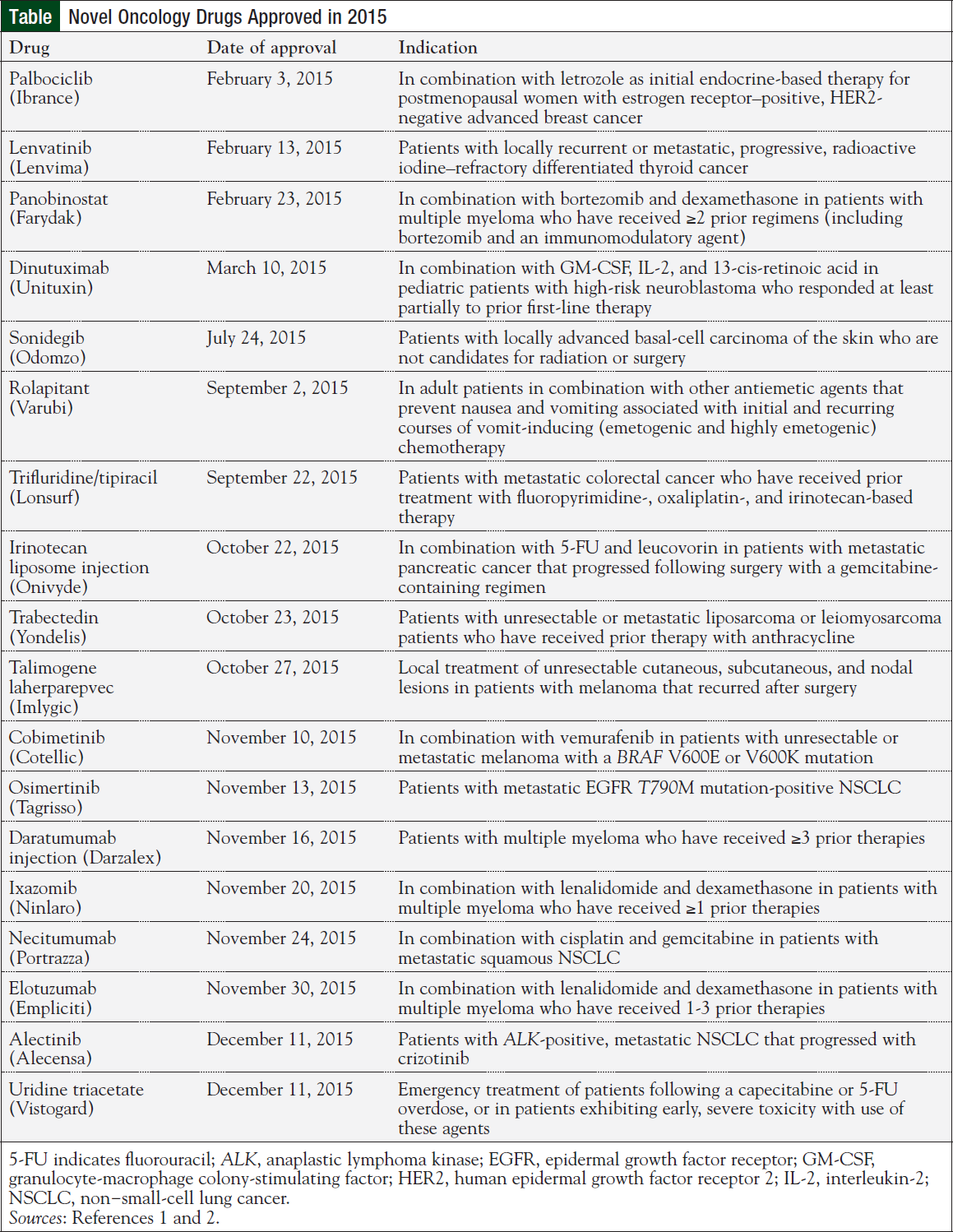
The number of oncology trials seeking new indications for new and known agents is continuing to grow at a fevered pitch. The fruition of this work is now being realized; in the past 12 months, there have been 36 hematology/oncology drug approvals, 18 of which involved new drug entities (Table).1,2 The majority of these agents are targeted agents, which are most effective and approved for a limited population of patients with cancer.
The origin of targeted therapy starts with the drug discovery process. Targeted therapies are created to disrupt a specific molecular target that is important to cancer cell growth, progression, and metastasis.3 With targeted therapies, the desired pharmacology (ie, molecular target) is identified before the compounds are created, whereas, with traditional drug discovery, older cancer drugs arose from the screening of libraries of compounds for activity, then solving the pharmacology.4,5
Clinically, targeted therapies should offer the advantage of better efficacy and less toxicity. The inherent benefit is in the early identification of a biomarker that can be used to select patients who will respond to the targeted treatment; identifying patients who will or will not benefit from treatment also translates to higher efficacy rates. Because many of these drugs target signaling pathways, phase 1 studies can target a dose that inhibits signaling through that pathway—most commonly a phosphorylation event. This approach allows for fewer toxic doses compared with traditional cytotoxic phase 1 trials that seek to determine the maximum tolerated dose.
The key to targeted therapy success is identification of a good molecular target.3 Benchtop scientists have been studying tumor cell biology for decades. Use of this knowledge to target tumors has continued to pick up pace in the past 20 years with improved molecular tools and proteomics. Efforts to translate this science into patient care and improved outcomes took some time; imatinib was arguably the first high-impact targeted therapy approved.
However, the new drugs (Table)—including those not listed but—approved in the past 12 months are clear evidence that the enormous amount of money and energy spent developing new targeted therapies is being translated into patient care.
As oncology pharmacists, it is crucial that we stay abreast of these new drugs to ensure that they are used in the appropriate patient populations, and that toxicity is prevented or quickly recognized and managed.
References
- US Food and Drug Administration. Hematology/oncology (cancer) approvals & safety notifications. www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm279174.htm. Updated March 30, 2016. Accessed March 31, 2016.
- US Food and Drug Administration. FDA approves new drug treatment for nausea and vomiting from chemotherapy. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm460838.htm. Updated September 2, 2016. Accessed April 4, 2016.
- National Cancer Institute. Targeted cancer therapies. www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet. Reviewed April 25, 2014. Accessed March 31, 2016.
- Lord CJ, Ashworth A. Biology-driven cancer drug development: back to the future. BMC Biology. 2010;8:38.
- Parulekar WR, Eisenhauer EA. Phase I trial design for solid tumor studies of targeted, non-cytotoxic agents: theory and practice. J Natl Cancer Inst. 2004;96:990-997.