Background: Chemotherapy-induced diarrhea (CID) is a predictable yet undertreated side effect of several frequently used chemotherapy agents and can lead to delays in treatment and poor quality of life. Although the exact cause of CID is not completely understood, various theories point to a multifactorial process resulting in an imbalance between the absorption and the secretion of fluid in the gastrointestinal tract. Medications such as loperamide and diphenoxylate plus atropine are frequently used along with nonpharmacologic measures to treat mild CID.
Objectives: To discuss the currently recommended treatment for CID, as well as other potential medications for the treatment and the prevention of CID. With the emergence of new therapeutic alternatives for severe CID, an update of the current treatment options is warranted.
Discussion: Although guidelines exist for the treatment of CID, patient needs often exceed these recommendations. Through different mechanisms of action, medications such as corticosteroids, antibiotics, glutamine, palifermin, and activated charcoal have been studied for the prevention of CID. For patients with treatment-resistant CID, small clinical trials suggest that probiotics or octreotide long-acting release may be an effective alternative.
Conclusion: Further investigations should be conducted with promising therapies for validation before being recommended for guideline inclusion for the treatment of CID.
Diarrhea is a well-recognized side effect that is associated with various phases of a patient with cancer’s treatment cycle. Radiotherapy, chemotherapy, infection, and graft-versus-host disease can all potentially augment this dose-limiting toxicity. Some regimens, especially those targeting colorectal cancer (CRC) and other malignancies of the gastrointestinal (GI) tract, are associated with an increased incidence of severe or refractory chemotherapy-induced diarrhea (CID). In some studies, CID has been reported as a side effect in up to 82% of patients with cancer, with up to 33% experiencing grades 3 and 4 diarrhea.1 GI toxicity has also been linked to many cases of death and is often an underrecognized and undertreated complication of chemotherapy.2
Severe diarrhea resulting in dehydration, neutro-penia, fever, malnutrition, renal insufficiency, infectious complications, or severe electrolyte imbalances can lead to hospitalization.2 The presence of CID can influence providers to change chemotherapy agents, reduce treatment doses, delay therapy, or even to discontinue therapy, leading to potentially worsened clinical outcomes.2 A study by Arbuckle and colleagues demonstrated that grades 1 and 2 diarrhea may lead to an alteration in chemotherapy for 11% of patients, whereas approximately 45% of patients experiencing any-grade CID required dose reduction in chemotherapy.3,4 Although it is well known that CID can result in significant morbidity and mortality, no comprehensive treatment guidelines exist for historically used and newly evaluated medications for the treatment of CID.
Mechanism of Chemotherapy-Induced Diarrhea
The exact mechanism of CID is not completely understood; however, various theories point to a multifactorial process resulting in an imbalance between the absorption and the secretion of fluid in the GI tract.5 Other contributing factors, such as diet, concomitant medications, and infectious complications, can enhance diarrhea in patients with cancer.4 The frequency of CID varies based on the chemotherapy regimen and on the administration schedule.
Table 1
Mechanism of Fluorouracil-Induced Diarrhea
Up to 50% of patients treated with weekly 5-fluorouracil in combination with leucovorin experience CID.6 Fluorouracil therapy results in mitotic arrest and apoptosis of the crypt cells in the GI tract.5 Necrosis of this tissue enhances the imbalance of the ratio of immature secretory crypt cells to mature villus enterocytes.7 It also causes bowel wall inflammation, thereby stimulating additional secretion of fluid and electrolytes into the intestinal lumen5 and significantly altering the osmotic gradient in the GI tract, which contributes to the increased secretion of fluid into the stool.8
Mechanism of Irinotecan-Induced Diarrhea
Irinotecan, a cornerstone in the management of CRC, with a 2-pronged effect, can induce acute (within 24 hours) and delayed (2-14 days postadministration) diarrhea.6 Irinotecan is a prodrug converted into its active form, SN-38, both of which are released into the feces by hepatobiliary and intestinal secretions.4 SN-38 is inactivated in the liver to SN-38G. As it eventually reaches the intestinal lumen, SN-38G is transformed back into its active form by beta-glucuronidase, an enzyme secreted by intestinal microflora, causing direct mucosal damage and toxicity.4 Irinotecan also induces the production of prostaglandin E2 and thromboxane A2, inflammatory cytokines, and tumor necrosis factor alpha, all causing additional mucosal damage.4
Additional Drugs Causing Chemotherapy-
Induced Diarrhea
Other chemotherapeutic regimens have been associated with diarrhea, although at a considerably lower rate than either fluorouracil or irinotecan. Epidermal growth factor receptor (EGFR)-targeted therapies result in grade 3 or greater diarrhea in <10% of cases.6 Patients treated with EGFR tyrosine kinase inhibitors (eg, erlotinib, gefitinib, or lapatinib) experience diarrhea in up to 60% of cases, with grades 3 and 4 diarrhea occurring much less often.6 Unlike irinotecan- and fluorouracil-based regimens, EGFR therapies rarely need to be dose-reduced as a result of severe diarrhea.6 The mechanism of CID for these therapies has not been adequately investigated.6
Therapy with ipilimumab, a human monoclonal anti–cytotoxic T-lymphocyte antigen (CTLA)-4 antibody used for the treatment of metastatic melanoma, often results in GI and skin toxicities.9 Weber and colleagues estimated the incidence of grade 2 or greater diarrhea between 32% and 35%.10 Blockade of CTLA-4 in the GI tract causes dysregulation of the mucosal immune system, resulting in colitis and diarrhea.9 This mechanism of diarrhea is significantly different from fluorouracil or irinotecan and is often not adequately treated with conventional therapies.
