase the enasidenib dose to 200 mg daily, which led to a decrease in pretreatment peripheral blasts of >50% within 1 month of escalated-dose therapy.
We noted a spike in absolute neutrophil count (ANC) to 7050, with a proportionate increase in white blood cells to 9400 4 days after increasing the dose of enasidenib. The increase in white blood cells was not associated with any fevers, respiratory symptoms, or fluid retention, and did not indicate cytoreduction. This could represent a leukemic-cell differentiation. The patient had stable disease for 4 months while receiving the escalated dose before proceeding to the next line of therapy (Figures 3-5).
Adverse events in this patient included skin rash, trace lower-extremity edema, joint pains, constipation, and hyponatremia.
In these 2 cases, we did not notice gastrointestinal adverse events or hyperbilirubinemia with the increased enasidenib dose. All adverse events were controlled with supportive measures, and neither of the patients required hospitalization.
Discussion
These 2 cases represent the outcome in selected patients who received an escalated dose of enasidenib 200 mg daily. Our goal was to generate a hypothesis of utilizing ramp-up dosing of enasidenib to optimize clinical response. This strategy could be used as a single agent or in combination with hypomethylating agents in the relapsed or refractory AML setting, and as first-line therapy in clinical trials.26
By evaluating the response to treatment after cycle 3 with repeated bone marrow evaluation with confirming persistence of IDH2 mutation, we could optimize the treatment regimen (if the patient shows initial response, defined as complete response, complete response with incomplete hematologic recovery, morphologic leukemia-free state, or partial response), aiming to decrease the probabilities of disease relapse and prolong relapse-free survival.
With a median overall survival of approximately 6 months with active therapy, relapsed or refractory AML represents a formidable challenge.27
Treatment decisions and prognosis depend on patient factors, such as performance status, comorbidities, length of relapse-free survival, age at relapse, and transplant eligibility, as well as disease factors, such as previous treatments and risk stratification.28,29 Therefore, the goals of treatment also differ for each patient, such as considering allogeneic stem-cell transplant for eligible patients versus a palliative approach to improve quality of life and extend life expectancy for other patients.
IDH mutations appear to be exclusive of mutations in TET2, with unclear prognostic value at this time. Approximately 8% to 28% of patients with AML carry IDH2 mutations. Mutations occur at the conserved arginine residues.30 IDH2-R140 and IDH2-R172 are the most common mutations, with IDH2-R140 being the more common.12
Galkin and Jonas showed that enasidenib improved overall survival among patients with relapsed or refractory AML, with a median overall survival of 9.3 months, and longer overall survival among patients who achieved complete or partial response.27 A total of 87% of patients who ultimately responded to therapy showed a response by cycle 5.27 Observing the evolution of response at each treatment cycle for the study patients showed that the median time to attain a first response was 1.9 months, and the median time to achieve complete response was 3.8 months.13,31
However, the phase 3 IDHENTIFY clinical trial comparing the safety and efficacy of enasidenib in the relapsed or refractory setting versus conventional care regimens failed to meet its primary end point in achieving an overall survival benefit. The safety profile for enasidenib was consistent with previously reported findings.32
Although the full evaluation of data from IDHENTIFY is yet to be complete, enasidenib remains a viable option in the treatment of patients with AML and IDH2 mutation, given its safety profile as a result of its role in cell metabolism rather than cytotoxic effect, with lower risk for myelosuppression and less likelihood of neutropenia, infectious complications, and requirement for blood products compared with conventional salvage chemotherapy.32 These factors, along with its oral route of administration, make enasidenib a preferable option over the conventional care regimens, as it would likely provide better quality of life.
The phase 1/2 clinical trial that led to approval of enasidenib15 was based on the success of preclinical studies using 2-HG levels as a surrogate of drug activity.31 Although the study established that the 100-mg daily dose is efficacious in achieving a median 93% plasma reduction of 2-HG in patients with IDH2-R140 mutation and a median of 28% in patients with AML and IDH2-R172 mutation,15 other studies have analyzed the mechanisms of enasidenib clinical activity and have established several important facts.19,21
These facts are: (1) among doses of <100 mg, 100 mg, or >100 mg, no dose-dependent 2-HG inhibition was observed; (2) enasidenib can inhibit the IDH2-mutated enzyme and suppress 2-HG, regardless of clinical response; (3) 2-HG suppression is not predictive of clinical response; (4) AUC increases in a dose-proportional manner from a 50-mg to 450-mg daily dose;
(5) clinical response to enasidenib is associated with leukemic-cell differentiation.19,21
For our 2 patients, we evaluated the response to enasidenib 100 mg daily at 3-month intervals. After noting evidence of partial response (defined as complete response with incomplete blood count recovery after 4 months of therapy in Case 1 and partial response after 6 months of therapy in Case 2), or early evidence of relapse (defined as worsening neutropenia in Case 1 and increasing peripheral blast count in Case 2), we started administering the escalated-dose protocol. This was aimed at enhancing leukemic-cell differentiation that could improve the clinical response, after informed consent and disclosing the off-label use of this dose for enasidenib.
