Background: The incidence of tumor lysis syndrome (TLS) has been reported in 42% of adults with hematologic malignancies and can result in serious laboratory findings and clinical manifestations. The clinical manifestations may be severe, leading to dialysis therapy and/or death. The exact incidence of these severe outcomes has not been determined; however, strategies to determine the risk for these complications have been proposed.
Objective: To evaluate current risk categories and strategies to determine their ability to predict the incidence of dialysis or mortality secondary to TLS.
Methods: A total of 1327 patients with cancer who were identified by an internal registry database from the University of Kentucky Healthcare were assessed for risk of TLS based on current guidelines and were stratified into low-, intermediate-, or high-risk categories. These categories were assessed to determine if there is a difference in the incidence of dialysis and/or the incidence of mortality among the risk groups for TLS, and to determine if baseline population characteristics or laboratory abnormalities can predict severe patient outcomes.
Results: Of the 1327 patients evaluated, 6 (0.5%) had clinically severe outcomes secondary to TLS. Patients with high or intermediate risk were significantly more likely to have clinically severe outcomes compared with low-risk patients (2.98% vs 0.09%; P = .001). Predictors for severe events included male sex, age, diagnosis of Burkitt’s lymphoma, abnormal renal laboratory parameters, and categorization into higher-risk groups.
Conclusion: The overall incidence of clinically severe outcomes associated with TLS is low. However, higher-risk patients are at a significantly increased risk for dialysis or for mortality based on the results of the present study. Multiple laboratory and demographic factors should be considered when creating future predictive models for the clinical manifestations of TLS.
Tumor lysis syndrome (TLS) is a complication of cancer therapy that leads to multiple abnormal laboratory findings and clinical manifestations. The overall incidence of TLS has been reported to be 42% in adults with hematologic malignancies; however, the incidence of patients with clinical manifestations may be 5% to 10% or less, and may depend on the type of cancer.1-3 TLS with severe clinical consequences (ie, requirement of dialysis or mortality) may occur even less frequently, although the incidence has not been fully elucidated in a real-world setting.
TLS is caused by the acute release of cellular components into the blood after the rapid destruction of malignant cells.4,5 The abrupt lysis of tumor cells causes the release of electrolytes, particularly potassium and phosphorus, from intracellular compartments, causing systemic hyperkalemia and hyperphosphatemia. Hypocalcemia can also occur secondary to hyperphosphatemia.5,6 In addition, release of purine nucleotides that are metabolized to uric acid can accumulate and crystallize in renal tubules, which will overwhelm normal glomerular filtration processes and lead to renal insufficiency or renal failure.7,8 Renal failure further perpetuates electrolyte abnormalities, resulting in even larger increases in potassium and phosphorus. Collectively, the syndrome can lead to clinical manifestations that include seizures, cardiac abnormalities, neuromuscular instability, the need for dialysis, and even death.9
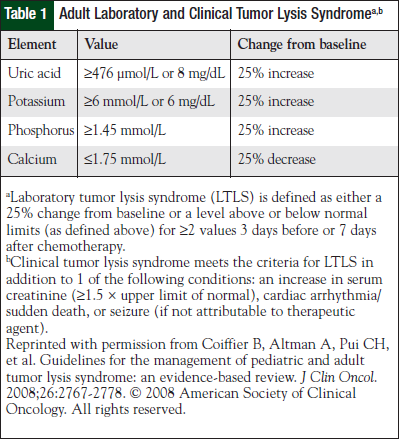
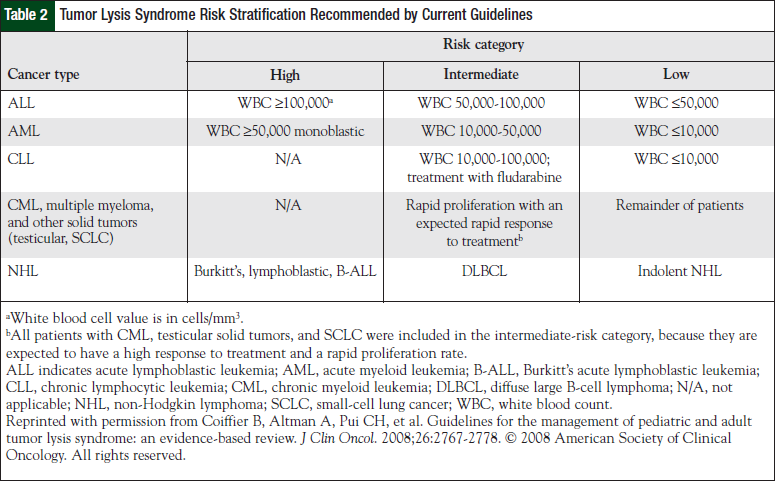