Assessment of Chemotherapy-Induced Diarrhea
Currently, there is a lack of comprehensive standardized assessment methods for CID.5 The most frequently used criteria for categorizing CID is the Common Terminology Criteria for Adverse Events, which assesses patient symptoms on a scale of 1 to 5 (Table 1).8,11 CID is classified into 2 categories, complicated and uncomplicated. Uncomplicated diarrhea is defined as patients with grade 1 or 2 diarrhea and no additional signs or symptoms. Complicated CID is classified by patients with grade 3 or 4 diarrhea or patients with grade 1 or 2 diarrhea and 1 additional risk factor, such as moderate-to-severe cramping, grade 2 or greater nausea and vomiting, decreased performance status, fever, sepsis, neutropenia, frank bleeding, or dehydration.
Guideline-Recommended Therapy for Uncomplicated Chemotherapy-Induced Diarrhea
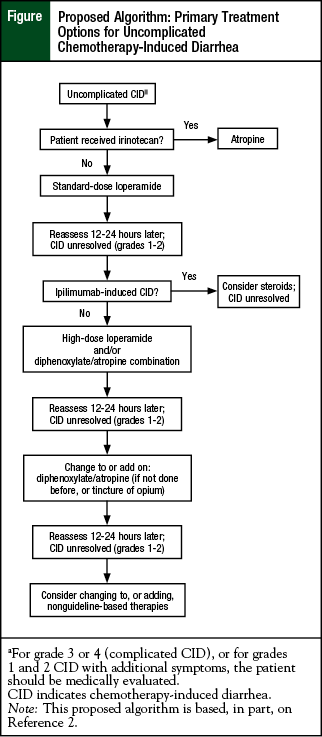
The last consensus conference for the management of CID was published in 1998,7 and the resulting guidelines were last updated in 2004.2 Loperamide, tincture of opium, and octreotide remain the only agents currently recommended by treatment guidelines, because of the lack of data supporting other therapies for CID (Figure).2
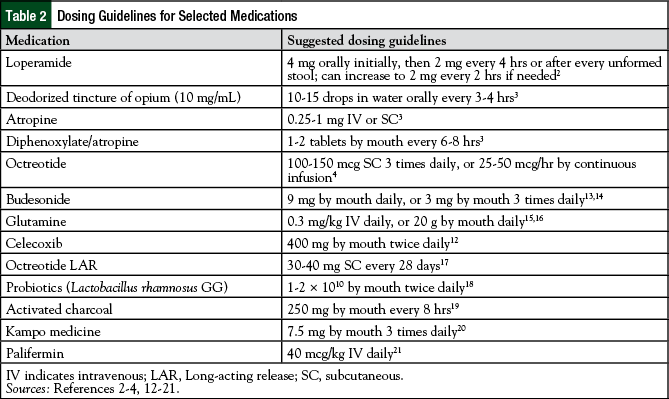
These guidelines recommend dietary modification, along with loperamide (4 mg initially, followed by 2 mg every 4 hours or after every unformed stool), as the standard first-line therapy for the treatment of uncomplicated CID (Table 2).2,4,12-21 Patients can discontinue loperamide therapy when they have been free of diarrhea for at least 12 hours.2
If diarrhea persists for more than 24 hours, high-dose loperamide (2 mg every 2 hours) is recognized as an appropriate therapeutic option in addition to initiating oral antibiotics for the prevention of infectious complications.2
If diarrhea persists for more than 48 hours with high-dose loperamide therapy, loperamide should be stopped and a second-line treatment should be considered when the patient is evaluated by a physician; options include subcutaneous (SC) octreotide, tincture of opium, or oral budesonide.2
Loperamide
Loperamide, a synthetic opiate derivative, is the initial drug of choice for CID; it has reduced the incidence of irinotecan-induced diarrhea from 80% to 9% in several studies.5,8 Loperamide acts as an antidiarrheal agent by exerting agonistic effects on opioid receptors in the GI tract, resulting in decreased peristalsis and increased fluid reabsorption.5 Loperamide is minimally absorbed and produces a limited side-effect profile.6 Although rare, loperamide can cause a paralytic ileus, and patients should be routinely monitored while using high-dose loperamide.5 Other side effects include abdominal pain, dry mouth, drowsiness, and dizziness.4 Although loper-amide has been proved to be extremely effective in uncomplicated diarrhea, its utility as monotherapy for severe diarrhea is limited.8,22
Tincture of Opium
Tincture of opium, like loperamide, works by slowing GI peristalsis and increasing intestinal transit time.4 No studies have specifically evaluated tincture of opium in the treatment of CID; however, it is frequently used as an antidiarrheal agent and can safely be used as a second-line therapy for refractory diarrhea.4 Common side effects are usually mild and include nausea and vomiting.4
Atropine
In the treatment of irinotecan-induced, acute-onset diarrhea, atropine monotherapy works as a competitive antagonist at anticholinergic receptors, typically dosed as 0.25 to 1 mg intravenous (IV) or SC.4 Grades 1 to 4 and grades 3 to 4 acute-onset diarrhea are typically seen in 51% and 8% of patients receiving irinotecan infusions, respectively.12 In a study by Yumuk and colleagues, 66 patients with metastatic CRC who received irinotecan were premedicated with 0.5 mg of SC atropine before their infusion. In a total of 444 infusions, acute-onset diarrhea was not seen in any of these patients.12
Figure 1
Diphenoxylate plus Atropine
There are little efficacy data supporting the use of diphenoxylate plus atropine compared with loperamide for the treatment of CID8; however, one double-blind study comparing these agents suggests that loperamide is the more effective agent.7 In one study of 614 patients who experienced acute diarrhea, the efficacy of loperamide was compared with diphenoxylate plus atropine.23 Patients were initially treated with loperamide 2 mg or with diphenoxylate 2.5 mg plus atropine 0.025 mg and were instructed to take an additional tablet after each unformed stool. Of patients in the loperamide group, 42% required 2 to 3 tablets to control diarrhea, whereas 2 to 3 tablets of diphenoxylate plus atropine controlled diarrhea in only 23% of patients. Diarrhea was controlled within 24 hours in 47% of patients in the loperamide group compared with 37% of patients in the diphenoxylate plus atropine group. In addition, within the 72-hour study period, fewer tablets of loperamide were required versus in the diphenoxylate plus atropine group (4.37 vs 5.75 tablets; P = .01).23
