Oral chemotherapy treatments were first approved in 1953 and include familiar agents such as chlorambucil, 6-mercaptopurine, and methotrexate, which are still used in the treatment of cancer today.1 Since the approval of capecitabine in 1998, there has been rapid development of new oral chemotherapies, a broad pharmacologic class that includes oral cytotoxic agents and small-molecule inhibitors that target surface proteins and tumor biologic pathways.1 This growth is expected to continue because oral agents comprise a significant proportion of the pipeline of oncology treatments that are currently being developed.1,2
Oral chemotherapy offers patients increased ease of administration and an improved quality of life (QOL).2 For example, oral treatments allow patients to avoid painful injections, prolonged infusions, and time spent at infusion centers, thereby interfering less with work and social activities.2 In addition, the self-administration of oral medications improves patient autonomy.2
In a study by Liu and colleagues, 33% of patients who responded to a survey noted a preference for the greater sense of control that oral chemotherapy afforded them versus intravenous chemotherapy.3 In that same survey, 80% of patients said that they would prefer oral chemotherapy over parenteral therapy as long as the agents were equally efficacious.3 In some cases, the oral administration of topoisomerases I inhibitors and fluoropyrimidines can provide prolonged drug exposure compared with parenteral therapy and therefore may be a more viable treatment delivery option.2
Even with these benefits, oral chemotherapy treatments have numerous unique challenges. Although cytotoxic parenteral chemotherapy regimens are typically given intermittently in cycles with a recovery period, many oral cancer therapies are often cytostatic and typically must be administered daily continuously.1 Thus, to maximize the effectiveness of oral chemotherapy regimens, patients must maintain high adherence to therapy.1
In a study of treatment with imatinib in patients with chronic myeloid leukemia, >90% adherence to therapy was associated with a significantly better probability of achieving major and complete molecular responses.4 Adherence is typically higher in patients who are older, have a higher education level, and are more affluent. In addition, regimens that have simpler dosing strategies are also associated with better adherence,2 whereas adherence is inversely related to the number of pills or capsules required daily and typically decreases with long-term treatment administration.2
Patients can face many barriers to adherence, including low health literacy, limited patient knowledge, patient forgetfulness, complex treatment administration instructions, challenging side effects, and expensive out-of-pocket (OOP) costs.5
In contrast to parenteral chemotherapy, a majority of the monitoring and management responsibilities for oral chemotherapy regimens are transferred from the hematology/oncology clinic to the patient.1,6,7 Historically, hematology/oncology practices were designed to facilitate the periodic administration of parenteral therapies and not for the self-administration of continuous oral therapies by patients.
Studies have shown that the systems designed to facilitate the safe administration of parenteral cancer therapies are often not robust enough for oral chemotherapy, and that institutions have been slow to adopt established safety checks for parenteral chemotherapy in the oral chemotherapy setting.1,6-12 As a result, patients receiving oral chemotherapy for the treatment of malignancies are often subject to a less standardized approach to care.1,6-12
To address the need for increased vigilance and standardized education for patients receiving oral chemotherapy, the American Society of Clinical Oncology developed core measures that are specific to oral chemotherapy for the Quality Oncology Practice Initiative, including monitoring patient adherence to oral chemotherapy.13 In addition, the Hematology/Oncology Pharmacy Association has published a pharmacy practice standard regarding best practices for the use of oral oncolytic treatments.14
The Vizient Cancer Care Committee was initiated in January 2018. Vizient is a member-driven healthcare performance improvement company that includes more than 25 member networks from various professional disciplines, topic interests or segments, and system-level executives.15 Vizient’s membership includes a diverse collection of academic medical centers, pediatric facilities, community hospitals, and integrated health delivery networks.15
The Cancer Care Committee is a subgroup of the pharmacy member network and consists of more than 50 member institutions. Serving as a forum for assessing and spreading best practices that are designed to improve all aspects of cancer care delivery, the committee’s work focuses on the expansion of the pharmacist’s role, the use of technology to improve safety and efficiency, and the implementation of value-based care.16
Because of the unique challenges surrounding oral chemotherapy, many hematology/oncology practices have developed comprehensive oral chemotherapy programs.6,8-11,17 These programs are aimed at optimizing the benefits of oral chemotherapy by assisting patients and providers in overcoming many of the unique barriers associated with these treatments while also improving patient safety.
We conducted a national survey to assess the components of oral chemotherapy management programs throughout the United States, as well as what program-associated metrics were being collected, in order to identify the key program monitoring parameters that can be used for the development of standard metrics that define the impact of oral chemotherapy programs.
