The use of oral antineoplastic therapy presents many challenges in the inpatient setting.1 The handling of oral antineoplastic drugs can place patients and caregivers at risk for exposure to hazardous medications without proper precautions.1 Complex regimens and dosing schemes increase the risks for prescribing and administration errors.2 Drug–drug interactions among medications used for acute care can also result in adverse events or decreased drug efficacy.3 Food–drug interactions may affect a patient’s overall exposure to oral antineoplastic drugs.4 These challenges could be even more complicated in the inpatient setting than in the outpatient setting because of fluctuating patient acuity.
Several national organizations have addressed these challenges by implementing guidelines for the safe use of oral antineoplastic drugs. For example, the American Society of Clinical Oncology (ASCO) and the Oncology Nursing Society (ONS) recommend including the full schedule, duration of therapy, and dose calculations on medication orders to reduce dispensing and administration errors.5 The American Society of Health-System Pharmacists (ASHP) further discusses how drug labels should also include supplemental drug administration instructions, including starting and completion dates, cautionary information regarding when the medications should be taken in relation to food and other medications, and drug handling precautions.6 Although these recommendations provide the best practices for the initial prescribing of drugs, they do not fully describe safeguards during transitions of care.5,6
Continuing oral antineoplastic therapy in the inpatient setting presents new safety and financial concerns.7 Complex antineoplastic regimens may be difficult to capture through medication reconciliation during admission, which can increase potential prescribing and administration errors.8 In addition, antineoplastic agents are being approved by the US Food and Drug Administration (FDA) at increasing rates.1 Acute care centers do not have the capacity to keep all oral antineoplastic agents on their formulary, because of these drugs’ high costs, and they may therefore have to use the patient’s own drug supply.
Although The Joint Commission addresses patients’ own medication use in standard MM.03.01.05, that standard only mandates that hospitals identify the medication and visually evaluate the integrity of the drug, and leaves the hospital to define its responsibilities for safe use.9 Thus, the use of a patient’s own medications could circumvent standard institutional safeguards, such as automated drug interaction screening, prepopulated labeling instructions, and automated dose checking.9,10
Healthcare workers can also be unintentionally exposed to hazardous oral antineoplastic agents during administration. All oral antineoplastic drugs should be assessed and labeled appropriately with precautions for hazardous medications.5,11,12 Nurses can then wear appropriate personal protective equipment (PPE) and decrease the risk for exposure to hazardous drugs.5,12 Institutions must find a system for alerting healthcare workers to potential hazards when using a patient’s own drug supply.
To address these concerns, our institution created nonbillable patient’s own antineoplastic medication drug files between July and September 2018. Now postimplementation, when a prescriber places an order to continue the patient’s own oral antineoplastic agent, a pharmacist would change the order to a medication-specific patient’s own antineoplastic medication drug file instead of verifying a generic patient’s own medication drug file.
Changing the generic patient’s own medication drug file to a medication-specific patient’s own antineoplastic medication order was done by pharmacists per TriStar Centennial Medical Center’s protocol and required dual pharmacist verification. Dosage form, including dose strength, had to be selected so that orders had the specific dose reported on the electronic medication administration record. Before the creation of a patient’s own antineoplastic medication drug files, the doses were reported as “1 each” or “2 each” for quantity, and often were not associated with an actual dosing strength (ie, mg dosing).
The medication-specific patient’s own antineoplastic medication drug files were created by an informatics pharmacist in collaboration with oncology pharmacists. The patient’s own antineoplastic medication drug files were created by modifying already existing drug files in the center’s database. By using the existing database, drug–drug interactions and drug dosing alerts would then signal on order verification. The patient’s own antineoplastic medication drug files were also modified to be nonbillable.
The center’s database’s drug files do not have any standardized, prepopulated label comments. As was recommended by the oncology pharmacists, standardized label comments were added for each medication-specific drug file to include handling precautions for hazardous medications, drug–food considerations, and other comments as applicable. Examples of labels before and after the implementation of the standardized antineoplastic drug files are provided online (Appendix Figure, available at the top of the article).
In this study, we evaluated the overall impact of implementing standardized files for a patient’s own antineoplastic medication on our ability to continue the use of oral antineoplastic drugs safely in the inpatient setting.