These 2 case reports show that by increasing the enasidenib dose, we can achieve more durable disease control and prolong the patient’s relapse-free survival. In the setting in which patients are unfit and/or ineligible for hematopoietic stem-cell transplantation, postponing the need for the next line of therapy is valuable, especially with limited success and less possibility of response.
With our limited cases of patients who received an increased dose of enasidenib, we did not report any treatment-emergent adverse events with increased dose. Common adverse events encountered were grade 1 and grade 2 diarrhea, and grade 1 lower-extremity edema and electrolyte disturbances; no gastrointestinal toxicity or hyperbilirubinemia were noted.
Increased enasidenib dose showed no evidence of differentiation syndrome in these patients. Although we noted leukocytosis in Case 2 shortly after increasing the dose, without any symptoms that would suggest differentiation syndrome and our patient did not need any cytoreductive therapy, we wish to emphasize the potential for an increased risk for differentiation syndrome with dose escalation. We therefore recommend close monitoring in the first 28 days of administering the increased dose of enasidenib, following the standard protocol for starting a new therapy.
Conclusion
Our 2 cases show that the initial response to standard-dose enasidenib could be optimized by dose escalation to 200 mg daily. Achieving meaningful response with enasidenib treatment in relapsed or refractory AML may be delayed, with a median time to first response of 1.9 months, and a median time to best response is 3.7 months; therefore, evaluation for response at 3-month intervals is needed. Evaluating the response to therapy earlier could provide an opportunity to optimize the treatment protocol. For initial responders to standard-dose enasidenib, evidenced by bone marrow evaluation with persistent positivity of IDH2 mutation, response could be optimized by increasing the enasidenib dose to 200 mg daily, with the potential to improve relapse-free survival.
Large multi-institutional randomized controlled studies are needed to test escalating the enasidenib dose and its impact on clinical outcomes. Studies to investigate the feasibility and safety of a ramp-up dosing strategy for initial responders to standard-dose enasidenib could also serve an important role.
Author Disclosure Statement
Dr Keruakous, Dr Schmidt, Dr Autry, Ms Ye, Ms Nguyen, and Mr Day have no conflicts of interest to report.
References
- Shallis RM, Boddu PC, Bewersdorf JP, Zeidan AM. The golden age for patients in their golden years: the progressive upheaval of age and the treatment of newly-diagnosed acute myeloid leukemia. Blood Rev. 2020;40:100639.
- Tenen DG, Hromas R, Licht JD, Zhang DE. Transcription factors, normal myeloid development, and leukemia. Blood. 1997;90:489-519.
- Eisfeld AK, Kohlschmidt J, Mims A, et al. Additional gene mutations may refine the 2017 European LeukemiaNet classification in adult patients with de novo acute myeloid leukemia aged <60 years. Leukemia. 2020;34:3215-3227.
- Mrózek K, Bloomfield CD. Chromosome aberrations, gene mutations and expression changes, and prognosis in adult acute myeloid leukemia. Hematology Am Soc Hematol Educ Program. 2006;169-177.
- Döhner H, Dolnik A, Tang L, et al. Cytogenetics and gene mutations influence survival in older patients with acute myeloid leukemia treated with azacitidine or conventional care. Leukemia. 2018;32:2546-2557.
- Liu XL, Liu HQ, Li J, et al. Role of epigenetic in leukemia: from mechanism to therapy. Chem Biol Interact. 2020;317:108963.
- Bewersdorf JP, Shallis R, Stahl M, Zeidan AM. Epigenetic therapy combinations in acute myeloid leukemia: what are the options? Ther Adv Hematol. 2019;10:1-19. doi:10.1177/2040620718816698.
- Wouters BJ, Delwel R. Epigenetics and approaches to targeted epigenetic therapy in acute myeloid leukemia. Blood. 2016;127:42-52.
- Shih AH, Meydan C, Shank K, et al. Combination targeted therapy to disrupt aberrant oncogenic signaling and reverse epigenetic dysfunction in IDH2- and TET2-mutant acute myeloid leukemia. Cancer Discov. 2017;7:494-505.