Methods
In October 2018, the Oral Chemotherapy Working Group, a project group of the Vizient Cancer Care Committee, conducted an online survey of the membership regarding their current oral chemotherapy practices. The survey questions were developed by the working group and included demographic information and specific oral chemotherapy management program–associated metrics that were chosen based on the working group members’ own experiences with oral chemotherapy management programs and were divided into the categories of safety, effectiveness, adherence, financial, and value outcomes.
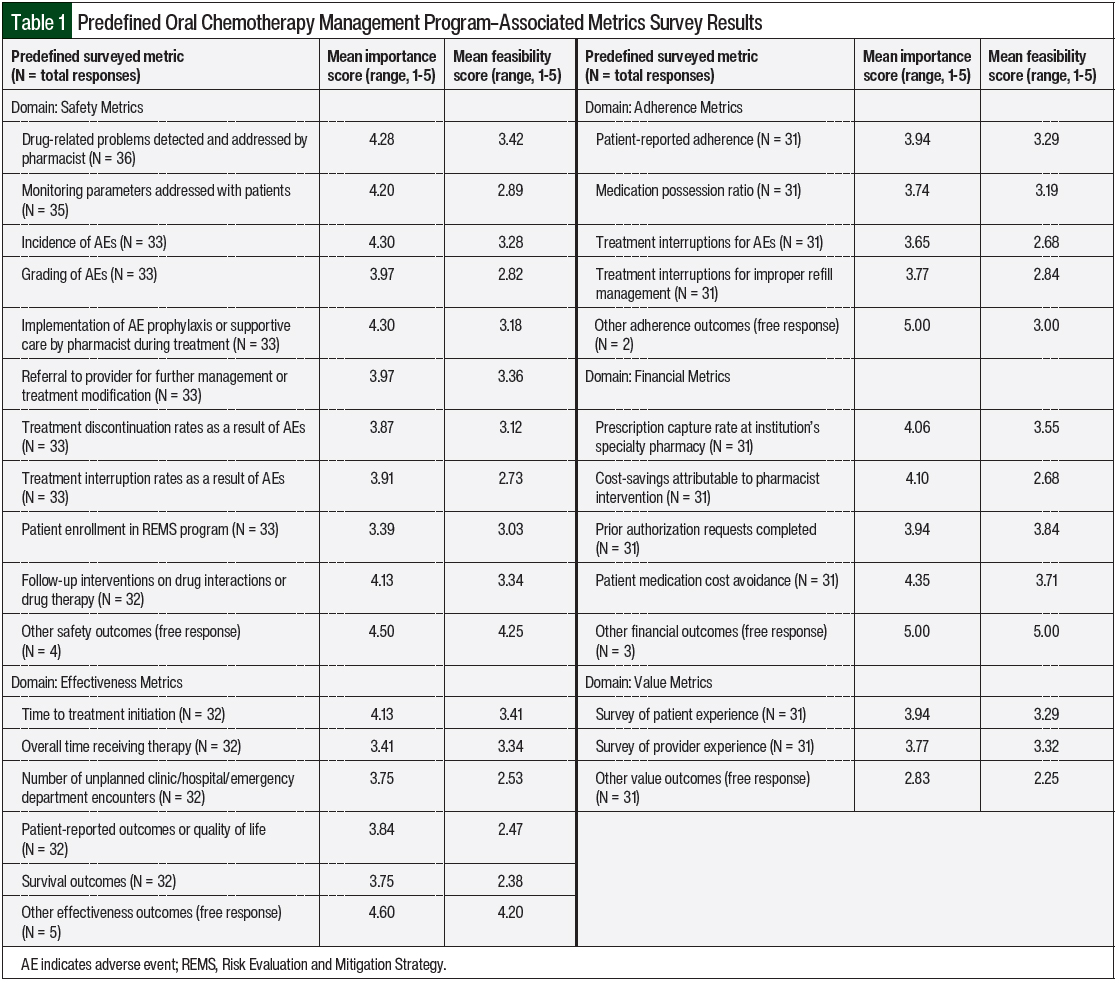
The respondents were asked to answer descriptive information about their institution, including if they had clinical pharmacists practicing in clinics, an on-site specialty pharmacy, and/or an established oral chemotherapy program. The respondents who indicated that their institution currently had an oral chemotherapy program were then asked if any safety, effectiveness, adherence, financial, or value outcomes were being collected as part of that program, as well as 25 specific predefined outcomes in those 5 categories. See Table 1 for the complete list of the predefined outcomes.
In addition to asking if their oral chemotherapy management program was currently collecting any specific outcomes, the respondents were also asked to rate each predefined outcome on a scale of 1 to 5 (5 being the highest) for the importance (how valuable you find this metric) and the feasibility (how achievable it would be for you to collect this metric) of collecting the outcome.
The survey was sent to all Vizient pharmacy network member institutions via postings to the discussion boards and listservs for the Cancer Care Committee and Pharmacy Cancer Care networks.
