Acute lymphoblastic leukemia (ALL) is a relatively rare cancer affecting 1.7 cases per 100,000 people annually in the United States, with a slightly higher incidence in males than females.1 ALL can occur throughout one’s lifetime, although the majority (54.2%) of cases are diagnosed in patients under age 20 years, with a peak incidence between age 3 and 5 years.1 With the current treatment options, approximately 90% of children with ALL can be cured.1,2 By contrast, the cure rate in adults is less than 50%, with the highest overall percentage of deaths caused by ALL occurring in patients between age 65 and 74 years.1,3
Particularly in adults with ALL, new treatment options are needed to improve survival. Blinatumomab represents a novel approach to the treatment of B-cell precursor ALL (B-ALL), by activating the patient’s own immune system. With its unique mechanism of action, blinatumomab can avoid the mechanisms of resistance associated with chemotherapy.
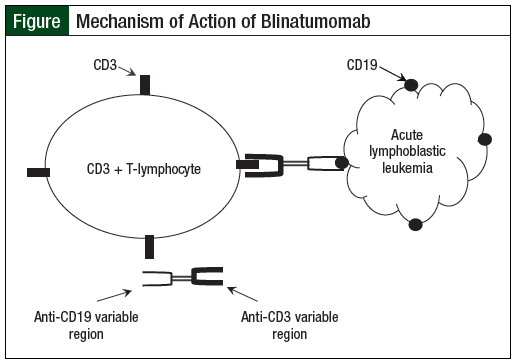
Mechanism of Action
ALL can be related to the patient’s B-cells or T-cells, with B-ALL representing the vast majority of cases.2 Many cell surface antigens, including CD19, CD20, CD22, and CD33, are expressed on the surface of B-ALL blasts. Because the B-cell lineage surface antigen CD19 is expressed on the surface of nearly all B-ALL blasts and plays a major role in establishing intrinsic B-cell–signaling thresholds, CD19 represents a particularly attractive therapeutic target for immunotherapy.4 Blinatumomab is a bispecific T-cell engager recombinant protein construct with dual affinity that binds to CD3 (expressed on the surface of all T-cells) and CD19 (expressed on the surface of normal B-lymphocytes and B-ALL blasts.5 With a molecular weight of approximately 54 kDa, blinatumomab is much smaller than an intact immunoglobulin (Ig)G monoclonal antibody, which typically has a molecular weight of 150 kDa.5 This difference in molecular weight gives blinatumomab a different pharmacokinetic profile compared with an intact IgG monoclonal antibody.
Blinatumomab, a unique recombinant protein, is produced in Chinese hamster ovary cells from genetic constructs encoding for the variable regions of murine anti-CD3 and anti-CD19 monoclonal antibodies. By binding to CD3 on T-cells and to CD19 on B-cells, blinatumomab activates endogenous CD3-positive cytotoxic T-cells to recognize and destroy CD19-positive ALL blasts (Figure). When binding to CD3, blinatumomab causes a transient proliferation of T-cells, a transient release of inflammatory cytokines, and mediates the production of cytotoxic proteins, all of which result in lysis of CD19-positive cells. This consequent T-cell–mediated lysis causes lysis of malignant B-cells, as well as of normal B-lymphocytes.5
Mechanisms of Resistance
Based on research and clinical observations, several possible mechanisms of resistance to blinatumomab have emerged.6-10 The proposed mechanisms of resistance include loss of CD19 expression, upregulation of programmed-cell death ligand 1 (PD-L1), elevated levels of regulatory T-cells (Tregs), and relapse in extramedullary sites. Several studies have documented the initial positive expression of the CD19 surface antigen that became negative in patients whose disease relapsed after treatment with blinatumomab.6-10 CD19 relapses occurred in approximately 20% of patients treated with blinatumomab.6-8,10
This shift from CD19-positive to CD19-negative ALL after relapse has been observed in 2 situations: (1) a myeloid lineage shift in patients with mixed-lineage leukemia during blinatumomab therapy,9 and (2) an unidentified molecular event driving resistance, as evidenced by a recent analysis of 4 CD19-negative ALL relapses after treatment with blinatumomab.10
Expression of PD-L1 on tumor cells has been associated with poor outcomes, because its upregulation on tumor cells has been shown to inhibit immune responses by causing T-cell dysfunction.11-12 Because blinatumomab exerts its effects by activating endogenous cytotoxic T-cells, upregulation of PD-L1 on ALL blasts has been proposed as a potential mechanism of resistance, suggesting that it could blunt or prevent the activation of a patient’s T-cells. Köhnke and colleagues reported a case of this proposed mechanism of resistance in an adult with relapsed or refractory B-ALL receiving treatment with blinatumomab.12
Tregs play a significant role in downregulating the immune response by secreting interleukin (IL)-10 and transforming growth factor-beta when activated.13 Duell and colleagues examined the role of Tregs in predicting the outcome of immunotherapy with blinatumomab in patients with relapsed or refractory ALL.14 They found that patients with a large number of Tregs were less likely to respond to blinatumomab therapy versus those with fewer Tregs. Although it is not standard practice to preemptively measure patients’ Tregs and/or deplete patients with elevated Tregs before initiating therapy with blinatumomab, these findings suggest that upfront removal of patients’ Tregs may restore proliferation of T-cells, improve response to blinatumomab therapy, and convert potential nonresponders to responders.14 However, these data are purely ex vivo and further validation of this technique is needed.