TLS may be classified as either a laboratory disorder (laboratory TLS [LTLS]) or as a laboratory disorder with clinical manifestations (clinical TLS [CTLS]). The current criteria for TLS are shown in Table 1.10 Guidelines have recently been proposed for the management of TLS in pediatric and adult patients for both laboratory and clinical manifestations.10 The guidelines support risk stratification among patients that is determined by cancer type and extent of disease (Table 2), as well as provide prophylaxis and treatment recommendations for each risk category.10
Protecting the kidneys is the primary focus of management, because the kidneys are essential to clearing the dying tumor’s metabolic products and electrolytes, which can accumulate to life-threatening concentrations. Because uric acid is the primary renal toxic product, minimizing the concentration in the renal tubule is paramount. This can be accomplished with allopurinol, which decreases the formation of uric acid through inhibition of xanthane oxidase, an enzyme that converts hypoxanthine and xanthine to uric acid.11,12
Alternatively, or in combination with allopurinol, rasburicase (recombinant urate oxidase) can be used, which metabolizes uric acid into the more water-soluble, nonrenally toxic allantoin.11,13-15 Hyperhydration is also often used; it provides benefit by diluting the concentration of uric acid in the renal tubule.10 The guidelines recommend that low-risk patients are monitored closely, intermediate-risk patients receive allopurinol and intravenous (IV) hydration, and high-risk patients receive recombinant urate oxidase and IV hydration. Select intermediate-risk patients in which hyperuricemia develops despite prophylactic allopurinol should also receive recombinant urate oxidase.10
Available evidence supports the risk stratification; however, the risk categories have not been adequately confirmed in a real-world setting.11,14,15 Current risk stratification methods have not been proved to predict the incidence of dialysis and mortality in patients at risk for TLS. The ability to predict patients at risk for renal failure and subsequent dialysis, for example, would be beneficial; it can be one of the most influential and costly manifestations of this disorder, because it increases mortality rates and length of hospital stay.2
The purpose of our study was to evaluate risk strategies and their ability to predict the incidence of clinically severe outcomes (eg, dialysis and mortality) secondary to TLS complications. This allowed us to determine if patients expected to be at higher risk according to proposed guidelines are more likely to suffer severe clinical outcomes secondary to TLS, and whether more aggressive and expensive pharmacologic management with recombinant urate oxidase should be considered. As a secondary analysis, we determined if selected pretreatment laboratory abnormalities translate into clinical outcomes and which factors are most predictive of clinically severe outcomes in patients at risk for TLS.
Patients and Methods
This study was designed as a retrospective review of patients at risk for TLS between January 1, 2005, and December 2, 2008, at a university hospital. The study was approved by the Institutional Review Board, and it adhered to appropriate policies and procedures. The primary end point was to determine if there is a difference in the incidence of dialysis and/or mortality in patients at low risk for developing TLS compared with those at high or intermediate risk based on current published guidelines for TLS risk. Secondary objectives were to determine if there is a difference in the incidence of dialysis and mortality between individual risk categories for TLS, and to determine if baseline population characteristics or laboratory abnormalities can predict these severe outcomes.