Methods
This single-center, retrospective, comparative study was conducted in a tertiary community medical center at TriStar Centennial Medical Center in Nashville, TN, which is associated with a cancer center and a research institute. This study was approved by our Institutional Review Board. The data were obtained from the electronic medical record (EMR) system for a patient’s own antineoplastic medication from January 2018 to June 2018 (ie, the preimplementation period) and from October 2018 to December 2018 (ie, the postimplementation period). Accordingly, patients were divided into a preimplementation and a postimplementation arm and the practice change was implemented between July and September 2018.
Patients were included if they were aged ≥18 years and had an order for a patient’s own antineoplastic drug. Patients were excluded if they received treatment from the pediatric oncology team, regardless of age, or if the patient’s own medication was an investigational agent. Patients who received oral chemotherapy agents for nononcologic indications (eg, hydroxyurea, methotrexate) were also excluded from the study. Patients who started taking antineoplastic drugs listed on the formulary, such as midostaurin, dasatinib, or venetoclax, were also excluded, because these were not the patient’s own supply.
The patients’ demographic data we collected included age, sex, hospital admitting location, hospital length of stay, and cancer diagnosis. The antineoplastic drug orders were reviewed for appropriateness, by collecting the name of the medication, drug administration dates, dosage form, drug strength, frequency of administration, dose, and food instructions. Concurrent medications were documented and were evaluated for drug interactions. Laboratory and additional monitoring parameters (eg, having an electrocardiogram) recommended by the drug’s prescribing information were collected and were evaluated for appropriateness.
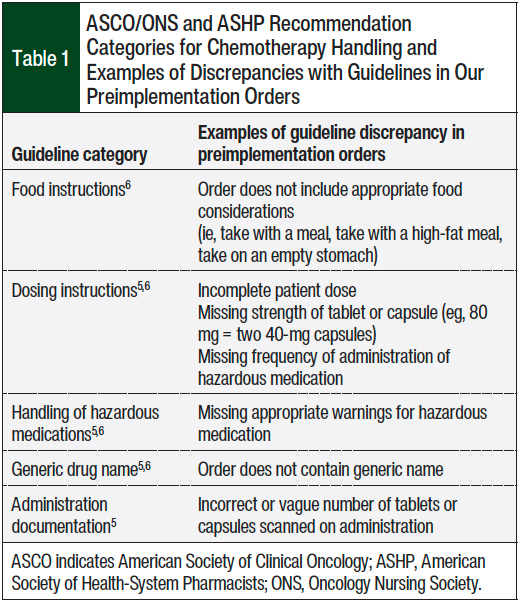
The study’s primary outcome was to compare the total number and type of discrepancies in the drug files before and after the implementation of the patient’s own antineoplastic medication in relation to the ASCO/ONS and ASHP recommendations.5,6 We classified the guideline recommendations into 5 categories, including food instructions, dosing instructions, handling of hazardous medications, drug’s generic name, and appropriate administration documentation (Table 1).5,6
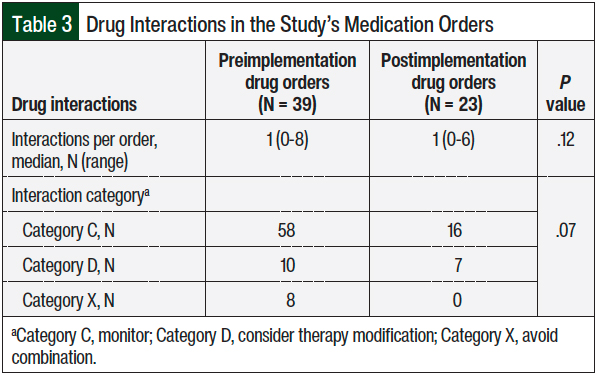
The secondary outcomes included screening for potential drug–drug interactions, as well as classifying the category of drug–drug interactions. Drug–drug interactions were categorized as follows: Category C, monitor; Category D, consider therapy modification; or Category X, avoid combination via a drug–drug interaction checker.