Diphenoxylate plus atropine can be used in combination with loperamide for the treatment of grade 1 or 2 diarrhea at a dosage of 1 to 2 tablets every 6 to 8 hours.4 Side effects include dry mouth, blurred vision, insomnia, and dyspepsia.4
Guideline-Recommended Therapy for Aggressive Chemotherapy-Induced Diarrhea
Standard- or high-dose loperamide therapy is often unsuccessful in the treatment of aggressive (complicated) CID in 9% to 30% of cases.7,22 Patients experiencing refractory grade 1 or 2 CID, as well as grade 3 or 4 CID can be treated more aggressively with SC octreotide acetate (100-150 mcg 3 times daily).2 The octreotide dose can be titrated until the symptoms of diarrhea are under control.2 In addition, patients should receive fluids and antibiotic therapy according to the guidelines.2 Hospitalization is often required for patients who are unable to be adequately rehydrated orally or those who have other complicating symptoms.1 Antidiarrheal treatment should be continued until the patient is symptom free for at least 24 hours.2 Patients experiencing grade 2 or greater diarrhea before a scheduled chemotherapy session should have treatment suspended until complete symptom resolution for at least 24 hours.4
Octreotide
Octreotide is a synthetic somatostatin analogue that regulates intestinal fluid and electrolyte transport.24 Octreotide inhibits the secretion of hormones in the gut, including serotonin, gastrin, insulin, and secretin.24 Through these mechanisms, octreotide increases GI tran-sit time and reduces intestinal secretions.8 Numerous studies have displayed the effectiveness of short-acting SC octreotide for the treatment of CID.25 In a study by Gebbia and colleagues, patients were treated with 500 mcg of SC octreotide 3 times daily compared with oral loperamide 4 mg 3 times daily in patients with grade 3 or 4 diarrhea.26 Complete resolution of diarrhea was seen in 80% of patients receiving octreotide compared with only 30% of patients treated with loperamide (P <.001).26
A dosage of SC octreotide 100 to 150 mcg 3 times daily has been shown in clinical trials to reduce grades 3 and 4 CID by 60% to 95%.1 As of the last revision (in 2004), the guidelines recommend a dose of 100 to 150 mcg of SC octreotide 3 times daily or 25 to 50 mcg hourly by continuous infusion.2 This dose can be increased up to 0.5 mg 3 times daily until diarrhea is under control.2
A study by Goumas and colleagues evaluated 100 mcg versus 500 mcg of SC octreotide in patients with grades 3 or 4 diarrhea after loperamide failure.27 Patients treated with 500 mcg had significantly more symptom control than those treated with the 100-mcg dose (approximately 90% vs 61%, respectively; P <.05), with a similar side-effect profile.2 Drawbacks for the use of octreotide include the administration of SC injection and a high cost profile.8 Side effects of SC octreotide include GI symptoms and, rarely, injection-site reactions.4
Antibiotics
Widespread inflammation and necrosis in the bowel predisposes patients to infections from opportunistic pathogens, especially if they are immunocompromised or neutropenic.5 Increased epithelial permeability, as well as a reduced immune system, enable microflora to translocate out of the GI tract, predisposing patients to potential life-threatening gram-negative sepsis.4 The guidelines suggest the initiation of antibiotics for patients who are experiencing diarrhea for more than 24 hours for the prevention of septic complications.2 An oral fluoroquinolone, such as ciprofloxacin, for 7 days, has been recommended by the Independent Panel for Management of Chemotherapy-Induced Diarrhea.2,4
Nonguideline-Based Therapies
Budesonide
Budesonide, an oral, topically active synthetic glucocorticoid, provides anti-inflammatory activity within the intestines.4,13 Historically, budesonide has been used as an anti-inflammatory agent in the treatment of inflammatory bowel disease (IBD). Of note, when patients with irinotecan-induced diarrhea underwent colonoscopy, findings were similar to those seen in patients with IBD.13 Budesonide decreases inflammation through the inhibition of mucosal prostaglandins within the intestines, restoring mucosal function and leading to intestinal fluid absorption.4 With a 90% first-pass effect in the liver, budesonide presents an improved safety profile compared with traditional oral glucocorticoids.14