A final observed limitation for blinatumomab is relapse of ALL in extramedullary sites, such as the cerebrospinal fluid and testis.6 Relapse in these sites may reflect occult disease in sanctuary sites that are not penetrated by blinatumomab and/or T-cells.
Pharmacokinetics and Pharmacodynamics
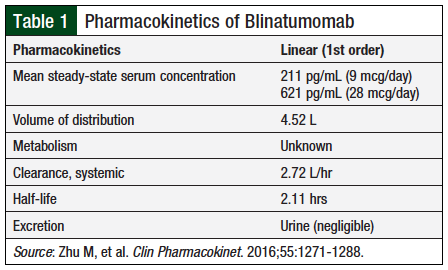
Zhu and colleagues conducted a comprehensive analysis of blinatumomab’s pharmacokinetics using data from 386 adults enrolled in 6 phase 1 or phase 2 clinical trials of non-Hodgkin lymphoma (NHL), relapsed or refractory ALL, or ALL with minimal residual disease (MRD).15 The analysis demonstrated that the pharmacokinetics of blinatumomab in adults are linear over a dosage range of 0.5 to 90 mcg/m2 daily (equivalent to doses of 9 to 162 mcg) and undergo fast clearance from the body.15 Considering the fast clearance blinatumomab exhibits, continuous infusion during 4 to 8 weeks best maintains effective concentrations to achieve adequate efficacy. When administered as a continuous infusion over 4 weeks per cycle in the study, the estimated mean volume of distribution was 4.52 L, the half-life was approximately 2.11 hours, and the estimated mean systemic clearance was 2.72 L per hour (Table 1).15
Similar to other small protein therapeutics, blinatumomab is catabolized into small peptides and amino acids and cleared from the circulation.15 Creatinine clearance (CrCl) is not a significant covariate of drug clearance, and no dose adjustment is currently recommended for patients with mild-to-moderate renal dysfunction (CrCl ≥30 mL/min). Because blinatumomab is not hepatically metabolized, dose adjustment based on hepatic function (defined by alanine transaminase or aspartate aminotransferase levels) does not appear to be necessary. Based on the study by Zhu and colleagues, blinatumomab exhibits a low probability of developing neutralizing antibodies, with fewer than 1% of patients developing them in their study.15
Zhu and colleagues also sought to identify factors that may alter the pharmacokinetics of blinatumomab, including disease type, baseline disease characteristics, and patient demographics.15 The results of their analysis suggest that the pharmacokinetics of blinatumomab are not influenced by disease type (ie, NHL, relapsed or refractory ALL, ALL with MRD), baseline disease characteristics (ie, Eastern Cooperative Oncology Group performance status, T-cell counts, or B-cell counts), or patient demographics (ie, age, sex, weight, or body size).15
Transient effects exerted by blinatumomab included upregulation of cell-adhesion molecules, release of inflammatory cytokines, proliferation of T-cells, production of cytolytic proteins, and elimination of CD19-positive B-cells, which ultimately result in T-cell redistribution, activation, and expansion; B-cell depletion; and transient cytokine elevation. At the dosage range described above, the pharmacodynamic profile does not differ on the basis of disease type.15
After initiation of a continuous infusion, when a new dose was given, or when a dose was escalated, peripheral CD3-positive T-cells declined briefly and quickly recovered to baseline values (a process known as T-cell redistribution).15 At doses of 5 mcg/m2 daily or more, a rapid decline in peripheral CD19-positive B-cells was observed, and this phenomenon appeared to be dose-dependent, considering that B-cell depletion was faster at higher doses of blinatumomab. Incomplete B-cell depletion was observed at doses of 0.5 to 1.5 mcg/m2 daily, as well as at higher doses in patients whose disease did not respond to treatment. Cytokine elevation occurred rapidly with the initiation of blinatumomab treatment, peaking on day 1, with a return to baseline by day 2. Cytokines IL-10, IL-6, and interferon (IFN)-gamma showed a substantial elevation, whereas cytokines tumor necrosis factor (TNF)-alpha and IL-2 showed a small elevation. Zhu and colleagues found a high level of interpatient variability in terms of cytokine levels. Higher initial doses of blinatumomab may be associated with a higher magnitude of cytokine elevation. This elevation might also be associated with tumor load (ie, B-cell counts).15
Zhu and colleagues also investigated the effect of blinatumomab on QT prolongation and the potential for drug–drug interactions.15 QT prolongation was not observed with increasing serum concentrations of blinatumomab.15 As a small protein, blinatumomab has limited distribution intracellularly into cardiac myocytes, and therefore likely does not directly inhibit potassium channels. Based on an in vitro assay with human hepatocytes, blinatumomab did not directly affect the activity of cytochrome (CY) P450 enzymes; however, possible drug interactions may exist because, as noted, blinatumomab causes a transient increase in cytokines, some of which may suppress CY enzyme activities.15
Clinical Trials
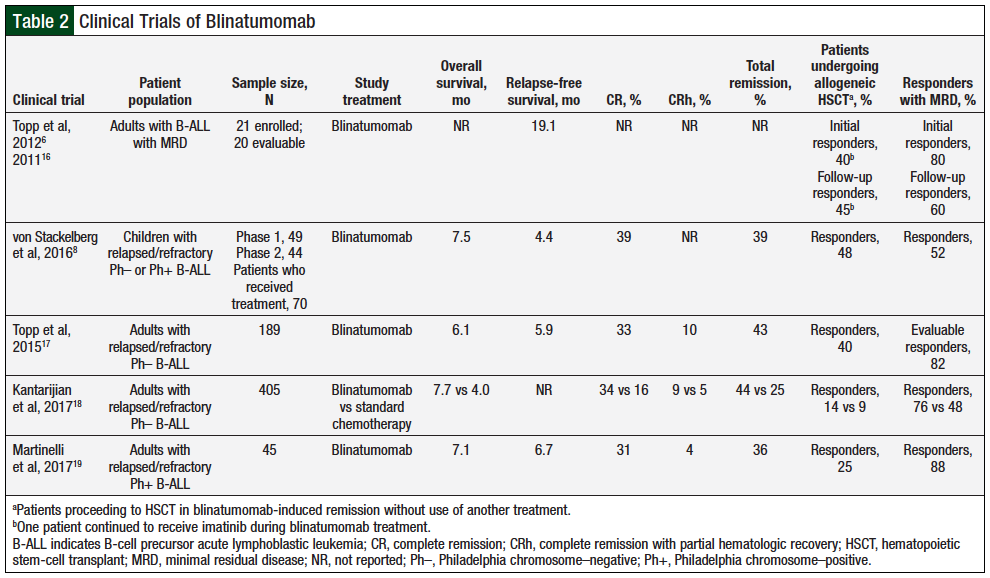