A total of 1327 patients with cancer were identified by an internal registry database and were risk stratified according to disease state and/or baseline white blood cell counts based on current guidelines (Table 2). Only the first antineoplastic treatment episode was considered for each individual, because the risk for TLS would be highest during the first course of treatment.
Administrative claims databases were used to determine if patients received dialysis or died after therapy. Medical charts, including inpatient and outpatient dialysis records, were analyzed for all database-identified patients to determine if death occurred within 1 month after treatment (consistent with TLS complications), or if they received dialysis within 2 weeks of therapy. These criteria were used to define a TLS-associated severe outcome. Patients were excluded only if they were aged ≤18 years. All therapies for the prevention or treatment of TLS were allowed in this study.
The incidence of severe clinical outcomes between the combined intermediate- and high-risk group were compared with those in the low-risk group, as well as between each individual risk group. When available, baseline laboratory and demographic data were collected to assess individual predictors for risk of clinical outcomes secondary to TLS compared with patients without these outcomes. Laboratory values were collected for 1 week before treatment, and the averages of the total values were considered baseline characteristics of each patient in the analysis.
Statistical Analysis
Demographic data and other patient characteristics were described using descriptive statistics. Differences between the intermediate- or high-risk category and the low-risk category were compared using a 2-tailed Fisher’s exact test. For analysis of differences between individual risk categories, a multivariate 2 × 3 Fisher’s exact test (2-tailed; 95% confidence interval) was performed, followed by a univariate analysis to detect differences between individual risk categories.
A logistic regression analysis was used to calculate the crude odds of characteristic variables predicting significant outcomes. A multivariate logistic regression model was then used to compare significantly different variables adjusted for the primary risk category to control for differences between risk groups. Binary covariates were encoded as 0 and 1, and cutoff values for continuous variables were not included. For all tests, P <.05 was considered significant. All statistical analyses were performed using STATA v10.1 (StataCorp; College Station, TX).
Results
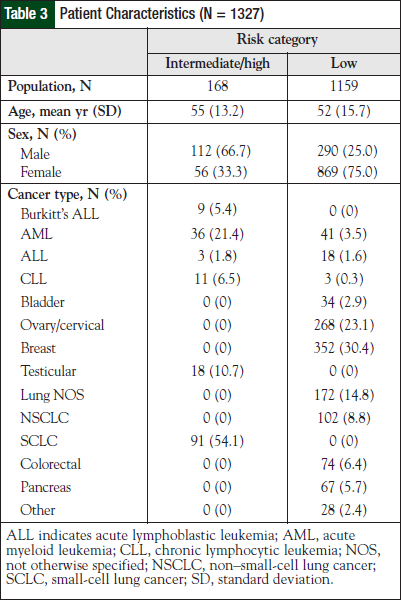
Of the 1327 patients who were included in the analysis, 168 patients were in the intermediate- or high-risk groups, and 1159 patients were in the low-risk group. Analysis of each individual risk group reveals that there were 1159 (87.4%), 141 (10.6%), and 27 (2.0%) individuals at low, intermediate, and high risk, respectively. Demographic information of the patients in the study is shown in Table 3.
View larger version
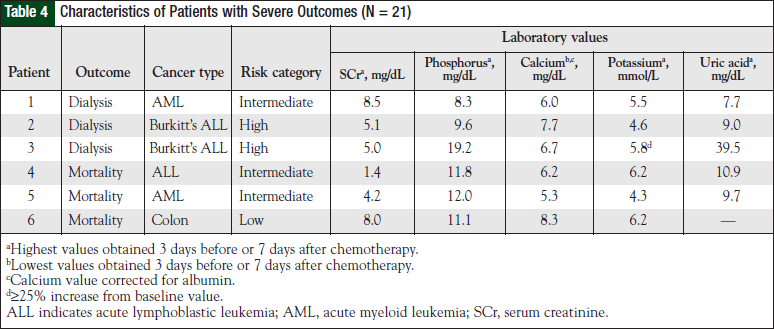
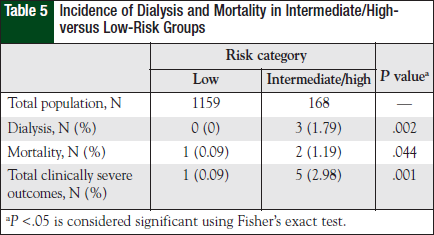
The overall incidence of clinically severe outcomes secondary to TLS was found in 6 (0.5%) patients (Table 4). Of these patients, 3 (0.2%) received dialysis and 3 died secondary to TLS complications (Table 5). Of the entire at-risk population, patients with intermediate or high risk were significantly more likely to have clinically severe outcomes compared with low-risk patients (0.09% vs 2.98%; P = .001). No patients had concomitant dialysis and mortality in the study.