In a study of 21 patients with loperamide-refractory diarrhea, budesonide decreased the severity of grade 3-4 diarrhea by at least 2 grades in 86% of patients treated with irinotecan.13 The dosage studied in this trial was 9 mg, given once daily for 3 to 5 days, starting the day of chemotherapy. Most patients saw a reduction in diarrhea within 24 to 48 hours.13 A second study evaluated the effects of budesonide (3 mg 3 times daily) in patients with advanced CRC.14 Patients in the budesonide arm experienced a shorter duration (1.8 vs 4.2 days, respectively) and fewer episodes (0.7 vs 2.2, respectively) of diarrhea than patients receiving placebo. Patients treated with budesonide also required fewer doses of loperamide (24.9 vs 36.2 capsules, respectively). Although findings were not statistically significant, a trend toward clinical improvement with budesonide therapy was demonstrated.14
Budesonide prophylaxis has been studied in the prevention of severe colitis, a common adverse effect of therapy with ipilimumab. However, Weber and colleagues found no difference between the treatment arms of ipilimumab plus budesonide or ipilimumab plus placebo in patients with grade 2 or higher diarrhea (33% vs 35%, respectively).10 Likewise, Berman and colleagues demonstrated that prophylactic oral budesonide did not prevent GI toxicity in patients undergoing treatment with ipilimumab.9 The authors theorized that the lack of effect was a result of insufficient amounts of budesonide reaching the distal colon.9
Corticosteroids in Ipilimumab-Induced Colitis
Although ipilimumab-associated colitis did not show response to prophylaxis with oral budesonide, response has been demonstrated with drug withdrawal and with systemic steroid administration.9,28 In 676 patients who were treated with ipilimumab for metastatic melanoma, the most common immune-related adverse event was diarrhea, with up to 32% of patients experiencing any grade of CID.29,30 Thirty-four patients experienced grade 3 to 5 enterocolitis.30 Of these patients, 29 (85%) were treated with high-dose corticosteroids (≥40-mg prednisone equivalent daily), with a median dose of 80 mg daily of prednisone or an equivalent.29,30 Complete resolution was seen in 74% of patients treated with steroids, 3% of patients improved to grade 2 severity, and 24% did not improve their Common Toxicity Criteria (CTC) score.30 Ipilimumab’s package insert recommends the discontinuation of therapy in patients experiencing severe enterocolitis and the initiation of systemic corticosteroids at a dose of 1 to 2 mg/kg daily of prednisone or an equivalent.30 When CID improves to grade 1 or less, steroids can be tapered over a duration of at least 1 month. Trials have shown that rapid steroid tapering can result in the recurrence or the worsening of CID in some patients.30
Glutamine
Glutamine, the most abundant amino acid in the body, serves as oxidative fuel for enterocytes.22 It is essential for normal intestinal structure, and it enhances nutrient transport in the GI tract.4 Over time, marked glutamine depletion develops in patients with cancer, enhancing the incidence and the severity of diarrhea. It has been proposed that glutamine supplementation may aid in protecting the gut mucosa from toxic chemotherapeutic agents.
In a randomized, double-blind, crossover study, Li and colleagues evaluated the prophylactic effect of glutamine in 54 patients with GI cancer.31 Patients were administered 20 g of IV alanyl-glutamine dipeptide (0.3 g/kg daily) on day 1 of chemotherapy and continued therapy for 5 days. Nausea and vomiting, as well as diarrhea, decreased significantly in patients treated with glutamine (P <.05). A 2001 study by Daniele and colleagues demonstrated that 18 g of oral glutamine daily increased intestinal absorption (P = .02) and decreased intestinal permeability (P = .04) to a greater extent than placebo in patients with CRC.16 Although the exact mechanism and extent with regard to decreasing CID remain uncertain, these studies have shown promise for patients with CID who are undergoing treatment for GI cancer.16,31
COX-2 Inhibitors
Clinical trials suggest that diarrhea may be induced by the overproduction of thromboxane A and prostaglandins in the GI tract.32 The body uses cyclooxygenase (COX), an enzyme found in normal tissue, to convert arachidonic acid into prostaglandins.32 The prostaglandin PGE2 stimulates mucous and chloride secretion from the epithelial cells in the colon, leading to significant diarrhea.22 In preclinical data, both COX-2 and PGE2 levels increased in a direct relationship to diarrhea incidence in rats given irinotecan.22 In this trial, celecoxib, a COX-2 selective inhibitor, at doses of ≥10 mg/kg daily was shown to reduce PGE2 levels, ameliorate diarrhea, and reduce weight loss while enhancing the anticancer effect of irinotecan.22
However, in a study by Maiello and colleagues, 81 patients with advanced CRC were randomized to receive FOLFIRI or FOLFIRI plus celecoxib.33 Celecoxib was given continuously as a 400-mg twice-daily dose starting on the first day of chemotherapy.33 No significant toxicity differences were seen between groups. Although preclinical trials suggest that celecoxib may be beneficial in alleviating CID, human studies have yet to show significant benefit of adding celecoxib to traditional chemotherapy.