Results
We received a total of 40 survey responses from 38 member institutions (Table 2). Two institutions had 2 different survey respondents; for these institutions, both responses were included. In all, 71% of the institutions reported that they had an established oral chemotherapy management program. All institutions had initiated this program within the past 10 years, and the most frequent duration of the program was 3 to 6 years.
A total of 68% of the respondents stated that pharmacy was a primary manager of the oral chemotherapy program, with nursing being the second most common discipline, at 20%. All respondents stated that they had clinical pharmacists embedded in their clinic, and 85% stated that they had a specialty pharmacy on-site.
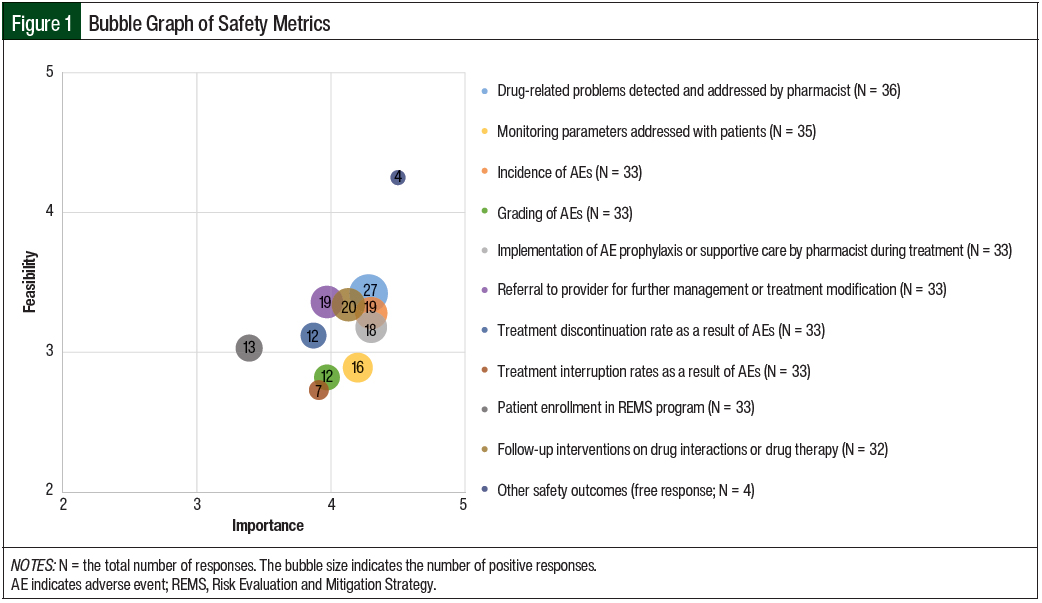
