Dual immune checkpoint inhibitors—such as cytotoxic T-lymphocyte–associated protein 4 (CTLA-4) inhibitors and programmed cell death 1 (PD-1) or PD ligand 1 (PD-L1) inhibitors—are now approved in multiple malignancies, including melanoma, renal-cell carcinoma, non–small-cell lung cancer, hepatocellular carcinoma, and microsatellite instability-high solid tumors, such as colorectal cancer.1 As these agents are being used more frequently by oncologists, we are also seeing a wide spectrum of immune-related adverse events (irAEs).
The CheckMate-067 and the CheckMate-069 clinical trials compared the use of combination checkpoint inhibitors versus a single checkpoint inhibitor, showing that grade 3 or 4 adverse events increased with dual agents compared with a single checkpoint inhibitor; however, these studies did not report cases of hematologic adverse events.2,3
Hematologic irAEs, such as anemia, neutropenia, and thrombocytopenia, have been reported in other clinical trials with dual immune checkpoint inhibitors, showing an overall hematologic adverse event rate of approximately 4%.4 The more frequently reported hematologic toxicities with immune checkpoint inhibitors are neutropenia, autoimmune hemolytic anemia, and autoimmune thrombocytopenia.4
Pure red-cell aplasia (PRCA) is a rare event. Delanoy and colleagues reviewed 3 databases and screened 948 patients with a variety of malignancies, of whom 35 had hematologic adverse events, but only 1 case of PRCA.5 Davis and colleagues examined more than 16 million reported irAEs with checkpoint inhibitors, which included 168 hematologic adverse events; of these adverse events, only 7 patients had PRCA, and 6 of them had melanoma.6 The onset of hematologic adverse events occurred earlier with a CTLA-4 inhibitor than with a PD-1 or PD-L1 inhibitor.
Currently, no standard therapy is available for steroid-refractory immune checkpoint inhibitor–mediated PRCA. Similarly, immune checkpoint inhibitor–mediated diabetes is uncommon and is usually managed with insulin, because its pathogenesis is similar to type 1 diabetes.7
Case Report
We present a 49-year-old man with a history of hypertension and vitiligo who was diagnosed with left-ocular melanoma after he complained of blurry vision when he would look up and to the left. The patient underwent enucleation of his left eye in April 2016, and was diagnosed with T3b ciliochoroidal melanoma in his left eye. Testing for BRAF mutation was negative. Positron emission tomography (PET) scanning at the time of diagnosis revealed no other sites of disease.
In spring 2018, the patient started to have neck and lower back pain. In May 2018, a PET scan revealed a hypermetabolic lesion, measuring 1.3 × 1.4 × 1.9 cm, in segment VII of the liver, as well as a hypermetabolic lesion in the posterior aspect of the L1 vertebra. A biopsy of the L1 vertebra revealed malignant cells consistent with metastatic uveal melanoma.
The patient started therapy with nivolumab 240 mg every 2 weeks in August 2018, and in September 2018 he received palliative radiation to the L1 spine (35 Gy/12 fractions) for pain control. In January 2019, a PET scan revealed a new hypermetabolic area in the right iliac bone, stable disease in the L1 vertebra, and no activity in the liver. This was deemed to be a mixed response, and, because the patient had no new symptoms, no change was instituted in his management.
A repeated PET scan in August 2019 revealed a new right frontal lobe mass measuring 2.3 × 1.9 cm, and increasing metabolic activity in the right iliac spine. Magnetic resonance imaging (MRI) of the brain confirmed the finding of the PET scan of a right frontal lobe mass, which measured 2.7 × 2.6 × 2.2 cm on the MRI. Aside from mild chronic lower back pain, the patient had no new bone pain or neurologic symptoms.
He was deemed to have progression of his melanoma and underwent hypofractionated stereotactic radiosurgery to the right frontal lobe mass (21 Gy/3 fractions) and radiation to the right iliac bone (20 Gy/5 fractions). Systemic therapy was changed to the combination of dual checkpoint inhibition with the PD-1 inhibitor nivolumab plus the CTLA-4 inhibitor ipilimumab every 3 weeks, for 4 cycles.
Approximately 3 weeks after completing his fourth cycle of nivolumab plus ipilimumab, the patient complained of severe fatigue. Laboratory tests showed that his hemoglobin level was 5.3 g/dL and the reticulocyte count was 0.005 M/µL, with a reticulocyte percentage of 0.22%. His hemoglobin level was normal during his last infusion of nivolumab and ipilimumab.