With regard to secondary objectives, the incidence of dialysis occurred in 7.4%, 0.7%, and 0.0% of patients in the high-, intermediate-, and low-risk groups, respectively. Mortality occurred in 0.0%, 1.4%, and 0.09% of patients in the high-, intermediate-, and low-risk groups, respectively. Univariate analysis revealed that of the total population, the intermediate- and high-risk groups had more severe outcomes compared with the low-risk group (P = .001 and P = .005, respectively). Individual outcomes of dialysis and mortality also had statistically significant differences in the higher-risk group. Mortality incidence was significantly higher in the intermediate-risk group compared with that of the lowrisk group (P = .033), and the incidence of dialysis was significantly higher in the high-risk group compared with that of the low-risk group (P = .001).
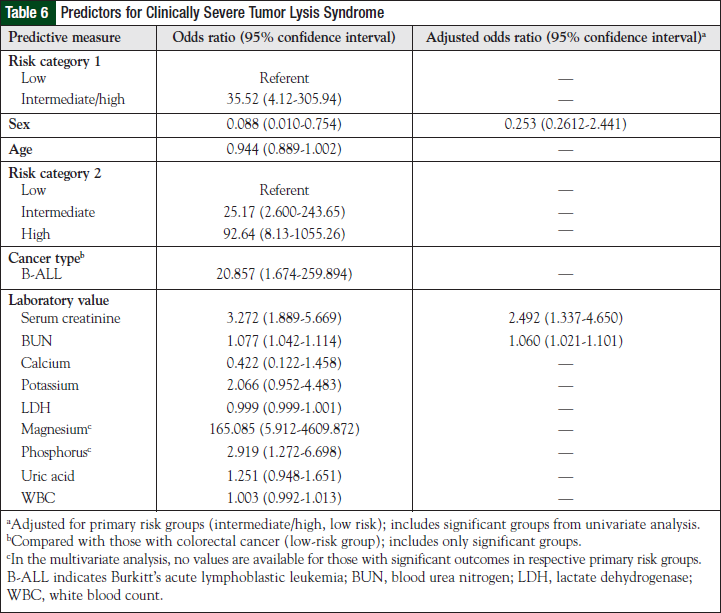
Baseline parameters most predictive of clinically significant outcomes based on the univariate analysis included male sex, age, and patients classified in the intermediate-, high-, and in the combined intermediateand high-risk groups. Patients with Burkitt’s lymphoma were most at risk compared with standard (ie, those with colorectal cancer) patients in the low-risk category. In addition, laboratory parameters, such as serum creatinine (SCr), blood urea nitrogen (BUN), magnesium, and phosphorus, were all significant predictors of clinically severe outcomes. When adjusting for the primary risk categories (intermediate-, high-, and low-risk groups), only SCr and BUN were significant predictors of severe TLS (Table 6).