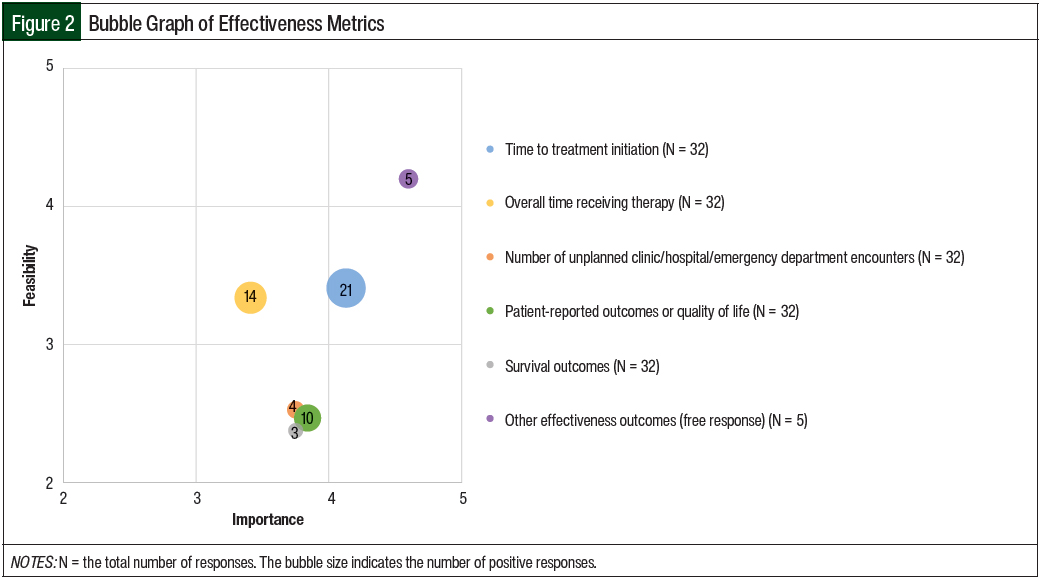
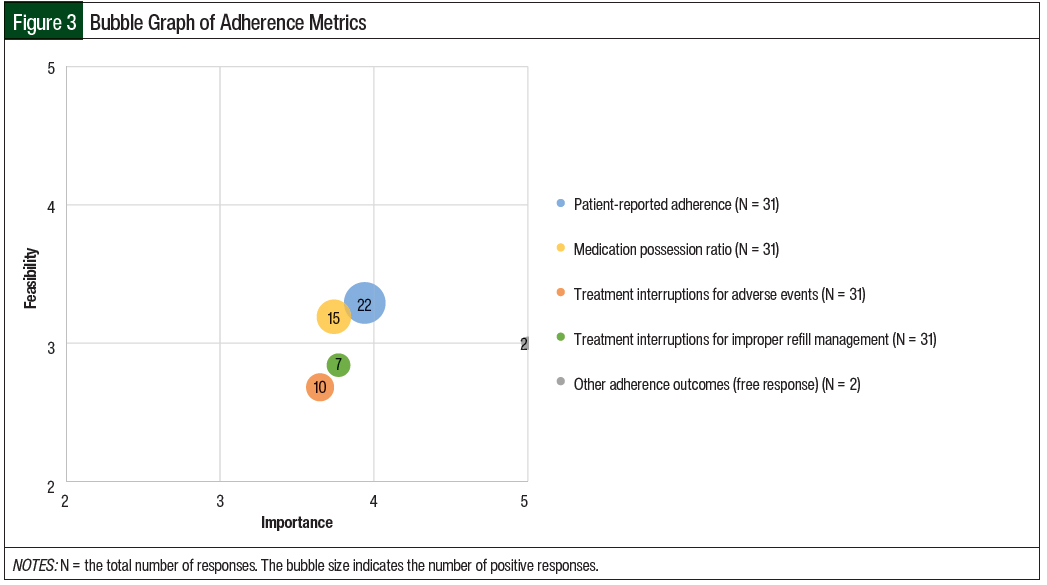
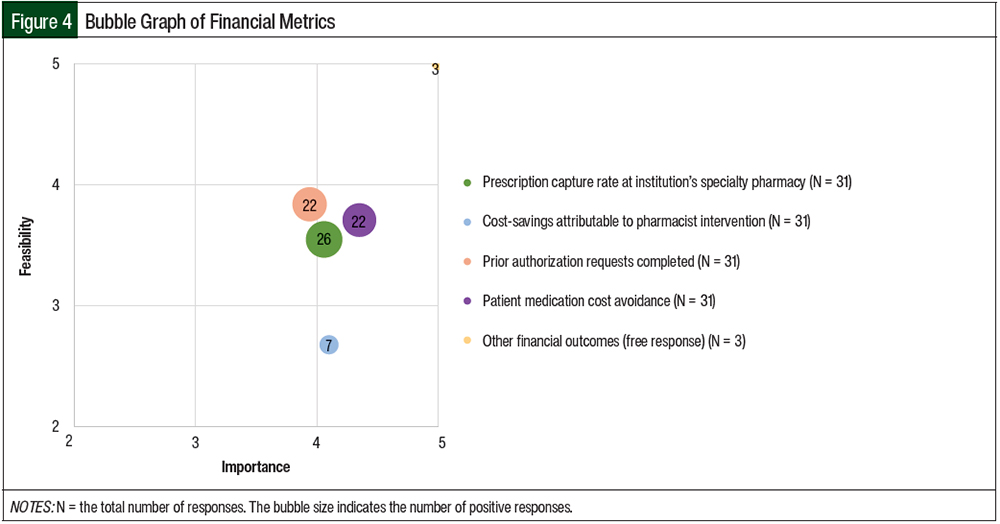
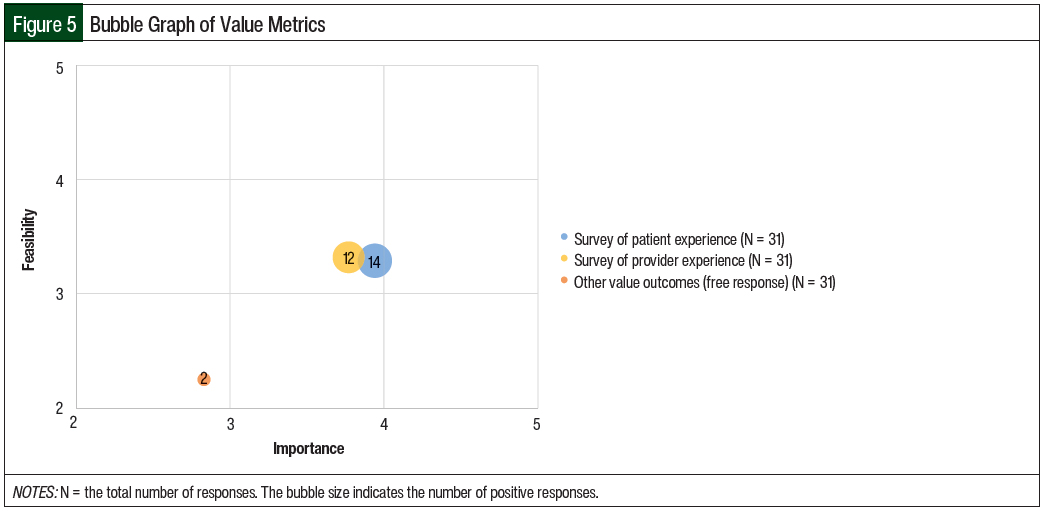
The mean results of the importance and feasibility ratings for the predefined oral chemotherapy management program–associated metrics are shown in the bubble charts in Figures 1 through 5 and in Table 1. The size of and number inside the bubbles are relative to the number of institutions that indicated they collect the outcomes. The bubbles’ placement on the graph is dependent on the outcomes’ mean scores for importance and feasibility. The x-axis scale is relative to the importance of the outcome. The y-axis scale is relative to the feasibility of the outcome. Ideal metrics are highly valuable and highly feasible.