A direct antiglobulin (Coombs) test showed a weak positive immunoglobulin (Ig)G autoantibody, but a normal bilirubin count, a normal haptoglobin count, and his lactate dehydrogenase (LDH) was mildly elevated, at 280 IU/L. It was therefore deemed that he was not in active hemolysis, and corticosteroid treatment was initiated.
Despite blood transfusions, the patient showed no improvement of symptoms, and 3 days later his blood glucose level was >700 mg/dL and his hemoglobin A1c level was 8.2%. We suspected that he had autoimmune nonhemolytic anemia and autoimmune diabetes as a result of his treatment with immune checkpoint inhibitors, because he had no known cardiovascular disease and was not obese. This prompted a bone marrow biopsy, which revealed a normal flow cytometry, and the cytogenetics were normal. His cellularity was high, but no erythroid precursors were detected on the marrow. His parvovirus B19 titers were negative.
Given the pertinent laboratory findings in this patient (Table), we suspected the diagnosis of PRCA. Although the direct Coombs test results showed a weak IgG antibody, given the normal bilirubin, low reticulocyte count, normal haptoglobin, and mildly elevated LDH, we were able to rule out autoimmune hemolytic anemia.
No specific immunophenotypic abnormalities and no blasts were detected, as well as no evidence of a T-cell or B-cell lymphoproliferative disorder. As shown in Figure 1, the patient had hypercellular marrow with cellularity of 80% and red-cell aplasia, because no erythroid precursors could be seen. Relative megakaryocyte and myeloid hyperplasia were detected, but no abnormal plasma cells were seen. The patient had mild-to-moderate reticulin fibrosis and increased iron stain, but flow cytometry showed no evidence of any blasts or T-cell or B-cell disorders. Cytogenetics showed a normal male karyotype and fluorescence in situ hybridization (FISH) showed no abnormalities.
The bone marrow biopsy confirmed red-cell aplasia. The differential diagnosis also included myelodysplastic syndromes (MDS), given his anemia and low reticulocyte count; however, the bone marrow biopsy showed no evidence of dysplasia. The FISH and cytogenetics showed no abnormalities for MDS, which was therefore ruled out. Parvovirus-induced red-cell aplasia was a consideration for this patient, because of his fatigue and myalgia. Although the bone marrow biopsy showed red-cell aplasia, the patient’s parvovirus titers were negative, which ruled out parvovirus B19–induced red-cell aplasia; therefore, we suspected that his PRCA was caused by the checkpoint inhibition therapy.
The patient was initially started with methylprednisolone 60 mg intravenous (IV) every 8 hours, as well as packed red blood cell (RBC) transfusions, as needed. He started treatment with metformin, 500 mg twice daily, for diabetes, because it was initially unclear that his condition was autoimmune-mediated. The patient was prescribed prednisone 1 mg/kg (weight, 95 kg) once his hemoglobin was close to 8 g/dL, with a planned slow taper. Despite steroid therapy, his reticulocyte count did not improve. He still required 1 packed RBC transfusion every 10 to 14 days. Because of the patient’s poor glucose control, he was switched to long-acting insulin 20 units at night, and short-acting insulin, 5 to 12 units, after every meal.
Despite these interventions, his blood glucose remained consistently >300 mg/dL, and his hemoglobin A1c increased to 9.9%. The prednisone dose was reduced to 20 mg daily to maintain a hemoglobin level between 7 g/dL and 8 g/dL. A PET scan in January 2019 showed mild persistent activity in the right iliac bone, but no new disease was seen, and the brain MRI showed a relatively stable right frontal lobe mass.
Because his hemoglobin and blood glucose levels were stable, the patient was rechallenged with nivolumab, and prednisone 10 mg daily was continued. However, after 2 weeks, his hemoglobin level decreased to 6.5 g/dL, and his blood glucose increased again to >350 mg/dL. As a result, his prednisone dose was increased again, to 1 mg/kg daily, and his insulin dose was increased to 40 units at night, and 10 to 20 units with meals, and treatment with nivolumab was discontinued.