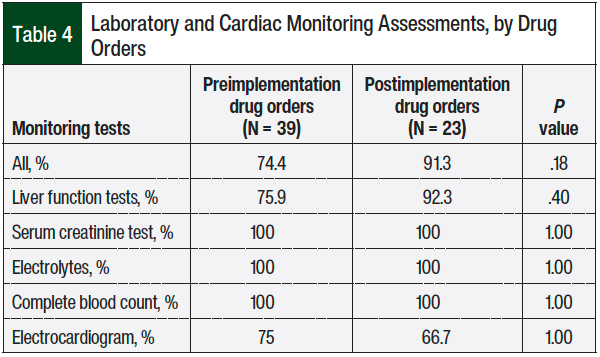
Additional secondary outcomes included evaluating if the recommended monitoring parameters (ie, liver function panel, renal panel, electrocardiogram, and complete blood count) were ordered at baseline. Descriptive statistics were used in the study for all baseline characteristics and categorical data, and to describe the frequency of medication adherence, drug interactions, and frequency of laboratory monitoring. Fisher’s exact test was used to assess statistical significance for categorical data. A Mann–Whitney U test was used to compare the continuous data between the 2 study groups.
Results
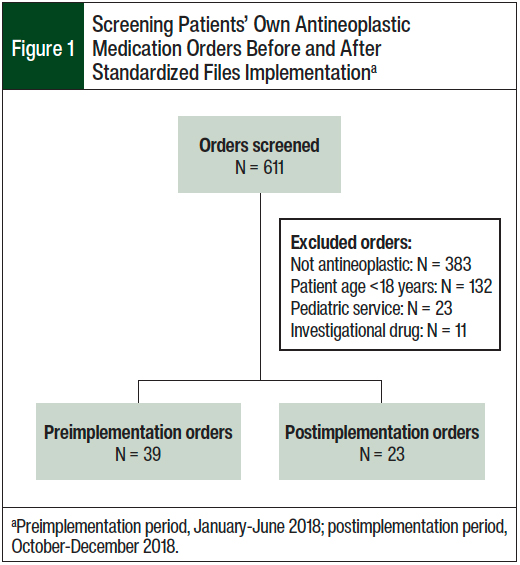
A total of 611 patient’s own medication orders were screened for study eligibility. The primary reason for order exclusion was that the ordered medication was not an antineoplastic drug. In all, a total of 39 medication orders (for 30 patients) were included for analysis in the preimplementation arm and 23 medication orders (for 17 patients) were included in the postimplementation arm. Figure 1 outlines the screening of a patient’s own medication orders that were included and excluded in this study.
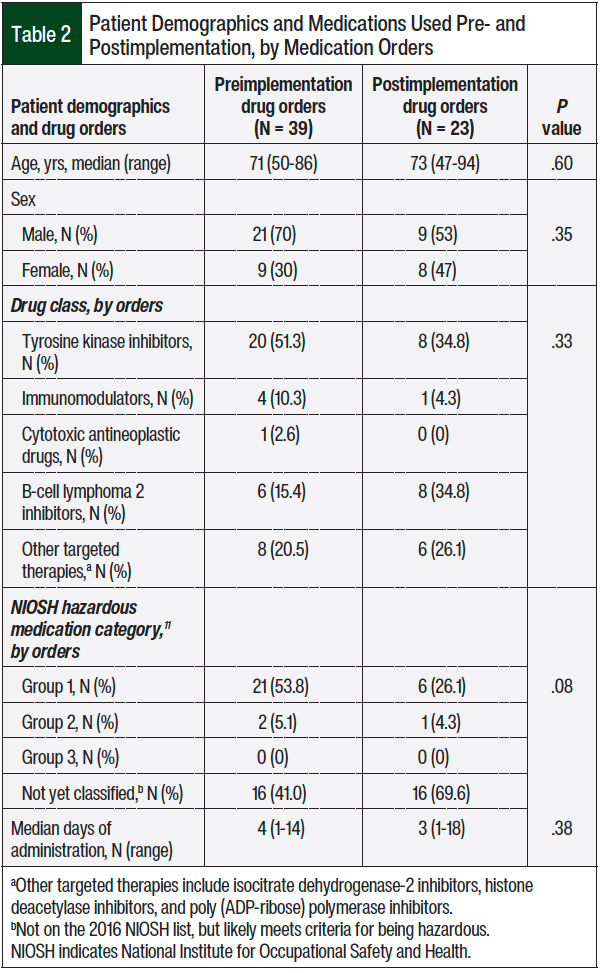
The patients’ age and sex were similar in the 2 groups (Table 2). The majority of the medications included in the study were the patient’s own tyrosine kinase inhibitors that were nonformulary, followed by B-cell lymphoma 2 inhibitors and other targeted agents. Most of the drugs were not yet classified by the National Institute for Occupational Safety and Health (NIOSH) because of their recent approval by the FDA, but these medications may still be considered hazardous, based on their structure and mechanism of action that are similar to other hazardous antineoplastic drugs.11
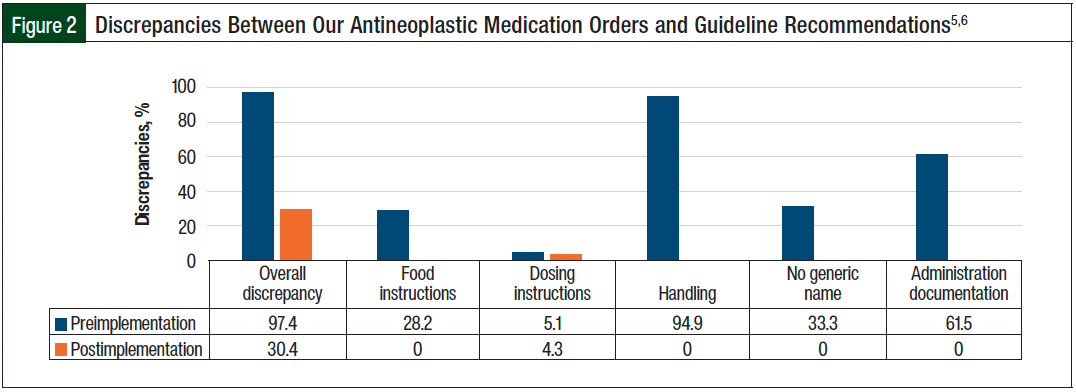
The use of a patient’s own antineoplastic medication drug files postimplementation significantly decreased the percentage of orders with discrepancies in relation to the guidelines5,6 compared with these drug files preimplementation (30.4% vs 97.4%, respectively; P <.001).
Specifically, compared with orders preimplementation, the patient’s own antineoplastic medication standardized drug files postimplementation decreased the medication orders without food instructions (28.2% vs 0%, respectively; P <.001), without generic names (33.3% vs 0%, respectively; P = .001), and without hazardous handling precautions (94.9% vs 0%, respectively; P <.001). The use of the patient’s own antineoplastic medication drug files also reduced the discrepancy with the guideline recommendations5,6 in terms of administration documentation (61.5% vs 0%, respectively; P <.001). No significant difference was seen in the percentage of orders with incomplete dosing instructions between the 2 arms (5.1% vs 4.3%, respectively; P = 1; Figure 2).