- Pollyea DA, Kohrt HE, Zhang B, et al. 2-hydroxyglutarate in IDH mutant acute myeloid leukemia: predicting patient responses, minimal residual disease and correlations with methylcytosine and hydroxymethylcytosine levels. Leuk Lymphoma. 2013;54:408-410.
- Figueroa ME, Abdel-Wahab O, Lu C, et al. Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell. 2010;18:553-567.
- Marcucci G, Maharry K, Wu YZ, et al. IDH1 and IDH2 gene mutations identify novel molecular subsets within de novo cytogenetically normal acute myeloid leukemia: a Cancer and Leukemia Group B study. J Clin Oncol. 2010;28:2348-2355.
- Stein EM, DiNardo CD, Fathi AT, et al. Molecular remission and response patterns in patients with mutant-IDH2 acute myeloid leukemia treated with enasidenib. Blood. 2019;133:676-687.
- Janin M, Mylonas E, Saada V, et al. Serum 2-hydroxyglutarate production in IDH1- and IDH2-mutated de novo acute myeloid leukemia: a study by the Acute Leukemia French Association group. J Clin Oncol. 2014;32:297-305.
- Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130:722-731.
- US Food and Drug Administration. FDA granted regular approval to enasidenib for the treatment of relapsed or refractory AML. August 1, 2017. www.fda.gov/drugs/resources-information-approved-drugs/fda-granted-regular-approval-enasidenib-treatment-relapsed-or-refractory-aml. Accessed January 18, 2021.
- Dogra R, Bhatia R, Shankar R, et al. Enasidenib: first mutant IDH2 inhibitor for the treatment of refractory and relapsed acute myeloid leukemia. Anticancer Agents Med Chem. 2018;18:1936-1951.
- Idhifa (enasidenib) tablets, for oral use [prescribing information]. Summit, NJ: Celgene Corporation; 2020.
- Amatangelo MD, Quek L, Shih A, et al. Enasidenib induces acute myeloid leukemia cell differentiation to promote clinical response. Blood. 2017;130:732-741.
- Wouters BJ. Hitting the target in IDH2 mutant AML. Blood. 2017;130:693-694.
- Li Y, Liu L, Gomez D, et al. Pharmacokinetics and safety of enasidenib following single oral doses in Japanese and Caucasian subjects. Pharmacol Res Perspect. 2018;6:e00436.
- Feng S, Zhou L, Zhang X, et al. Impact of ELN risk stratification, induction chemotherapy regimens and hematopoietic stem cell transplantation on outcomes in hyperleukocytic acute myeloid leukemia with initial white blood cell count more than 100 × 109/L. Cancer Manag Res. 2019;11:9495-9503.
- Harada Y, Nagata Y, Kihara R, et al; for the Japan Adult Leukemia Study Group. Prognostic analysis according to the 2017 ELN risk stratification by genetics in adult acute myeloid leukemia patients treated in the Japan Adult Leukemia Study Group (JALSG) AML201 study. Leuk Res. 2018;66:20-27.
- Döhner H, Estey E, Grimwade D, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424-447.
- Kluetz PG, Chingos DT, Basch EM, Mitchell SA. Patient-reported outcomes in cancer clinical trials: measuring symptomatic adverse events with the National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). Am Soc Clin Oncol Educ Book. 2016;35:67-73.
- Stein EM, DiNardo CD, Fathi AT, et al. Ivosidenib or enasidenib combined with intensive chemotherapy in patients with newly diagnosed AML: a phase 1 study. Blood. 2020 Oct 5. Epub ahead of print.
- Galkin M, Jonas BA. Enasidenib in the treatment of relapsed/refractory acute myeloid leukemia: an evidence-based review of its place in therapy. Core Evid. 2019;14:3-17.
- Breems DA, Van Putten WLJ, Huijgens PC, et al. Prognostic index for adult patients with acute myeloid leukemia in first relapse. J Clin Oncol. 2005;23:1969-1978.
- DeWolf S, Tallman MS. How I treat relapsed or refractory AML. Blood. 2020;136:1023-1032.
- Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43:1541-1551.
- Abou Dalle I, DiNardo CD. The role of enasidenib in the treatment of mutant IDH2 acute myeloid leukemia. Ther Adv Hematol. 2018;9:163-173.
- Bristol Myers Squibb. Bristol Myers Squibb provides update on phase 3 IDHENTIFY trial in patients with relapsed or refractory acute myeloid leukemia. August 25, 2020. https://news.bms.com/news/details/2020/Bristol-Myers-Squibb-Provides-Update-on-Phase-3-IDHENTIFY-Trial-in-Patients-with-Relapsed-or-Refractory-Acute-Myeloid-Leukemia/default.aspx. Accessed January 19, 2021.