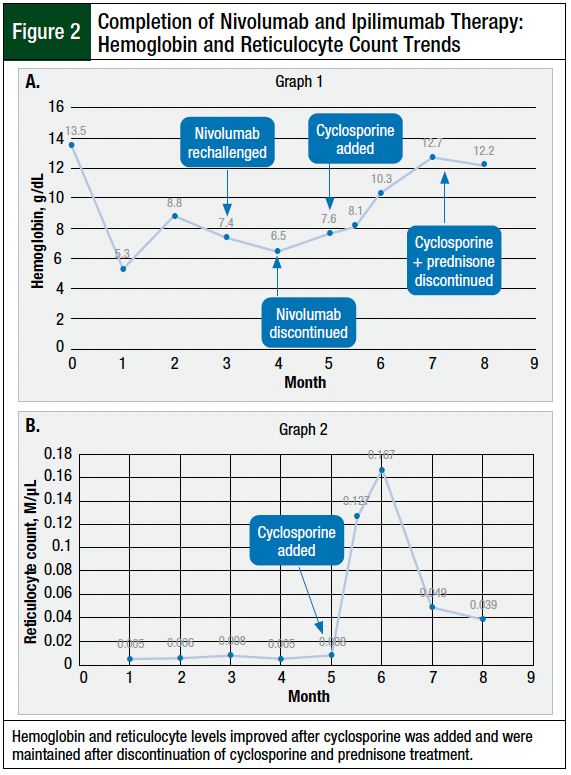
Despite a daily prednisone dose of 40 mg to 60 mg, the patient’s hemoglobin remained between 6.5 g/dL and 7.5 g/dL (Figure 2A) and his reticulocyte count did not increase to more than 0.008 M/µL (0.29%; Figure 2B). He remained transfusion-dependent (1 unit of blood per week) for approximately 6 weeks, despite prednisone therapy; therefore, he was deemed steroid-refractory.
The patient was then started with cyclosporine 2 mg/kg twice daily (400-mg daily dose) along with prednisone 20 mg daily. We were aware of the category C drug–drug interaction between cyclosporine and amlodipine, which potentially increases the serum concentrations of cyclosporine; however, we did not think there was a need to reduce the dose initially. After 3 days, the patient started to have palpitations, and at that point we reduced the cyclosporine dose to 1 mg/kg twice daily (200-mg daily dose) and changed amlodipine to metoprolol.
Two weeks after starting cyclosporine therapy, the patient’s reticulocyte count increased to 0.127 M/µL (4.44%) and his hemoglobin level rose to 8.5 g/dL. After 1 month, his hemoglobin increased to >10 g/dL for the first time since his PRCA diagnosis, and the reticulocyte count increased to 0.167 M/µL (4.63%). The cyclosporine and prednisone were tapered over 3 to 4 weeks, maintaining a hemoglobin level >10 g/dL and a normal reticulocyte count. The patient did not require any blood transfusions while receiving cyclosporine.
An unexpected result of the use of cyclosporine was that the patient’s blood glucose was never above 150 mg/dL, and we were able to taper down his insulin requirement to 10 U daily. His hemoglobin A1c decreased from 9.9% to 6.8% after cyclosporine treatment. A PET scan performed after he completed cyclosporine therapy showed the same mild, persistent activity in the right iliac spine, and no recurrence of melanoma.
The patient is currently not receiving any immunosuppressant therapies or insulin. We decided that immunotherapy would be reinitiated only when he has recurrence of the melanoma. Four months after the patient discontinued treatment with nivolumab, his PET scan showed no active cancer.
Discussion
PRCA is a rare adverse effect of immune checkpoint inhibitor therapy and, to our knowledge, is only reported in a few case reports.8,9 In general, primary PRCA is a known autoimmune disorder. An increase in gamma-delta T-cells that inhibit the burst-forming and colony-forming unit-erythroid precursors is believed to be one of the mechanisms of PRCA.10 Immune checkpoint inhibitors can increase CD3+/CD8+ T-cells, and these activated T-cells may result in PRCA.11 However, the mechanism of PRCA during immune checkpoint inhibitor therapy has not yet been proved.
PRCA should be suspected if a patient suddenly has anemia associated with a low reticulocyte count. Because autoimmune hemolytic anemia is a more common cause of anemia,5 checking for hemolysis (ie, bilirubin levels, LDH levels, direct antiglobulin test [Coombs], and haptoglobin and reticulocyte counts) is necessary.
In our patient, although the Coombs test was positive for IgG autoantibody, there was no indication of active hemolysis, and the bone marrow biopsy confirmed erythroid hypoplasia. However, cases have been reported of a combination of autoimmune hemolytic anemia and PRCA.8,12 Furthermore, in one case, the patient had autoimmune hemolytic anemia and then had PRCA a week later, because that patient had a poor reticulocyte response.9 It is, therefore, important to perform a bone marrow biopsy, which can reveal a PRCA, and can sometimes assess the T-cell response or proliferation that can occur with immune checkpoint inhibitors.