Safety
Of the respondents, 58% indicated that they were collecting safety outcomes, and 10 different outcomes were specifically assessed through the survey (Table 1 and Figure 1).
The most frequently collected safety outcomes were drug-related problems detected and addressed by pharmacists (68%), follow-up interventions on drug interactions or drug therapy (50%), the incidence of adverse events (48%), referral to a provider for further management of an adverse event or oral chemotherapy treatment modification (48%), and pharmacists’ implementation of an adverse-event prophylaxis or supportive-care measures during treatment (45%).
Combined, these 5 outcomes had a mean importance score of 4.20 and a mean feasibility score of 3.32. In general, the grading of adverse events was not collected (30%) and had a lower feasibility score (mean, 2.82) than the other metrics; similar results were seen for treatment discontinuation rates as a result of adverse events, treatment interruption rates resulting from adverse events, and patient enrollment in a Risk Evaluation and Mitigation Strategy program.
Only 40% of respondents were collecting data about addressing treatment monitoring parameters with patients, such as the QT interval, left ventricular ejection fraction, or blood glucose levels; this had a mean importance score of 4.20 and a mean feasibility score of 2.89.
Effectiveness
A total of 45% of the respondents indicated that they were collecting effectiveness data as a component of their oral chemotherapy program. The effectiveness outcomes that were collected included the time to treatment initiation (53%); overall time receiving therapy (35%); patient-reported outcomes or QOL (25%); the number of unplanned clinic, hospital, or emergency department encounters (10%); and survival outcomes (8%).
The importance of collecting the time to treatment initiation was rated the highest of all of the effectiveness outcomes (mean, 4.13); the other effectiveness measures were rated a mean of 3.41 to 3.84 in terms of importance. The feasibility of collecting the effectiveness data varied more widely and with lower scores than the importance scores (Table 1 and Figure 2).
In terms of feasibility scores, collecting information on the time to treatment initiation had the highest feasibility score (mean, 3.41) whereas the feasibility of collecting the impact on survival outcomes had the lowest score (mean, 2.38). A total of 5 respondents also indicated in a free-response field that their programs were collecting additional effectiveness metrics, including tracking tumor markers, the rates of major molecular responses in patients with chronic myeloid leukemia, and patient satisfaction outcomes.
Adherence
When asked about adherence outcomes, 58% of the respondents said that adherence outcomes were being collected through their program. The most common adherence measures that the respondents collected were patient-reported adherence (55%) and medication possession ratios (38%; Table 1 and Figure 3).
Fewer respondents collected the number of treatment interruptions resulting from adverse events (25%) or from improper refill management (18%). A free-response field was completed by 2 respondents who were collecting the proportion of days covered (5%).
Overall, the adherence metrics received a mean importance score of 4.02 and a mean feasibility score of 3.00.
Financial
In the survey, 53% of respondents reported collecting financial outcomes. Prescription capture rate was the main outcome collected, with 65% of the respondents collecting the number of prescriptions written at their facility that were subsequently dispensed by the institution’s specialty pharmacy. The respondents also reported high collection rates of data evaluating the number of prior authorization requests completed as well as the avoidance of medication cost by patients.
In all, 58% of the respondents indicated that the number of prior authorizations completed was being collected at their institutions. This metric was rated close to 4 for importance (mean, 3.94) and feasibility (mean, 3.84; Table 1 and Figure 4). Patient medication cost avoidance, which was defined as the amount of OOP money saved by the use of patient assistance or other related programs, was being tracked by 55% of the respondents, with a mean importance score of 4.35 and a mean feasibility score of 3.71.