To date, several phase 1 and phase 2 clinical trials and 1 phase 3 clinical trial examining the safety and efficacy of blinatumomab in relapsed or refractory B-ALL have been conducted (Table 2).6,8,16-19 Based on the results of the phase 2 clinical trial data,17 in December 2014, the US Food and Drug Administration (FDA) granted accelerated approval for blinatumomab for the treatment of Philadelphia chromosome (Ph)-negative (Ph–) relapsed or refractory B-ALL in adults and pediatric patients. And in July 2017, the FDA extended the accelerated approval to full approval based on the results of the phase 3 clinical trial TOWER.18 At the same time, the FDA also expanded blinatumomab’s indication to include the treatment of Ph-positive (Ph+) relapsed or refractory B-ALL in adults and pediatric patients, supported by data from the phase 2 clinical trial ALCANTARA.19
MRD that reappears or persists after treatment is an indicator of resistance to chemotherapy. In an open-label, multicenter, single-arm, phase 2 clinical trial, blinatumomab was evaluated to determine its efficacy in 21 adults with MRD-positive B-ALL.6,16 This clinical trial also evaluated secondary end points, including the safety and tolerability, pharmacokinetics, and pharmacodynamics of blinatumomab. Patients received blinatumomab given as a continuous intravenous (IV) infusion at a dose of 15 mcg/m2 daily for 4 weeks, followed by a 2-week treatment-free period (1 cycle). Notably, this dosing regimen differed from that of other clinical trials by not using a stepwise dosing regimen. Patients completing at least 1 treatment cycle (N = 20) were included in the analysis.6,16
Of these 20 patients, 16 (80%) achieved an MRD response (<1 × 10–4), and at long-term (33 months) follow-up, 12 patients were still in complete remission (CR). Of the 20 patients, 9 (45%) had an allogeneic hematopoietic stem-cell transplant (HSCT) after treatment with blinatumomab, and at 33-month follow-up, 6 of those 9 patients were still in hematologic CR, with a 65% rate of relapse-free survival. Of the 11 patients who did not undergo allogeneic HSCT, 6 were in ongoing hematologic CR, with 60% relapse-free survival at 31-month follow-up. One patient had a seizure during the first cycle, which led to the permanent discontinuation of blinatumomab, and was not evaluable for response.6,16 These favorable preliminary results led to a larger phase 2 clinical trial in relapsed or refractory Ph– B-ALL.17
A larger phase 2 clinical trial with 189 patients sought to assess the activity and safety profile of blinatumomab in primary relapsed or refractory Ph– B-ALL.17 In this international, multicenter, single-arm, open-label, phase 2 study, patients received blinatumomab in a stepwise dosing regimen (9 mcg daily for the first 7 days, then 28 mcg daily for 21 days) by continuous IV infusion, followed by 2 weeks of rest every 6 weeks (1 cycle). Patients included in this trial were refractory to primary induction therapy, relapsed within 12 months of first remission, relapsed within 12 months of receiving an allogeneic HSCT, or were nonresponsive to, or relapsed after, first salvage therapy or beyond. The primary end point was CR or CR with partial hematologic recovery (CRh) occurring within the first 2 cycles.17
After 2 cycles, 43% of patients had achieved CR (33%) or CRh (10%). The secondary end points included relapse-free survival, overall survival, proportion of responders undergoing allogeneic HSCT after achieving blinatumomab-induced CR or CRh, 100-day mortality after allogeneic transplantation and the incidence and severity of adverse events. MRD response was noted as a prespecified exploratory end point. For the 43% of responders, the median relapse-free survival was 5.9 months (6.9 months for patients in CR and
5.0 months for patients in CRh), with a median follow-up of 8.9 months. For all 189 patients included in the study, the median overall survival was 6.1 months. Of patients achieving CR or CRh, 40% (32) of patients proceeded to allogeneic HSCT while still in remission; of these patients, 5 had received an allogeneic HSCT before entering the study. The 100-day mortality rate after transplantation was 11%.17
In terms of the exploratory secondary end point of MRD response, 82% of the evaluable study population achieved a reduction in MRD to less than 10–4, and those who reached an MRD status had a longer relapse-free survival (6.9 months) and overall survival (11.5 months) versus those who did not achieve MRD response (2.3 months and 6.7 months, respectively).17
Based on the accelerated FDA approval, a prospective, randomized, multi-institutional phase 3 clinical trial, the TOWER, was conducted to evaluate the efficacy and safety of blinatumomab versus standard chemotherapy in heavily pretreated adults with Ph– B-ALL.18 Patients were enrolled if their disease was refractory to primary induction therapy or to salvage chemotherapy, in first relapse, with the first remission lasting less than 12 months, in second or greater relapse, or in relapse after allogeneic HSCT. Patients were assigned in a 2:1 ratio to receive blinatumomab or a standard chemotherapy regimen of the investigator’s choice. In all, 405 patients were randomized based on age, previous salvage therapy, and previous allogeneic HSCT. Of the 405 patients, 376 received open-label treatment. All patients were included in the efficacy analyses, and all patients who received at least 1 dose of the study treatment were included in the safety analyses. The study primary end point was overall survival, which was significantly greater in the blinatumomab group, with a median overall survival of 7.7 months versus 4.0 months in the chemotherapy group. Secondary end points included CR, CRh, or CR with incomplete hematologic recovery (CRi) within 12 weeks, event-free survival, MRD remission, rate of allogeneic HSCT, and adverse event rates.18