There were no differences in the median number of drug interactions per medication order (1 vs 1; P = .12) and in the drug interaction severity classification (ie, monitor, consider therapy modification, avoid combination) between the 2 arms (Table 3).
The patients’ baseline laboratory and cardiac monitoring assessments were higher in the postimplementation arm than in the preimplementation arm, although this was not statistically significant (91.3% vs 74.4%, respectively; P = .18; Table 4). Overall, the rates of monitoring were high in both groups.
Discussion
Using nonbillable, standardized drug files decreased the percentage of medication orders that had a discrepancy with the guideline recommendations.5,6 The drug files also alerted nurses and healthcare providers about potentially hazardous antineoplastic drug exposure during administration and improved proper documentation in the electronic medication administration record.
Before implementation of the drug files, drug doses would display as “1 each” or “2 each” on the electronic medication administration record, but it would be hard to distinguish if “1 each” meant 1 tablet or 1 dose. One dose could be multiple tablets; thus, the administration documentation was often vague and hard to interpret correctly. After the implementation of the drug files, the electronic medication administration record would display the actual dose (ie, mg) administered. Because antineoplastic medications are not readily available on hospital formularies, and the use of a patient’s own antineoplastic medication is anticipated to increase, these standardized drug files present a possible solution to this complex problem.
Given the recent focus on the safe handling of hazardous medications in the US Pharmacopoeia <800>,12 it is imperative that appropriate warnings are included on the medication’s label. Exposure to antineoplastic agents can result in negative side effects, including skin rashes, an increased risk for cancer, and reproductive adverse events.11 In our study, the implementation of standardized drug files ensured that all medications had hazardous drug warning labels. These drug files also alerted healthcare workers about the potential for hazardous medications, which may lead to increased appropriate PPE use.