Long-Acting Octreotide
In the GI tract, octreotide suppresses gastric emptying and inhibits active chloride secretion in the small intestine, allowing for water and electrolyte reabsorption.25 Octreotide long-acting release (LAR) is the intramuscular form of octreotide, which is slowly released over a period of 4 to 6 weeks.25 A case series by Rosenoff and colleagues evaluated 3 patients with severe refractory diarrhea who were treated with 30 mg of octreotide LAR every 28 days.34 All 3 patients experienced prompt resolution of diarrhea, improved quality of life, and completion of full-dose chemotherapy.34
In regard to the dosing of octreotide LAR, data supporting the use of 30-mg versus 40-mg dosing every 28 days has not been convincing.17 In the STOP trial, an open-label, randomized multicenter study by Rosenoff and colleagues, 147 patients with active or previous CID were randomized to receive 30 mg or 40 mg of octreotide LAR every 28 days.17 The primary end point was the proportion of patients with grade 3 or 4 diarrhea during the study period; secondary end points included the number of patients requiring IV fluids or changes in chemotherapy dosage, as well as a quality-of-life survey.17 Although fewer patients in the 40-mg treatment group experienced CID (48.4% vs 61.7% in the 30-mg group), differences did not reach significance (P = .14); however, with both dosages, the amount of CID was reduced significantly, because all patients experienced CID with previous cycles. The authors concluded that although octreotide LAR can be used safely and effectively for CID, no specific prophylactic dosing recommendations (30 mg vs 40 mg) can be made at this time.17 Octreotide is generally reserved as a second-line treatment of CID after patients fail treatment with loperamide, based on the increased drug cost of octreotide.6 Octreotide LAR could be considered for patients with refractory CID as a part of their CID management when other alternatives fail.17
Probiotics
Probiotics, nonpathogenic microorganisms such as Lactobacillus rhamnosus, Lactobacillus acidophilus, and bifidobacterium, have been extensively studied in the prevention of diarrhea associated with irritable bowel syndrome and Crohn’s disease.4,6 The possible mechanisms of action include providing a protective physical barrier from infectious bacteria, degrading carcinogens, and producing anti-inflammatory effects on the bowel mucosa.35 One clinical trial assessed the efficacy of Lactobacillus rhamnosus GG in reducing fluorouracil-induced CID in patients with CRC.18 L rhamnosus GG was administered orally twice daily at a dosage of 1 to 2 × 1010 daily for 24 weeks. A significant reduction in grade 3 to 4 diarrhea (22% vs 37% in the control group, respectively; P = .027) was seen with probiotic therapy.18 In addition, decreased abdominal discomfort and fewer dosage reductions in chemotherapy were seen with the use of probiotics.18 Immunocompromised patients should, however, be cautious of severe infections, such as sepsis, resulting from the use of probiotics.4 In that trial, no blood cultures from any of the patients grew Lactobacillus during the study period18; however, case reports have identified probiotics as a source of clinical bloodstream infections, especially in immunocompromised patients.36
Activated Charcoal
Activated charcoal, an adsorbent, has been used in the acute treatment of drug overdoses and poisonings.4 Its use as a prophylactic CID agent presents mechanistic potential, because it decreases enterohepatic cycling and increases SN-38 (irinotecan’s toxic form) clearance from the gut.6 A 2008 study by Sergio and colleagues evaluated the use of activated charcoal prophylaxis in children who were receiving a chemotherapy regimen of irinotecan plus either cisplatin plus doxorubicin (N = 20) or carboplatin (N = 2).19 Activated charcoal was given as a 250-mg capsule starting the evening before chemotherapy and every 8 hours thereafter until the end of the cycle.19 Loperamide was given at the onset of diarrhea (2 mg every 2 hours). Twenty-eight events of diarrhea were recorded, with a frequency of 28.88% in the activated charcoal group and 71.42% in the control group (P = .002).19 Grade 3 diarrhea was significantly more frequent in the control group (42.85% vs 2.22%, respectively). Children in the activated charcoal group completed their chemotherapy cycles more often, and compliance was nearly doubled. No significant adverse events to activated charcoal were reported in this trial.19
Michael and colleagues completed a similar study of activated charcoal in adults.37 Patients received 5 mL of aqueous Charcodote (1000 mg activated charcoal) plus 25 mL of water the evening before and subsequently every 8 hours for 48 hours postirinotecan chemotherapy during their first cycle.37 The patients then served as their own control, and they received no activated charcoal during their second cycle. Grade 3 to 4 diarrhea was present in 7.1% versus 25% of patients in cycles 1 and 2, respectively. The use of loperamide increased to more than 10 tablets in 25% of patients and in 54% of patients in cycles 1 and 2, respectively. The patients received 98% of their planned chemotherapy in the first cycle as opposed to only 70% in the second cycle. Again, activated charcoal was well tolerated and had excellent patient compliance.37 In these 2 trials, prophylactic use of activated charcoal has shown decreased grades 3 and 4 diarrhea and loperamide use, while optimizing the amount of irinotecan that could be administered.19,37 A phase 3, randomized controlled trial should be considered to validate these results.
Neomycin
Irinotecan-induced diarrhea is enhanced by the intestinal bacterial production of beta-glucuronidases, causing the transformation of SN38-G into its active form, SN-38.38 Several trials have evaluated the use of neomycin, a poorly absorbed aminoglycoside antibiotic, in its effect in treating CID through the reduction of intestinal microflora.39 In patients with CRC who are experiencing delayed-type diarrhea after their first irinotecan cycle, 1000-mg neomycin 3 times daily was administered for 2 days before and for 5 days after their second cycle.39 Of the 7 patients in the trial, 5 experienced no diarrhea after the second treatment course with neomycin (P = .03).39 Fecal cultures did not reveal any neomycin-resistant or pathogenic microorganisms. In addition, neomycin did not alter the plasma kinetics of SN-38 or irinotecan, and thus did not alter the efficacy of chemotherapy.39
Another study evaluated the combination of neomycin 25,000 IU plus bacitracin 2500 IU dosed 1000 mg 3 times daily for days 2 to 5 and days 16 to 19 of each cycle.40 All 15 patients with diarrhea in the first cycle had complete resolution of diarrhea for cycles 2 to 4.40 Neomycin was also studied as a prophylactic regimen at a dose of 660 mg 3 times daily for 3 days, starting 2 days before receiving irinotecan (350 mg/m2 once every 3 weeks).41 The overall incidence, severity, and duration of diarrhea were not statistically significant between the neomycin arm and placebo (P = .33); however, neomycin did show a 45% lower incidence of grade 3 delayed-onset diarrhea and a reduced duration of diarrhea by 0.9 days.41 The variation seen among trials could result from a difference in the treatment dose and duration of neomycin.