Blinatumomab resulted in better outcomes versus chemotherapy in all secondary efficacy end points. With respect to 12-week remission rates, patients who received blinatumomab achieved CR, CRh, or CRi at a higher rate than those who received standard chemotherapy (CR, 34% vs 16%, respectively; CR/CRh/CRi, 44% vs 25%, respectively). Of the patients achieving CR, CRh, or CRi, 76% of those receiving blinatumomab achieved MRD-negativity versus 48% of patients in the chemotherapy group. Event-free survival was higher for those receiving blinatumomab than those receiving chemotherapy, as was median duration of remission (7.3 vs 4.6 months, respectively). Overall, 24% of patients in each blinatumomab group proceeded to allogeneic HSCT; however, 14% of these patients proceeded to transplant without the use of another treatment compared with only 9% of the patients in the chemotherapy group.18
Most recently, the results of the phase 2 clinical trial assessing the efficacy of blinatumomab for the treatment of relapsed or refractory Ph+ B-ALL were published.19 The ALCANTARA clinical trial was an open-label, single-arm, multicenter, phase 2 study of blinatumomab in 45 patients with relapsed or refractory Ph+ B-ALL who had relapsed after, or were refractory to, at least one second-generation or later tyrosine kinase inhibitor (TKI) or were intolerant to second-generation or later TKIs and intolerant or refractory to imatinib. Of these 45 patients, 36% achieved CR or CRh within the first 2 cycles (the primary end point). Key secondary end points included MRD response rate during the first 2 treatment cycles, relapse-free survival, overall survival, and allogeneic HSCT after blinatumomab-induced remission. Of the patients achieving CR or CRh, 88% also achieved a complete MRD response. The median relapse-free survival was 6.7 months and the median overall survival was 7.1 months. Four (25%) of the patients with a continuous blinatumomab-induced remission proceeded to allogeneic HSCT, with a 100-day posttransplant mortality rate of 25% after receiving blinatumomab only. These data indicate the efficacy of treating relapsed or refractory Ph+ B-ALL patients with blinatumomab.19
To assess the potential use of blinatumomab in pediatric patients, von Stackelberg and colleagues conducted a multicenter, single-arm, open-label phase 1 and phase 2 study to assess safety, to determine the maximum tolerated dosage, and to evaluate the complete remission rate within the first 2 cycles of blinatumomab treatment in children younger than 18 years with Ph+ or Ph– relapsed or refractory B-ALL.8 During phase 1, 49 patients received dosages of 5, 15, or 30 mcg/m2 daily or a stepwise dosing regimen of 15/30 mcg/m2 daily (ie, 15 mcg/m2 daily for the first 7 days, followed by 30 mcg/m2 daily thereafter). The recommended dosage was a stepwise dosage of 5 mcg/m2 daily for the first 7 days and 15 mcg/m2 daily for 21 days, followed by 2 weeks of recovery; after this determination was made, 26 additional patients received treatment to further assess the pharmacokinetics and safety of blinatumomab before progressing to phase 2. In total, 70 patients (ie, 26 in phase 1 and 44 in phase 2) received treatment at the recommended stepwise 5/15-mcg/m2 daily dosing regimen. Of these patients, 39% achieved CR within the first 2 cycles (primary outcome for phase 2), with 52% of those responders achieving a complete MRD response. Among the responders, 48% underwent allogeneic HSCT after blinatumomab-induced remission. The median relapse-free survival for responders was 4.4 months and the median overall survival for all 70 patients who received the recommended dosages was 7.5 months.8
To date, allogeneic HSCT remains the only known curative treatment for patients with relapsed or refractory ALL. A concern with the use of blinatumomab before allogeneic HSCT is blinatumomab’s activation of T-lymphocytes, which could increase morbidity and mortality associated with transplant, particularly in terms of increasing the incidence and intensity of graft-versus-host disease (GVHD). Studies in adults and in children indicate that an allogeneic HSCT is feasible after treatment with blinatumomab,8,16-19 and the 100-day mortality with blinatumomab before transplant is similar to salvage chemotherapy before transplant.20 With relapse-free survival of more than 7 months, blinatumomab provides additional time for patients who need to identify a suitable matched donor for allogeneic HSCT, without significantly increasing the incidence of GVHD; however, further study is needed.