Only 18% of the respondents reported that cost-savings attributable to pharmacist interventions were currently being measured. This outcome had a mean importance score of 4.10, but a mean feasibility score of 2.68. Other financial metrics that the respondents reported collecting in a free-response field included revenue growth and lost revenue.
Value
A total of 35% of the respondents indicated that they were collecting value outcomes as part of their oral chemotherapy services. The most common value outcome collected was a survey of patients’ experiences with pharmacy services; 35% of the respondents confirmed they were collecting this information. Surveys of providers’ experiences with pharmacy services were collected by 30% of the respondents.
The respondents rated the importance of collecting the patient experience (mean, 3.94) higher than the importance of collecting the provider experience (mean, 3.77; Table 1 and Figure 5). The respondents felt that the feasibility of collecting the patient and provider experiences were similar (mean, 3.29 and 3.32, respectively). In a free-response answer, 1 respondent reported surveying other supportive-care staff.
Discussion
At the time our survey was conducted, almost 75% of the responding organizations already had an oral chemotherapy management program in place. In addition, 3 survey respondents that did not yet have a formal program established indicated that pharmacy was still one of the disciplines primarily involved in the management of oral chemotherapy. This could indicate that these respondents felt that even though their institutions did not have a formal oral chemotherapy program, pharmacists at their institution were informally involved with oral chemotherapy–related processes.
The surveyed institutions were primarily large academic medical centers. The respondents had already incorporated clinical pharmacists into the clinic, and the vast majority also had an on-site specialty pharmacist. The presence of these resources may have impacted the respondents’ feasibility scores on the surveyed outcomes, and thus a limitation of this work is that our results may not be broadly generalizable to institutions that are currently lacking these pharmacy resources, such as smaller rural clinics or community hospitals.
In addition, when asked if their institution was collecting any outcomes for each of the 5 different outcome domains, some survey respondents reported that their institution was not collecting outcomes in one of the domains; however, they later responded affirmatively when asked if their institution was collecting a specifically surveyed predefined oral chemotherapy management program–associated metric, which could indicate that the respondent had not previously considered that metric to be related to that domain.
Although the survey respondents were asked to self-assess the importance and feasibility of each predefined oral chemotherapy management program–associated metric, when combined, the feasibility scores had a larger range and lower average than the importance scores. One explanation for this could be the inherent difficulties associated with collecting the metrics associated with established oral chemotherapy management programs, which can be extremely labor intensive, especially when considering the high volume of results that are likely needed to draw meaningful conclusions.
In addition to the work required to collect the data, the time and effort required to ensure the accuracy of the data and to analyze the data are also significant. This further emphasizes the importance of developing and disseminating standardized metrics that are meaningful and can be easily and practically collected by institutions.
Standardized metrics will also allow for cross-institutional collation and/or comparison. Future goals could include the development of more automated data collection and reporting tools that use these standardized metrics within electronic health records (EHRs) and pharmacy dispensing software to further reduce administrative and logistical burdens.
Safety
The results from our survey demonstrate the inherent difficulties that exist with collecting safety-related metrics that are associated with oral chemotherapy monitoring programs. From a safety perspective, monitoring patients while they are receiving oral chemotherapy often relies on patient self-reporting, and the documentation of treatment interruptions and discontinuations often lacks accuracy in the medical record.
Following the recommended laboratory, procedure, or imaging monitoring relies on accurate patient education, appropriate scheduling, and patient adherence to the recommendations. Although Common Terminology Criteria for Adverse Events grading is standard in clinical trials, this grading was viewed by survey respondents as unlikely to be feasible to perform during daily clinical practice.
Tracking drug-related problems that are identified and addressed by pharmacists, interventions regarding drug interactions, and the pharmacist-led implementation of supportive-care or adverse-event prophylactic measures can show the direct clinical impact of pharmacists on these oral chemotherapy monitoring programs, and it is not surprising that these were some of the predefined safety metrics with the highest mean importance scores.
Effectiveness
Only half of the surveyed programs were collecting any effectiveness data as a part of their oral chemotherapy programs. Interestingly, effectiveness metrics were perceived by the survey respondents to be the least important and least feasible outcomes to collect. The survey respondents indicated that the time to treatment initiation was the most important predefined effectiveness metric for oral chemotherapy management programs, and they ranked its importance higher than QOL or survival outcomes; this could be correlated to its comparatively higher mean feasibility score in relation to the other effectiveness metrics.
The respondents could also have thought it more likely that oral chemotherapy management programs could have a demonstrative impact on the time to treatment initiation versus the other surveyed effectiveness outcomes. In addition, for institutions with their own on-site specialty pharmacy, this could be a useful metric to track their performance relative to outside, largely insurance company– or pharmacy benefit manager–owned specialty pharmacies.