Another area of concern is identifying potential drug interactions with oral antineoplastic medications. Drug interactions have resulted in treatment nonresponse, increased healthcare costs, and even increased mortality.13 The database that was integrated into the EMR at TriStar Centennial Medical Center used for the patient’s own antineoplastic medication orders was not the same as the interaction checker used in the retrospective analysis of the patient’s own generic medication orders. However, the database that was integrated into the EMR did display the same levels of drug interactions, including Category C, Category D, and Category X drug interactions.
Although the difference between pre- and postimplementation of the drug files was not significant, no contraindicated drug interactions were found after the implementation of the standardized antineoplastic drug files. The lack of statistical difference is likely a result of the small sample size in this study. However, thanks to their ability to screen for drug interactions with the implementation of standardized drug files, prescribers and pharmacists can now intervene and make appropriate recommendations for dose adjustments or recommend alternative therapies, which could improve patient safety and medication efficacy.
The goal of this study was to evaluate the safety of a patient’s own antineoplastic medication orders when these replaced a patient’s own generic medication order. The strengths of this study include the use of nationally recognized recommendations from ASCO/ONS5 and ASHP6 to evaluate patients’ medication orders for discrepancies between these recommendations and previous practice. In our study, the safety and monitoring parameters were standardized and were evaluated according to the recommendations in the prescribing information for each nonformulary antineoplastic drug. The patient’s own antineoplastic medication orders required a pharmacist’s double check to decrease the risk for error.
There are several practical considerations for the use of this strategy to enhance patient and healthcare worker safety. Close collaboration with informatics pharmacists was absolutely vital to the success of the implementation of standardized antineoplastic medication files. Oncology pharmacists created an extensive list with label comments of the most frequently used antineoplastic drugs for informatics pharmacists to create the patient’s own antineoplastic drug files.
Several in-services and education handouts were provided to the pharmacy department before the process could be implemented, which occurred from July to September 2018. If a patient’s own medication came across for order verification but did not have a medication-specific drug file, an oncology pharmacist would be contacted. The oncology pharmacist would create appropriate label comments and notify the informatics pharmacists about the need to create a new patient’s own antineoplastic drug file. The creation of new drug files would typically take 1 to 2 weeks.
Limitations
The limitations of this study include its retrospective nature, the small sample size, and the recent implementation of the standardized drug files. Because of these constraints, the data collection period ended in December 2018. The close proximity to the time of the standardized files implementation and the data collection might have heightened awareness of pharmacists during order verification, and might have increased the potential for bias because of increased vigilance toward these antineoplastic orders.
There is also a risk for errors related to order verification when pharmacists substitute the patient’s own generic medication with the patient’s own antineoplastic medication. To prevent an increased risk for such transcribing errors when the drug file was changed, the patient’s own antineoplastic medications required a pharmacist’s double check.
In addition, we were not able to assess in this study healthcare workers’ compliance with the use of appropriate PPE. At the time of the data collection and submission to publication, the NIOSH List of Hazardous Drugs in Healthcare Settings 2018 was still in draft form and was not yet finalized. The data from this study were analyzed per the published NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2016.11 Currently, several 2020 draft versions are available,14-16 which can help to provide further guidance on managing hazardous drugs in the healthcare setting.
Furthermore, we did not include or evaluate investigational antineoplastic drugs, which may also have drug interactions or carry hazardous risks for healthcare workers. Formulary antineoplastic drugs, such as certain tyrosine kinase inhibitors and venetoclax for specific indications, were also excluded from this study. Formulary antineoplastic drugs are entered as part of the chemotherapy regimen at TriStar Centennial Medical Center and are added to the electronic medication administration record with billable, standardized drug files.
Moreover, we did not find any significant difference in the appropriateness of ordering baseline laboratory tests or cardiac assessments between the 2 groups. This lack of difference may be explained by patient hospitalization and acuity of illness, resulting in thorough laboratory and cardiac monitoring as the standard of care.
Conclusion
The results of this study indicate that the use of standardized drug files for antineoplastic medications is a reasonable strategy to improve patient and healthcare worker safety when using a patient’s own oral (ie, nonformulary) antineoplastic drugs in the inpatient setting.
The next steps of our project are to allow a patient’s own antineoplastic medication orders to be orderable via computerized physician order entry, instead of only the patient’s own generic medication orders, to decrease the need for pharmacists to substitute drug files.