Safety and Tolerability
As in most immunotheries, the more common adverse events observed with blinatumomab include flulike symptoms, such as pyrexia, headache, chills, and fatigue, as well as neutropenia, febrile neutropenia, and infection. The prescribing information for blinatumomab includes a black box warning of 2 types of serious and potentially fatal adverse reactions—cytokine release syndrome (CRS) and neurologic toxicities.21
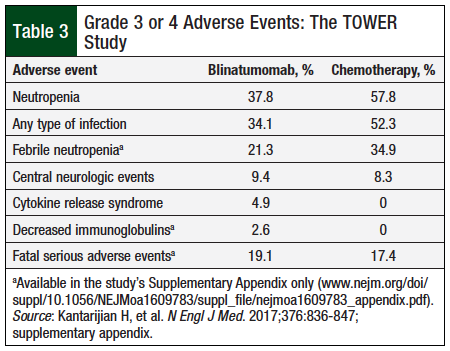
In the TOWER clinical trial, 99% of patients who received blinatumomab and 99% of patients who received chemotherapy had an adverse event. Grade 3 or 4 adverse events occurred slightly more frequently in the chemotherapy arm (91.7%) versus the blinatumomab arm (86.5%), whereas fatal, serious adverse events occurred slightly more often in the blinatumomab arm (19.1%) versus the chemotherapy arm (17.4%) (Table 3).18
CRS is an immediate and potentially fatal complication linked to blinatumomab. This adverse reaction develops shortly after initiating the first dose, and patients can have an infusion-related reaction with symptoms that include fever, headache, nausea, hypotension, weakness, and fatigue. The mechanism of this potentially fatal cytokine release is mediated through a severe inflammatory response, including vascular leak, hypotension, pulmonary edema, and coagulopathy, which ultimately results in multi-organ system failure.21,22
To investigate these reactions further, concentrations of selected cytokines that are often released by activated T-lymphocytes were measured in serum samples taken from patients with MRD-positive ALL in a study that focused on the patients’ immuno-pharmacologic response to blinatumomab; the patients did not receive dexamethasone as a premedication. Although vast patient variability existed, cytokines detected during the first 2 days of the start of infusion included IL-2, IL-4, IL-6, IL-10, IFN-gamma, and TNF-alpha.23
This study demonstrated that after the first 2 days of therapy initiation, cytokine levels began to decline quickly and were minimally detectable later in treatment, and generally returned to baseline by the end of the first week of treatment. Of note, on initiation of the subsequent cycles of therapy, this cytokine release was not detectable, with the exception of IL-10 elevation in the second and third cycles, although of a reduced magnitude than its elevation in cycle 1. This massive release of cytokines has also been studied in relation to the initial starting dose that patients receive, and it has been shown that patients who start with step-dose therapy have lower cytokine release (ie, changes in clinical lab) at the 2 dose intensities, making this the preferable dosing strategy.23
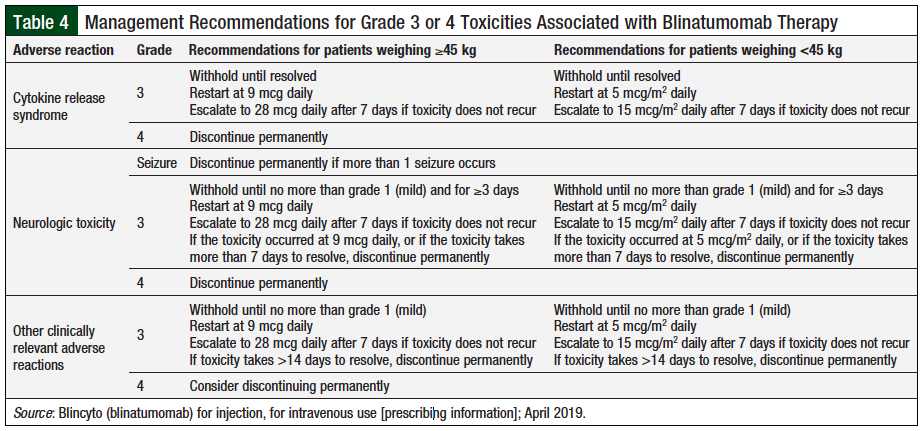
Although this adverse effect is considerable, it has been shown to be manageable. The manufacturer of blinatumomab recommends that the drug be discontinued in patients with grade 4 CRS (Table 4)21; however, a case study reported an adult patient who was successfully restarted with blinatumomab therapy after grade 4 CRS.23 After the patient had stabilized and recovered, the decision was made to reinitiate therapy, but with an additional step in dosing to titrate slower, and more frequent lab monitoring was performed during therapy. This patient was able to complete 5 cycles of blinatumomab therapy without further issue and ultimately achieved complete molecular remission.23 It is important to note that fatal adverse effects can be avoided in patients with any grade CRS, by discontinuing the infusion, administering IV tocilizumab (8 mg/kg), and/or corticosteroids.22
Some degree of cytokine release would be expected as a result of blinatumomab’s mechanism of T-cell activation; furthermore, it has been postulated to correlate with drug efficacy; however, studies have not proved this theory. The magnitude of this CRS has not been shown to correspond to the targeting of B-cells in the peripheral blood or bone marrow, nor has it been shown to correlate with the patient’s clinical response to blinatumomab.24 With early recognition and swift initiation of IV tocilizumab therapy, patients are able to recover from this life-threatening side effect. More studies are needed to evaluate the role of genetic testing to identify patients at high risk for CRS.