Effectiveness outcomes also had the highest number of free-response answers by the survey respondents. These free-response answers had higher mean feasibility and importance scores than any of the predefined effectiveness outcomes in the survey. Demonstrating the effectiveness of oral chemotherapy programs can be important when justifying the impact of such programs.
That being said, the real-time tracking of metrics such as the total time receiving therapy and patient-reported outcomes, QOL, or survival-related outcomes would likely be logistically challenging and would likely not have much use beyond the reports providing real-world comparison data to established clinical trials.
The identification of metrics that can track the effectiveness of oral chemotherapy programs and can be collected more feasibly than the predefined effectiveness outcomes included in this survey is clearly needed.
Adherence
A majority of responding institutions indicated that they had some method for collecting oral chemotherapy adherence as a part of their oral chemotherapy monitoring program. Multiple methods are used to monitor drug adherence, including patient self-reporting, patient diaries, prescription refill history, pill counts, drug plasma levels, various microelectronic monitoring systems, and digital medications.
Consistency within the literature is lacking, and therefore clinical practices regarding how to measure adherence to oral chemotherapies vary. The most accessible methods (eg, self-reporting, patient diaries) lack accuracy, whereas the most accurate methods (eg, microelectronic monitoring systems, digital medicine) are often cost- and resource-prohibitive.
Our survey aimed to determine how organizations are systematically tracking adherence to oral chemotherapy. Patient-reported adherence was the most frequently used metric and was viewed as only the second-most important, but most feasible, measure to track among the respondents. The survey respondents viewed the proportion of days covered, which was a free-response option, as the most important adherence metric. This is expected given its wide use and built-in inclusion in some pharmacy dispensing software programs and EHRs.
It should be noted, however, that proportion of days covered as the basis in pharmacy claims data does not account for clinically appropriate gaps in therapy nor patients who receive free drug via drug manufacturers’ assistance programs. This is an important caveat considering that the often extremely high cash prices of newer targeted oral cancer therapies, as well as the laws preventing patients who have Medicare and Medicaid coverage from using copay cards, can lead to a significant proportion of patients participating in oral chemotherapy monitoring programs that use these drug manufacturer assistance programs.
Financial
A majority of participating institutions had methods in place to collect data regarding the financial outcomes associated with oral chemotherapy and the institutions’ corresponding monitoring programs. Cancer is one of the costliest diseases to treat, and new oral targeted therapies for cancer continue to contribute to the rising costs of cancer care. For patients who are receiving oral chemotherapy, OOP costs can be a key barrier preventing adherence to a treatment plan.
Oral chemotherapy drugs are most often covered under the patient’s pharmacy benefit. Many plans have moved more cost-sharing onto patients, which may be catastrophic when a patient is newly diagnosed with cancer. The increased costs associated with cancer treatment can cause financial distress and anxiety for patients.
Our survey’s results demonstrate that pharmacy services supporting cancer care could make a great impact in the facilitation of prior authorizations and obtaining financial assistance for patients. These metrics are a good indicator of the impact that these programs have on the patient experience and on the reduction of financial barriers to medication adherence. Both metrics could also be useful in tracking performance relative to outside specialty pharmacy programs for the institutions that have their own specialty pharmacy.
There is a need for assistance in tracking cost avoidance as a result of these programs. One opportunity that was identified in the survey results was the measurement of cost-savings attributable to pharmacists’ interventions, which can be useful for pharmacy position justification, as well as for demonstrating the financial value of these oral chemotherapy monitoring programs. Only a small percentage of institutions were collecting those data; however, the respondents rated this metric as highly important, but relatively difficult to collect.
Value
Less than 50% of the surveyed oral chemotherapy programs were formally collecting value outcomes on patients’ or providers’ experiences with pharmacy services. Only 35% of the respondents stated that they were formally collecting patient satisfaction metrics regarding pharmacy services.
The patient experience is an important metric for any healthcare setting, and ensuring that patients feel satisfied with the care they receive should be a baseline metric incorporated by every oral chemotherapy program.
The determination of a standardized patient experience survey and the use of this survey by oral chemotherapy management programs would allow for benchmarking and cross-institutional comparisons. The majority of the respondents stated that their oral chemotherapy programs were being primarily managed by pharmacists and nurses.
Providers are a key component of the oral chemotherapy care delivery team, and it is important to ensure that they are engaged and satisfied with such programs. Only 30% of the surveyed programs were evaluating providers’ experiences, which can be a valuable tool in the continuous quality improvement of the program to ensure that all key contributors are able to provide input on how this complex patient care process can be optimized.