Cefpodoxime
Despite the potential risk for antibiotic-induced diarrhea, a number of antibiotics have shown promise in decreasing the incidence and the severity of CID.4 The third-generation cephalosporin, cefpodoxime, possesses the benefit of eliminating Escherichia coli, a known beta-glucuronidase producer, while not eradicating the anaerobes that are important for intestinal colonization. A phase 1, prospective, pediatric study performed by McGregor and colleagues investigated if this oral cephalosporin would allow for the dose escalation of irinotecan.42 In this study, 17 patients were treated with escalating levels of irinotecan starting at 20 mg/m2 per dose, the previously established maximum tolerated dose, for days 1 to 5 and days 8 to 12 of a 21-day course. Cefpodoxime (10 mg/kg daily, divided twice daily) was given to patients starting 2 days before chemotherapy and was continued as long as the patient was participating in the study.42
This study demonstrated that with the addition of cefpodoxime, the maximum tolerated dose of irinote-can could be increased to 30 mg/m2 per dose; however, diarrhea and diarrhea-associated dehydration remained the major dose-limiting toxicities when doses exceeded 30 mg/m2. Although prolonged administration of antibiotics may have the ability to increase the incidence of infections, none were demonstrated in this trial.42 Based on these results, further trials with cefpodoxime in children and adults are warranted and are under way.
Levofloxacin plus Cholestyramine
Flieger and colleagues hypothesized that the combination of cholestyramine, a bile acid chelator that reduces enterohepatic recirculation, plus levofloxacin to inhibit beta-glucuronidase production would be beneficial in patients with CID.43 Of patients with colorectal adenocarcinoma, 51 were treated with levofloxacin 500 mg daily and cholestyramine 4 g 3 times daily beginning the day before irinotecan administration and continuing for 3 days thereafter. The treatment of acute-onset diarrhea and delayed-onset diarrhea with standard doses of atropine and loperamide, respectively, was offered if necessary. Of the total patients in this trial, 78% reported no diarrhea. Only 22% of patients developed grade 1 to 2 diarrhea, 2% developed grade 3 diarrhea, and no patients developed grade 4 diarrhea.43 This prospective study illustrated that intestinal microflora suppression in combination with the reduction of enterohepatic recirculation of active chemotherapy provides suppression of diarrhea to well below the normal incidence of 40%.43 In addition, the short duration of antibiotic therapy supports a prompt recovery time of intestinal microflora, thereby decreasing potential adverse effects.
Kampo Medicine (Hangeshashin-To)
Hangeshashin-to (TJ-14) is a Chinese herbal product that is used in the treatment of acute gastroenteritis and that contains baicalin, a beta-glucuronidase inhibitor.44,45 Based on same mechanistic theory proposed for the use of oral antibiotics, TJ-14 may reduce active SN-38 concentrations in the intestine.44 A randomized, single-center trial by Mori and colleagues was conducted to investigate if the administration of TJ-14 would prevent and control CID.20 The trial included 41 patients with advanced non–small-cell lung cancer who were treated with irinotecan and cisplatin.20 The patients in the treatment arm were given TJ-14 at a dose of 7.5 g 3 times daily beginning more than 3 days before chemotherapy.20 Treatment with TJ-14 continued for a minimum of 21 days after the start of treatment with irinotecan plus cisplatin.20 Loperamide was administered to patients with severe diarrhea (grade ≥2). All but 2 patients (95%) in the TJ-14 group experienced some grade of diarrhea; however, the treatment arm did show an improved overall grade of diarrhea (P = .044) and a significantly lower incidence of grades 3 and 4 diarrhea (P = .018).20 The frequency and duration of diarrhea between the groups showed no difference. The major side effect of TJ-14, constipation, occurred in 11% of patients.20
Palifermin
Palifermin, a recombinant form of human keratinocyte growth factor (KGF), has been approved to reduce the incidence and the duration of severe oral mucositis in patients with hematologic malignancies who are receiving myelotoxic therapy that requires hematopoietic stem-cell support.46 The binding of KGF to its receptor results in the proliferation and differentiation of epithelial cells in multiple tissues, including the buccal mucosa, esophagus, stomach, and small intestine.46 Gibson and colleagues tested the efficacy of palifermin as an antidiarrheal agent in rats that were treated with irinotecan chemotherapy.47 One large dose (10 mg/kg) of palifermin was compared with multiple small doses (3 mg/kg daily for 3 days) or with placebo before administration of chemotherapy. The animals receiving palifermin prophylaxis had less severe diarrhea (single dose, 5%; multiple dose, 11%; and placebo, 28%; P <.05) in addition to maintaining their body weight.47
In a study by Rosen and colleagues, 64 patients with metastatic CRC being treated with fluorouracil plus leucovorin were receiving palifermin (40 mcg/kg) for 3 consecutive days before 2 chemotherapy cycles.21 Although the incidence of severe mucositis in patients treated with palifermin was half that of the placebo control group (P = .016) and reduced the need for chemotherapy dose reductions, the incidence of diarrhea did not differ between the groups.21 Grade 2 or higher CTC was observed in 20% of patients receiving placebo and in 18% of patients receiving palifermin during cycle 1.21 The most common adverse reactions to palifermin include skin toxicities (ie, rash, erythema) and oral toxicities (ie, reversible tongue thickening and tongue discoloration and alteration in taste).46
Other Treatments for Chemotherapy-Induced Diarrhea
Although not included in this review because of limited human data and the severity of side-effect profiles, thalidomide, cyclosporine, and racecadotril have been considered and have been used in limited studies as treatment options for CID.