Of all the adverse events exhibited by blinatumomab therapy, neurologic toxicity is the most puzzling, because the drug’s mechanism of action does not suggest this possibility. This serious and potentially fatal adverse reaction can manifest as headaches, tremor, speech disorders, confusion, loss of balance, seizures, and may eventually progress to encephalopathy. In the TOWER clinical trial, only 9.4% of patients had a neurologic event of grade 3 or 4.18 However, in the ALCANTARA clinical trial, 47% of all the patients had some degree of neurologic insult (7% at grade 3 or 4, most often paresthesia and confusion).19 von Stackelberg and colleagues found neurologic adverse events to be rare in the pediatric population, in comparison with adult data, and no patients discontinued treatment because of an associated neurologic side effect.8 If such a side effect is recognized early, the blinatumomab infusion should be discontinued, dexamethasone administered, and the adverse reaction may be reversible. For these reasons, the drug manufacturer recommends hospitalization for the initiation of infusion and any subsequent dose increase.21
Dosing Recommendations
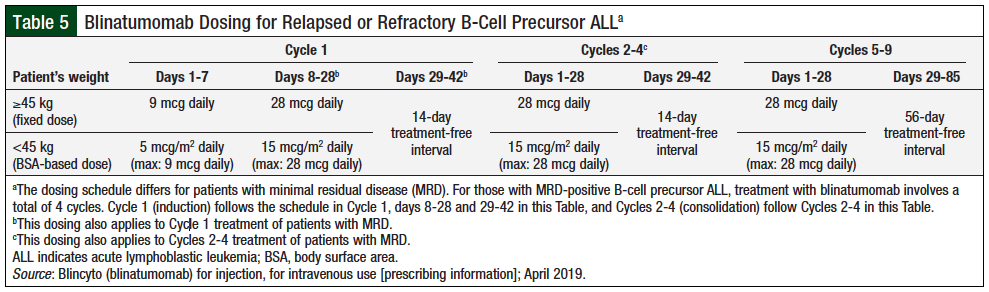
Based on the results of, and recommendations from, the clinical trials, patients diagnosed with relapsed or refractory B-ALL should receive the first cycle of blinatumomab induction as a 28-day infusion, followed by a 14-day treatment-free interval, for a total of 42 days.18,21 A treatment course consists of up to 2 cycles of induction followed by an additional 3 cycles for consolidation treatment, for a total of 5 cycles. Patients may receive continued therapy for an additional 4 cycles (Table 5).21 For patients initiating treatment, hospitalization is recommended for the first 9 days of the first cycle, the first 2 days of the second cycle, and if treatment is interrupted for more than 4 hours. In addition, it is recommended that adults receive 20 mg of dexamethasone, or dexamethasone 5 mg/m2 in pediatric patients, 1 hour before infusion of the first dose for each cycle, before any escalation in dose, and before reinitiating treatment if it was interrupted for more than 4 hours.21
In the event of adverse events, specific protocols for discontinuing blinatumomab therapy or modifying the dose, depending on the severity of the reaction, have been developed (Table 4).21 It should be noted, however, that if the interruption after an adverse event lasts no more than 7 days, the patient should resume the same cycle in a hospital setting for a total of 28 days of infusion, including the days before and after the interruption in that particular cycle. If an interruption as a result of an adverse event lasts more than 7 days, the patient should begin a new cycle.21
Cost of Therapy
Although blinatumomab therapy demonstrates a significant improvement in overall survival, relapse-free survival, and response rates versus chemotherapy, the cost of treatment remains a significant issue. One package of the drug contains a 35-mcg vial of blinatumomab, a 10-mL vial of IV solution stabilizer required to prevent absorption of the drug into the infusion bag, and tubing used for administration. The average wholesale cost of each package ranges from approximately $3464 to $3815.25,26 With the requirement of up to 1 vial daily for 28 days, the approximate cost for 1 cycle of blinatumomab would be between $97,000 and $107,000. Considering that patients may receive up to 5 cycles during a full treatment course, the drug acquisition costs alone could reach approximately $535,000. When evaluating treatment options, we must keep in mind that this estimate does not include additional expenses, such as hospitalization costs or supportive care, nor does it factor in productivity loss from workdays lost for the patient or the caregiver.25,26
In October 2015, the Centers for Medicare & Medicaid Services (CMS) announced their decision to make payment exceptions for blinatumomab as “new technology” to hospitals that are treating patients with relapsed or refractory B-ALL whose treatment costs did not fit into Medicare’s standard lump-sum reimbursements. CMS’s decision was swayed after additional clinical trials were presented in an effort to demonstrate blinatumomab’s efficacy versus chemotherapy. This decision is important for increasing access to healthcare, which will improve patients’ response rates, relapse-free survival, and overall survival.27