Limitations
Additional limitations of this study include our reliance on voluntary survey responses to listserv posts and a lack of consistency in survey answers among both respondents from the same institution as well as individual survey respondents providing conflicting responses if they were collecting outcomes in 1 of the domains compared with the predefined survey metric within said domain.
Conclusion
Using a sampling of Vizient’s member institutions, the results of our survey demonstrate the heterogeneity of the monitoring practices that are associated with oral chemotherapy programs across the United States. There is a clear need for the establishment and dissemination of best practices for monitoring the impact of these programs to bring clarity and consistency to these programs and to further enhance the quality and safety of patient care.
Future objectives include the identification and distribution of said best practices, as well as standardized metrics that assess the impact of oral chemotherapy programs that are highly valuable and highly feasible to collect. Other goals include identifying the strategies to overcome barriers faced by institutions that are attempting to measure these metrics to best operationalize the collection of these standardized benchmarks.
Funding Source
This research was supported by Vizient, Inc.
Author Disclosure Statement
Dr Taucher was an employee of G1 Therapeutics through October 2021; Dr Fahey, Dr Barbour, Dr Golf, Dr Malard Johnson, Dr Morgan, Dr Rhoades, and Dr Valgus have no conflicts of interest to report.
References
- Weingart SN, Brown E, Bach PB, et al. NCCN Task Force Report: oral chemotherapy. J Natl Compr Canc Netw. 2008;6(suppl 3):S-1–S-14.
- O’Neill VJ, Twelves CJ. Oral cancer treatment: developments in chemotherapy and beyond. Br J Cancer. 2002;87:933-937.
- Liu G, Franssen E, Fitch M, Warner E. Patient preference for oral versus intravenous palliative chemotherapy. J Clin Oncol. 1997;15:110-115.
- Marin D, Bazeos A, Mahon FX, et al. Adherence is the critical factor for achieving molecular responses in patients with chronic myeloid leukemia who achieve complete cytogenetic responses on imatinib. J Clin Oncol. 2010;28:2381-2388.
- Jin J, Sklar GE, Min Sen Oh V, Chuen Li S. Factors affecting therapeutic compliance: a review from the patient’s perspective. Ther Clin Risk Manag. 2008;4:269-286.
- May P, LaPlant K, McGee A. Practice model: establishing and running an oral chemotherapy management clinic. Asia Pac J Oncol Nurs. 2017;4:299-303.
- Griffin MC, Gilbert RE, Broadfield LH, et al. Comparison of independent error checks for oral versus intravenous chemotherapy. J Oncol Pract. 2016;12:168-169, e180-e187.
- Mackler E, Scappaticci GB, Salgado TM, et al. Impact of a statewide oral oncolytic initiative on five participating practices. J Oncol Pract. 2018;14:e304-e309.
- McNamara E, Redoutey L, Mackler E, et al. Improving oral oncolytic patient self-management. J Oncol Pract. 2016;12:e864-e869.
- Mulkerin DL, Bergsbaken JJ, Fischer JA, et al. Multidisciplinary optimization of oral chemotherapy delivery at the University of Wisconsin Carbone Cancer Center. J Oncol Pract. 2016;12:e912-e923.
- Battis B, Clifford L, Huq M, et al. The impacts of a pharmacist-managed outpatient clinic and chemotherapy-directed electronic order sets for monitoring oral chemotherapy. J Oncol Pharm Pract. 2017;23:582-590.
- Weingart SN, Flug J, Brouillard D, et al. Oral chemotherapy safety practices at US cancer centres: questionnaire survey. BMJ. 2007;334:407. doi: 10.1136/bmj.39069.489757.55.
- American Society of Clinical Oncology Quality Oncology Practice Initiative. 2023 QOPI reporting tracks. https://practice.asco.org/sites/default/files/drupalfiles/2023-01/2023-QOPI-Reporting-Track-Public-Posting.pdf. Accessed March 29, 2013.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association best practices for the management of oral oncolytic therapy: pharmacy practice standard. J Oncol Pract. 2019;15:e346-e355.
- Vizient. 2022 Corporate Responsibility Report. www.vizientinc.com/-/media/documents/sitecorepublishingdocuments/public/Vizient_Corporate_Responsibility_Report.pdf. Accessed March 28, 2023.
- Vizient Newsroom. Members of Vizient Pharmacy Network Transform Health Care through Collaboration, Innovation and Best Practice. https://newsroom.vizientinc.com/en-US/releases/members-vizient-pharmacy-network-transform-health-care-through-collaboration-innovation-and-best-practices. Accessed April 2, 2023.
- Dürr P, Schlichtig K, Kelz C, et al. The randomized AMBORA trial: impact of pharmacological/pharmaceutical care on medication safety and patient-reported outcomes during treatment with new oral anticancer agents. J Clin Oncol. 2021;39:1983-1994.