Additional studies are needed to evaluate the strategies for safe handling of hazardous medications and decreasing discrepancies with guideline recommendations to apply during transitions of care. All institutions should consider reviewing and creating safety mechanisms for continuing the use of outpatient antineoplastic medications in the inpatient setting.
Acknowledgment
This research was supported by HCA Healthcare and/or an HCA Healthcare–affiliated entity. The views expressed in this article represent those of the authors and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Author Disclosure Statement
Dr Li, Dr Parsons, Dr Siler, and Dr Matthews have no conflicts of interest to report.
References
- Weingart SN, Brown E, Bach PB, et al. NCCN Task Force Report: oral chemotherapy. J Natl Compr Canc Netw. 2008;6(suppl 3):S1-S14.
- Weingart SN, Toro J, Spencer J, et al. Medication errors involving oral chemotherapy. Cancer. 2010;116:2455-2464.
- van Leeuwen RWF, Brundel DHS, Neef C, et al. Prevalence of potential drug–drug interactions in cancer patients treated with oral anticancer drugs. Br J Cancer. 2013;108:1071-1078.
- Segal EM, Flood MR, Mancini RS, et al. Oral chemotherapy food and drug interactions: a comprehensive review of the literature. J Oncol Pract. 2014;10:e255-e268.
- Neuss MN, Polovich M, McNiff K, et al. 2013 updated American Society of Clinical Oncology/Oncology Nursing Society chemotherapy administration safety standards including standards for the safe administration and management of oral chemotherapy. J Oncol Pract. 2013;9(2 suppl):5s-13s. Erratum in: J Oncol Pract. 2013;9:265.
- Goldspiel B, Hoffman JM, Griffith NL, et al. ASHP guidelines on preventing medication errors with chemotherapy and biotherapy. Am J Health Syst Pharm. 2015;72:e6-e35.
- Mulkerin DL, Bergsbaken JJ, Fischer JA, et al. Multidisciplinary optimization of oral chemotherapy delivery at the University of Wisconsin Carbone Cancer Center. J Oncol Pract. 2016;12:e912-e923.
- Bartel SB. Safe practices and financial considerations in using oral chemotherapeutic agents. Am J Health Syst Pharm. 2007;64(suppl 5):S8-S14.
- Uselton JP, Kienle PC, Murdaugh LB. Assuring Continuous Compliance with Joint Commission Standards: A Pharmacy Guide. 8th ed. Bethesda, MD: American Society of Health-System Pharmacists; 2010.
- Norstrom PE, Brown CM. Use of patients’ own medications in small hospitals. Am J Health Syst Pharm. 2002;59:349-354.
- National Institute for Occupational Safety and Health/Centers for Disease Control and Prevention. NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, Publication No 2016-16. September 2016. www.cdc.gov/niosh/docs/2016-161/pdfs/2016-161.pdf?id=10.26616/NIOSHPUB2016161. Accessed August 24, 2021.
- Gabay M. USP <800>: handling hazardous drugs. Hosp Pharm. 2014;49:811-812.
- Sharma M, Holmes HM, Mehta HB, et al. The concomitant use of tyrosine kinase inhibitors and proton pump inhibitors: prevalence, predictors, and impact on survival and discontinuation of therapy in older adults with cancer. Cancer. 2019;125:1155-1162.
- National Institute for Occupational Safety and Health/Centers for Disease Control and Prevention. Draft. NIOSH List of Hazardous Drugs in Healthcare Settings, 2020. www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT-NIOSH-Hazardous-Drugs-List-2020.pdf. Accessed August 24, 2022.
- National Institute for Occupational Safety and Health/Centers for Disease Control and Prevention. Draft. Managing Hazardous Drug Exposures: Information for Healthcare Settings, 2020. www.cdc.gov/niosh/docket/review/docket233c/pdfs/DRAFT-Managing-Hazardous-Drug-Exposures_Information-for-Healthcare-Settings.pdf. Accessed August 24, 2022.
- National Institute for Occupational Safety and Health/Centers for Disease Control and Prevention. Draft. Procedures for Developing the NIOSH List of Hazardous Drugs in Healthcare Settings. Version February 24, 2020. https://downloads.regulations.gov/CDC-2020-0046-0002/content.pdf. Accessed August 24, 2022.