Considering the high acquisition costs of blinatumomab, Delea and colleagues evaluated the cost-effectiveness of blinatumomab versus standard chemotherapy using results from the TOWER study.25 They found higher total costs (ie, drug acquisition cost, administration, use of subsequent allogeneic HSCT, additional salvage therapy, and terminal care) with blinatumomab ($395,094) versus standard chemotherapy ($214,452).25 The major difference in this total cost was the drug cost of blinatumomab versus chemotherapy. When we factor in quality-adjusted life-years (QALY), because blinatumomab does improve survival, blinatumomab is estimated to cost approximately $110,108/QALY, which is below the threshold accepted for other cancer treatments of $150,000/QALY.25,28,29 The pharmacoeconomic value of blinatumomab therapy is derived from the increased overall survival, as well as increased health-related quality-of-life benefit over standard chemotherapy.25
Conclusion: Future Outlook
During the past 2 decades, increased insight into, and understanding of, the pathogenesis of ALL, coupled with the identification of specific targets for therapy, have led to the development of novel therapeutic options resulting in significant advances in the treatment of ALL and, ultimately, improved outcomes. In the past 2 decades, several new therapies have been approved by the FDA for ALL, providing more treatment options beyond chemotherapy, and many of these agents have become standard treatment options for patients with relapsed or refractory B-ALL.30
Although many innovative treatment options exist based on patient-specific characteristics, such as age and presence or absence of the Ph, blinatumomab is unique in its ability to cover all these spectrums and has been granted approval for use in adults and pediatric patients, and in patients with Ph+ or Ph–. It also is approved as a single agent and is not required to be used in conjunction with chemotherapy.21 The 2018 National Comprehensive Cancer Network (NCCN) guidelines recommend blinatumomab as a preferred treatment for patients with relapsed or refractory Ph– B-ALL. The NCCN guidelines also recommend blinatumomab therapy for patients with Ph+ B-ALL after failure of 2 TKIs.30
With its mechanism of action requiring T-lymphocytes, combining blinatumomab with myelosuppressive chemotherapy should likely be avoided. Using blinatumomab sequentially with myelosuppressive chemotherapy may be a rational way to incorporate blinatumomab earlier in therapy. In addition, blinatumomab could be used for molecular relapse after allogeneic HSCT to augment donor lymphocyte infusions.31 Although currently only FDA approved for the treatment of relapsed or refractory B-ALL, in the future, blinatumomab may provide extended therapeutic options for other CD19-positive diseases, such as multiple myeloma and NHL.32 In addition, several studies evaluating the use of blinatumomab in newly diagnosed B-ALL and in combination with other targeted therapies are currently ongoing, and the results of these trials may further delineate blinatumomab’s place in therapy.
Author Disclosure Statement
Dr Drawdy, Dr Jones, and Dr Hall have no conflicts of interest to report.
References
- National Cancer Institute. Cancer stat facts: leukemia - acute lymphocytic leukemia (ALL). https://seer.cancer.gov/statfacts/html/alyl.html. Accessed April 29, 2019.
- Hunger SP, Mullighan CG. Acute lymphoblastic leukemia in children. N Engl J Med. 2015;373:1541-1552.
- Jabbour E, O’Brien S, Konopleva M, Kantarjian H. New insights into the pathophysiology and therapy of adult acute lymphoblastic leukemia. Cancer. 2015;121:2517-2528.
- Raponi S, De Propris MS, Intoppa S, et al. Flow cytometric study of potential target antigens (CD19, CD20, CD22, CD33) for antibody-based immunotherapy in acute lymphoblastic leukemia: analysis of 552 cases. Leuk Lymphoma. 2011;52:1098-1107.
- Przepiorka D, Ko C-W, Deisseroth A, et al. FDA approval: blinatumomab. Clin Cancer Res. 2015;21:4035-4039.
- Topp MS, Gökbuget N, Zugmaier G, et al. Long-term follow-up of hematologic relapse-free survival in a phase 2 study of blinatumomab in patients with MRD in B-lineage ALL. Blood. 2012;120:5185-5187.
- Kantarjian H, Jabbour E, Topp MS. Blinatumomab for acute lymphoblastic leukemia. N Engl J Med. 2017;376:e49.
- von Stackelberg A, Locatelli F, Zugmaier G, et al. Phase I/phase II study of blinatumomab in pediatric patients with relapsed/refractory acute lymphoblastic leukemia. J Clin Oncol. 2016;34:4381-4389.