Discussion
Since the initiation of our study, other risk stratification and treatment guidelines for TLS have been published.13,16,17 To our knowledge, however, this is the only study that has compared the risk for TLS with severe clinical outcomes according to current guidelines. Our study shows an overall low incidence of dialysis or mortality, regardless of risk category. Of the total at-risk population, only 0.2% of the patients received dialysis, and 0.2% died from TLS. This is very similar to data reported by Annemans and colleagues, in which the overall incidence of dialysis secondary to TLS was 1.3%, and 0.8% of the total population died from the consequences of TLS.2 Multiple other studies have also shown overall low rates of CTLS after treatment with chemotherapy, although most studies did not primarily assess clinically severe outcomes.1,3,18
Despite the overall low incidence of severe clinical outcomes in our study, it was confirmed that those most at risk (intermediate/high-risk group) had an increased incidence of clinically severe outcomes compared with those at low risk. Significant differences were found between these 2 groups for dialysis and for mortality. These findings support current recommendations for more aggressive prophylaxis (ie, recombinant urate oxidase) in higherrisk patients, despite increased costs associated with the therapy.10
However, there are no randomized, controlled studies that have fully analyzed the clinical benefit of aggressive management for the prevention of clinically severe outcomes in the setting of TLS. Urate oxidase has been shown to be safe and effective in reducing serum uric acid concentrations in various populations with overall low incidence of dialysis or mortality, but this has not been extensively compared with more conventional strategies, such as allopurinol or IV hydration.14,15,19,20
Only 1 patient died in the low-risk group. This patient had colorectal cancer and LTLS, with no other documented source of mortality according to available records. Despite this patient, analysis of the entire at-risk population reveals that the low-risk group had an overall lower incidence of clinically severe outcomes compared with the higher-risk groups. This reached statistical significance with dialysis compared with high-risk individuals and mortality with intermediate-risk individuals.
Predictive models for TLS are currently lacking. An analysis of patients with acute myeloid leukemia (AML), however, revealed that baseline lactate dehydrogenase, serum uric acid concentrations, and male sex were significant predictive values for TLS in this specific patient population.21 Montesinos and colleagues revealed increased risks of LTLS and CTLS in patients with AML with elevated SCr, uric acid, and white blood cell counts.3
Our univariate analysis did not reproduce identical results, because lactate dehydrogenase, serum uric acid, and white blood cell counts were not positive predictors of clinically severe TLS. However, male sex and SCr did predict the incidence of severe outcomes. In addition, baseline BUN, magnesium, and phosphorus were all positive predictors for clinically severe TLS.
With regard to cancer type, patients with Burkitt’s lymphoma were at highest risk for significant outcomes compared with those at low risk. Using data from the univariate analysis, our multivariate analysis revealed that BUN and SCr were again predictors for clinically significant TLS when adjusted for primary risk groups, suggesting that baseline renal dysfunction is an important factor in predicting severe clinical outcomes of TLS. Future studies should consider these risk predictors for the creation of predictive models.
Limitations There are limitations to our study. Our review is retrospective, and therefore prospective studies are encouraged to support validity of the data. In addition, collection methods for our data may have missed patients who have received dialysis at an outside institution.
Our results also showed a low number of patients with severe outcomes, which limits our ability to appropriately incorporate the regression analysis and enhances our probability of error.
In addition, our data do not assess the influence of therapies on the incidence of TLS. Patients were not stratified by risk according to therapies received for TLS prevention and treatment, although allopurinol use in higher-risk patients is the standard approach, and the overall use of recombinant urate oxidase therapy is low at our institution.
Finally, for a majority of the patients, our data did not include the possibility of concomitant nephrotoxic agents that might have been utilized before or during the period of active tumor lysis. Patients found to have severe outcomes, however, did not have any evidence of the use of medications that significantly affect renal function.
Conclusion
Despite these limitations, our study provides evidence that higher-risk populations truly are at higher risk for severe CTLS. As a result, more aggressive therapies, such as recombinant urate oxidase, may need to be prescribed to prevent these outcomes in select intermediate- or high-risk populations as current guidelines suggest. In addition, multiple laboratory and demographic factors should be considered when creating predictive models for CTLS. Based on our findings, we encourage future randomized, prospective trials to analyze current therapies and their abilities to prevent and treat severe clinical outcomes in the setting of high-risk TLS.