Conclusion
Although guidelines exist for the treatment of CID, patient needs often exceed these recommendations. The majority of the clinical trials discussed in this article were limited by their small population size and their short-term follow-up. Further investigation should be conducted with promising therapies for validation before recommendation for guideline inclusion. Furthermore, with the emergence of new therapeutic alternatives in patients with severe CID, an update to the current treatment guidelines is warranted. n
Author Disclosure Statement
Dr Koselke and Dr Kraft reported no conflicts of interest.
References
1. Maroun JA, Anthony LB, Blais N, et al. Prevention and management of chemotherapy-induced diarrhea in patients with colorectal cancer: a consensus statement by the Canadian working group on chemotherapy-induced diarrhea. Curr Oncol. 2007;14:13-20.
2. Benson AB 3rd, Ajani JA, Catalano RB, et al. Recommended guidelines for the treatment of cancer treatment-induced diarrhea. J Clin Oncol. 2004;22:2918-2926.
3. Arbuckle RB, Huber SL, Zacker C. The consequences of diarrhea occurring during chemotherapy for colorectal cancer: a retrospective study. Oncologist. 2000;5:250-259.
4. Richardson G, Dobish R. Chemotherapy induced diarrhea. J Oncol Pharm Pract. 2007;13:181-198.
5. Kornblau S, Benson AB, Catalano R, et al. Management of cancer treatment-related diarrhea. Issues and therapeutic strategies. J Pain Symptom Manage. 2000;19:118-129.
6. Stein A, Voigt W, Jordan K. Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management. Ther Adv Med Oncol. 2010;2:51-63.
7. Wadler S, Benson AB 3rd, Engelking C, et al. Recommended guidelines for the treatment of chemotherapy-induced diarrhea. J Clin Oncol. 1998;16:3169-3178.
8. Saltz LB. Understanding and managing chemotherapy-induced diarrhea. J Support Oncol. 2003;1:35-46; discussion 38-41, 45-46.
9. Berman D, Parker SM, Siegel J, et al. Blockade of cytotoxic T-lymphocyte antigen-4 by ipilimumab results in dysregulation of gastrointestinal immunity in patients with advanced melanoma. Cancer Immun. 2010;10:11.
10. Weber J, Thompson JA, Hamid O, et al. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin Cancer Res. 2009;15:5591-5598.
11. National Cancer Institute. Common Terminology Criteria for Adverse Events, v4.03. National Cancer Institute, National Institutes of Health, Department of Health and Human Services; June 14, 2010.
12. Yumuk PF, Aydin SZ, Dane F, et al. The absence of early diarrhea with atropine premedication during irinotecan therapy in metastatic colorectal patients. Int J Colorectal Dis. 2004;19:609-610
13. Lenfers BH, Loeffler TM, Droege CM, Hausamen TU. Substantial activity of budesonide in patients with irinotecan (CPT-11) and 5-fluorouracil induced diarrhea and failure of loperamide treatment. Ann Oncol. 1999;10:1251-1253.
14. Karthaus M, Ballo H, Abenhardt W, et al. Prospective, double-blind, placebo-controlled, multicenter, randomized phase III study with orally administered budesonide for prevention of irinotecan (CPT-11)-induced diarrhea in patients with advanced colorectal cancer. Oncology. 2005;68:326-332.
15. Howard A, Hoffman J, Sheth A. Clinical application of voriconazole concentrations in the treatment of invasive aspergillosis. Ann Pharmacother. 2008;42:1859-1864.
16. Daniele B, Perrone F, Gallo C, et al. Oral glutamine in the prevention of fluorouracil induced intestinal toxicity: a double blind, placebo controlled, randomised trial. Gut. 2001;48:28-33.
17. Rosenoff SH, Gabrail NY, Conklin R, et al. A multicenter, randomized trial of long-acting octreotide for the optimum prevention of chemotherapy-induced diarrhea: results of the STOP trial. J Support Oncol. 2006;4:289-294.
18. Österlund P, Ruotsalainen T, Korpela R, et al. Lactobacillus supplementation for diarrhoea related to chemotherapy of colorectal cancer: a randomised study. Br J Cancer. 2007;97:1028-1034.
19. Sergio GC, Felix GM, Luis JV. Activated charcoal to prevent irinotecan-induced diarrhea in children. Pediatr Blood Cancer. 2008;51:49-52.
20. Mori K, Kondo T, Kamiyama Y, et al. Preventive effect of kampo medicine (hangeshashin-to) against irinotecan-induced diarrhea in advanced non-small-cell lung cancer. Cancer Chemother Pharmacol. 2003;51:403-406.