- Zoghbi A, Winkler B, zur Stadt U, et al. Lineage switch under blinatumomab of a relapsed common ALL co-expressing myeloid markers without MLL rearrangement. Blood. 2016;128:5196.
- Braig F, Brandt A, Goebeler M, et al. Resistance to anti-CD19/CD3 BiTE in acute lymphoblastic leukemia may be mediated by disrupted CD19 membrane trafficking. Blood. 2017;129:100-104.
- Shi L, Chen S, Yang L, Li Y. The role of PD-1 and PD-L1 in T-cell immune suppression in patients with hematological malignancies. J Hematol Oncol. 2013;6:74-79.
- Köhnke T, Krupka C, Tischer J, et al. Increase of PD-L1 expressing B-precursor ALL cells in a patient resistant to the CD19/CD3-bispecific T cell engager antibody blinatumomab. J Hematol Oncol. 2015;8:111.
- Miyara M, Sakaguchi S. Natural regulatory T cells: mechanisms of suppression. Trends Mol Med. 2007;13:108-116.
- Duell J, Dittrich M, Bedke T, et al. Frequency of regulatory T cells determines the outcome of the T-cell-engaging antibody blinatumomab in patients with B-precursor ALL. Leukemia. 2017;31:2181-2190.
- Zhu M, Wu B, Brandl C, et al. Blinatumomab, a bispecific T-cell engager (BiTE) for CD-19 targeted cancer immunotherapy: clinical pharmacology and its implications. Clin Pharmacokinet. 2016;55:1271-1288.
- Topp MS, Kufer P, Gökbuget N, et al. Targeted therapy with the T-cell–engaging antibody blinatumomab of chemotherapy-refractory minimal residual disease in B-lineage acute lymphoblastic leukemia patients results in high response rate and prolonged leukemia-free survival. J Clin Oncol. 2011;29:2493-2498.
- Topp MS, Gökbuget N, Stein AS, et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 2015;16:57-66. Erratum in: Lancet Oncol. 2015;16:e158.
- Kantarjian H, Stein A, Gökbuget N, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med. 2017;376:836-847.
- Martinelli G, Boissel N, Chevallier P, et al. Complete hematologic and molecular response in adult patients with relapsed/refractory Philadelphia chromosome–positive B-precursor acute lymphoblastic leukemia following treatment blinatumomab: results from a phase II, single-arm, multicenter study. J Clin Oncol. 2017;35:1795-1802.
- Kashyap A, Wingard J, Cagnoni P, et al. Intravenous versus oral busulfan as part of a busulfan/cyclophosphamide preparative regimen for allogeneic hematopoietic stem cell transplantation: decreased incidence of hepatic venoocclusive disease (HVOD), HVOD-related mortality, and overall 100-day mortality. Biol Blood Marrow Transplant. 2002;8:493-500.
- Blincyto (blinatumomab) for injection, for intravenous use [prescribing information]. Thousand Oaks, CA: Amgen; April 2019.
- Teachey DT, Rheingold SR, Maude SL, et al. Cytokine release syndrome after blinatumomab treatment related to abnormal macrophage activation and ameliorated with cytokine-directed therapy. Blood. 2013;121:5154-5157. Erratum in: Blood. 2016;128:1441.
- Marini BL, Sun Y, Burke PW, Perissinotti AJ. Successful reintroduction of blinatumomab in a patient with relapsed/refractory acute lymphoblastic leukemia following grade 4 cytokine release syndrome. J Oncol Pharm Pract. 2018;24:67-73.
- Klinger M, Brandl C, Zugmaier G, et al. Immunopharmacologic response of patients with B-lineage acute lymphoblastic leukemia to continuous infusion of T cell–engaging CD19/CD3-bispecific BiTE antibody blinatumomab. Blood. 2012;119:6226-6233.
- Delea TE, Amdahl J, Boyko D, et al. Cost-effectiveness of blinatumomab versus salvage chemotherapy in relapsed or refractory Philadelphia-chromosome-negative B-precursor acute lymphoblastic leukemia from a US payer perspective. J Med Econ. 2017;20:911-922.
- Marini BL, Wechter AR, Burke PW, et al. Minimizing waste during preparation of blinatumomab infusions. Am J Health Syst Pharm. 2016;73:19-20.
- Pear R. Medicare, reversing itself, will pay more for an expensive new cancer drug. August 8, 2015. www.nytimes.com/2015/08/09/us/medicare-reversing-it self-will-pay-more-for-an-expensive-new-cancer-drug.html. Accessed July 15, 2017.
- Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness—the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371:796-797.
- Hillner BE, Smith TJ. Efficacy does not necessarily translate to cost effectiveness: a case study in the challenges associated with 21st-century cancer drug pricing. J Clin Oncol. 2009;27:2111-2113.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Acute Lymphoblastic Leukemia. Version 1.2018. March 12, 2018. www.nccn.org/professionals/physician_gls/pdf/all.pdf. Accessed September 10, 2017.
- Thomas X. Blinatumomab: a new era of treatment for adult ALL? Lancet Oncol. 2015;16:6-7.
- Oak E, Bartlett NL. Blinatumomab for the treatment of B-cell lymphoma. Expert Opin Investig Drugs. 2015;24:715-724.