Author Disclosure Statement
Dr Sutphin is on the advisory board of Amgen. Dr Wirth, Dr Steinke, Dr Lawson, Dr Blechner, and Dr Adams have reported no conflicts of interest.
References
- Hande KR, Garrow GC. Acute tumor lysis syndrome in patients with high-grade non-Hodgkin’s lymphoma. Am J Med. 1993;94:133-139.
- Annemans L, Moeremans K, Lamotte M, et al. Incidence, medical resource utilisation and costs of hyperuricemia and tumour lysis syndrome in patients with acute leukaemia and non-Hodgkin’s lymphoma in four European countries. Leuk Lymphoma. 2003;44:77-83.
- Montesinos P, Lorenzo I, Martin G, et al. Tumor lysis syndrome in patients with acute myeloid leukemia: identification of risk factors and development of a predictive model. Haematologica. 2008;93:67-74.
- Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol. 2004;127:3-11.
- Fleming DR, Doukas MA. Acute tumor lysis syndrome in hematologic malignancies. Leuk Lymphoma. 1992;8:315-318.
- Jeha S. Tumor lysis syndrome. Semin Hematol. 2001;38(4 suppl 10):4-8.
- Wolf G, Hegewisch-Becker S, Hossfeld DK, Stahl RA. Hyperuricemia and renal insufficiency associated with malignant disease: urate oxidase as an efficient therapy? Am J Kidney Dis. 1999;34:E20.
- Arrambide K, Toto RD. Tumor lysis syndrome. Semin Nephrol. 1993;13:273-280.
- Davidson MB, Thakkar S, Hix JK, et al. Pathophysiology, clinical consequences, and treatment of tumor lysis syndrome. Am J Med. 2004;116:546-554.
- Coiffier B, Altman A, Pui CH, et al. Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence-based review. J Clin Oncol. 2008;26:2767- 2778.
- Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97:2998-3003.
- Hande KR, Hixson CV, Chabner BA. Postchemotherapy purine excretion in lymphoma patients receiving allopurinol. Cancer Res. 1981;41:2273-2279.
- Pession A, Masetti R, Gaidano G, et al. Risk evaluation, prophylaxis, and treatment of tumor lysis syndrome: consensus of an Italian expert panel. Adv Ther. 2011;28:684-697.
- Pui CH, Mahmoud HH, Wiley JM, et al. Recombinant urate oxidase for the prophylaxis or treatment of hyperuricemia in patients with leukemia or lymphoma. J Clin Oncol. 2001;19:697-704.
- Coiffier B, Mounier N, Bologna S, et al. Efficacy and safety of rasburicase (recombinant urate oxidase) for the prevention and treatment of hyperuricemia during induction chemotherapy of aggressive non-Hodgkin’s lymphoma: results of the GRAAL1 (Groupe d’Etude des Lymphomes de l’Adulte Trial on Rasburicase Activity in Adult Lymphoma) study. J Clin Oncol. 2003;21:4402-4406.
- Tosi P, Barosi G, Lazzaro C, et al. Consensus conference on the management of tumor lysis syndrome. Haematologica. 2008;93:1877-1885.
- Cairo MS, Coiffier B, Reiter A, Younes A. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. Br J Haematol. 2010;149:578-586.
- Cheson BD, Frame JN, Vena D, et al. Tumor lysis syndrome: an uncommon complication of fludarabine therapy of chronic lymphocytic leukemia. J Clin Oncol. 1998;16:2313-2320.
- Teo WY, Loh TF, Tan AM. Avoiding dialysis in tumour lysis syndrome: is urate oxidase effective?—a case report and review of literature. Ann Acad Med Singapore. 2007;36:679-683.
- Pui CH, Relling MV, Lascombes F, et al. Urate oxidase in prevention and treatment of hyperuricemia associated with lymphoid malignancies. Leukemia. 1997;11:1813-1816.
- Mato AR, Riccio BE, Qin L, et al. A predictive model for the detection of tumor lysis syndrome during AML induction therapy. Leuk Lymphoma. 2006;47:877-883.