21. Rosen LS, Abdi E, Davis ID, et al. Palifermin reduces the incidence of oral mucositis in patients with metastatic colorectal cancer treated with fluorouracil-based chemotherapy. J Clin Oncol. 2006;24:5194-5200.
22. Yang X, Hu Z, Chan SY, et al. Novel agents that potentially inhibit irinotecan-induced diarrhea. Curr Med Chem. 2005;12:1343-1358.
23. Dom J, Leyman R, Schuermans V, Brugmans J. Loperamide (R 18 553), a novel type of antidiarrheal agent. Part 8: clinical investigation. Use of a flexible dosage schedule in a double-blind comparison of loperamide with diphenoxylate in 614 patients suffering from acute diarrhea. Arzneimittelforschung. 1974;24:1660-1665.
24. Rubenstein EB, Peterson DE, Schubert M, et al. Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer. 2004;100(9 suppl):2026-2046.
25. Prommer EE. Established and potential therapeutic applications of octreotide in palliative care. Support Care Cancer. 2008;16:1117-1123.
26. Gebbia V, Carreca I, Testa A, et al. Subcutaneous octreotide versus oral loper-amide in the treatment of diarrhea following chemotherapy. Anticancer Drugs. 1993;4:443-445.
27. Goumas P, Naxakis S, Christopoulou A, et al. Octreotide acetate in the treatment of fluorouracil-induced diarrhea. Oncologist. 1998;3:50-53.
28. Kahler KC, Hauschild A. Treatment and side effect management of CTLA-4 antibody therapy in metastatic melanoma. J Dtsch Dermatol Ges. 2011;9:277-286.
29. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
30. Yervoy (ipilimumab) [package insert]. Princeton, NJ: Bristol-Myers Squibb; 2011.
31. Li Y, Ping X, Yu B, et al. Clinical trial: prophylactic intravenous alanyl-glutamine reduces the severity of gastrointestinal toxicity induced by chemotherapy—a randomized crossover study. Aliment Pharmacol Ther. 2009;30:452-458.
32. Fakih MG, Rustum YM. Does celecoxib have a role in the treatment of patients with colorectal cancer? Clin Colorectal Cancer. 2009;8:11-14.
33. Maiello E, Giuliani F, Gebbia V, et al. FOLFIRI with or without celecoxib in advanced colorectal cancer: a randomized phase II study of the Gruppo Oncologico dell’Italia Meridionale (GOIM). Ann Oncol. 2006;17 (suppl 7):vii55-vii59.
34. Rosenoff SH. Octreotide LAR resolves severe chemotherapy-induced diarrhoea (CID) and allows continuation of full-dose therapy. Eur J Cancer Care (Engl). 2004;13:380-383.
35. Miller AC, Elamin EM. Use of probiotics for treatment of chemotherapy-induced diarrhea: is it a myth? JPEN J Parenter Enteral Nutr. 2009;33:573-574.
36. Salminen MK, Tynkkynen S, Rautelin H, et al. Lactobacillus bacteremia during a rapid increase in probiotic use of lactobacillus rhamnosus GG in Finland. Clin Infect Dis. 2002;35:1155-1160.
37. Michael M, Brittain M, Nagai J, et al. Phase II study of activated charcoal to prevent irinotecan-induced diarrhea. J Clin Oncol. 2004;22:4410-4417.
38. Schmittel A, Jahnke K, Thiel E, Keilholz U. Neomycin as secondary prophylaxis for irinotecan-induced diarrhea. Ann Oncol. 2004;15:1296.
39. Kehrer DF, Sparreboom A, Verweij J, et al. Modulation of irinotecan-induced diarrhea by cotreatment with neomycin in cancer patients. Clin Cancer Res. 2001;7:1136-1141.
40. Alimonti A, Satta F, Pavese I, et al. Prevention of irinotecan plus 5-fluorouracil/leucovorin-induced diarrhoea by oral administration of neomycin plus bacitracin in first-line treatment of advanced colorectal cancer. Ann Oncol. 2003;14:805-806.
41. de Jong FA, Kehrer DF, Mathijssen RH, et al. Prophylaxis of irinotecan-induced diarrhea with neomycin and potential role for UGT1A1*28 genotype screening: a double-blind, randomized, placebo-controlled study. Oncologist. 2006;11:944-954.
42. McGregor LM, Stewart CF, Crews KR, et al. Dose escalation of intravenous irinotecan using oral cefpodoxime: a phase I study in pediatric patients with refractory solid tumors. Pediatr Blood Cancer. 2012;58:372-379.
43. Flieger D, Klassert C, Hainke S, et al. Phase II clinical trial for prevention of delayed diarrhea with cholestyramine/levofloxacin in the second-line treatment with irinotecan biweekly in patients with metastatic colorectal carcinoma. Oncology. 2007;72:10-16.
44. Sharma R, Tobin P, Clarke SJ. Management of chemotherapy-induced nausea, vomiting, oral mucositis, and diarrhoea. Lancet Oncol. 2005;6:93-102.
45. Kase Y, Hayakawa T, Takeda S, et al. Pharmacological studies on antidiarrheal effects of hange-shashin-to. Biol Pharm Bull. 1996;19:1367-1370.
46. Kepivance (palifermin injection) [package insert]. Thousand Oaks, CA: Amgen; 2012.
47. Gibson RJ, Bowen JM, Keefe DM. Palifermin reduces diarrhea and increases survival following irinotecan treatment in tumor-bearing DA rats. Int J Cancer. 2005;116:464-470.