The management of hematologic irAEs has not been well-established, because of the paucity of cases. The National Comprehensive Cancer Network guidelines discuss the more common adverse events, such as dermatitis, colitis, hepatitis, and pneumonitis.13 Corticosteroids (prednisone or methylprednisolone) are the first-line treatment for immune-mediated side effects.13 For steroid-refractory cases, infliximab improves colitis, and data show that mycophenolate mofetil can be used for the treatment of hepatitis and pneumonitis.14
The first-line treatment of choice for immune checkpoint inhibitor–mediated PRCA has been corticosteroids.15 However, the response rate to corticosteroids in PRCA, regardless of etiology, is approximately 40%.15 In a patient with anti–CTLA-4–mediated PRCA who was refractory to a corticosteroid, IV immunoglobulin has been used as a treatment.16
In general, for patients with acquired PRCA that is steroid-refractory, medications such as cyclosporine, rituximab, cyclophosphamide, and azathioprine have been used as treatment.17 Because the response rate to cyclosporine in acquired PRCA is approximately 70%,17 we decided to use cyclosporine as our second-line therapy. Our patient was able to achieve a response to cyclosporine within 1 month.
Similarly, diabetes mellitus caused by immune checkpoint inhibitor therapy is a rare adverse effect, and the mechanism is believed to be upregulation of cytotoxic T-cells causing damage to the pancreatic islet cells.18 Although anti–glutamic acid decarboxylase (GAD) 65 antibodies resulting from T-cell activation is another potential mechanism for immunotherapy-mediated diabetes, cases of immune checkpoint inhibitor–mediated type 1 diabetes mellitus have not always shown increased levels of anti-GAD antibodies.19
It is important to distinguish between immune checkpoint inhibitor–mediated hyperglycemia and actual diabetes mellitus as the clinical manifestation, because the disease management may differ. Most cases of immune checkpoint inhibitor–mediated diabetes mellitus are managed with insulin therapy, with or without metformin.18
Historically, steroids have not effectively managed immune checkpoint inhibitor–mediated diabetes and are counterintuitive to the effort to control blood glucose.20 Our patient had no response to prednisone, and, despite receiving a high dose of insulin, he still had significant hyperglycemia. However, after the initiation of cyclosporine therapy, his insulin requirement and blood glucose improved.
Conclusion
Our case report demonstrates the use of cyclosporine as a potential therapeutic option for steroid-refractory PRCA. Based on this case, cyclosporine may be used in patients who have brittle diabetes that results from immune checkpoint inhibitor treatment. Although PRCA may be a rare hematologic irAE, it should be suspected in patients with anemia, reticulocytopenia, and no evidence of active hemolysis. A bone marrow biopsy should be performed in all patients with suspected hematologic irAEs. Steroids are still considered the first-line therapy for checkpoint inhibitor–mediated PRCA, but cyclosporine may be used in steroid-refractory cases, as shown in our case.
Checkpoint inhibitor–mediated diabetes can be managed with insulin. As the indications for immunotherapy increase, new hematologic irAEs may occur, and further studies may be warranted to determine the most appropriate management of steroid-refractory hematologic irAEs. As shown in our case, cyclosporine can also improve checkpoint inhibitor–mediated diabetes, which should also be investigated in future studies.
Author Disclosure Statement
Dr Padhi, Dr White, and Dr Cetin have no conflicts of interest to report.
References
- Vaddepally RK, Kharel P, Pandey R, et al. Review of indications of FDA-approved immune checkpoint inhibitors per NCCN guidelines with the level of evidence. Cancers (Basel). 2020;12:738.
- Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373:23-34. Erratum in: N Engl J Med. 2018;379:2185.
- Hodi FS, Chesney J, Pavlick AC, et al. Combined nivolumab and ipilimumab versus ipilimumab alone in patients with advanced melanoma: 2-year overall survival outcomes in a multicentre, randomised, controlled, phase 2 trial. Lancet Oncol. 2016;17:1558-1568.
- Hellmann MD, Paz-Ares L, Caro RB, et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. 2019;381:2020-2031.
- Delanoy N, Michot JM, Comont T, et al. Haematological immune-related adverse events induced by anti-PD-1 or anti-PD-L1 immunotherapy: a descriptive observational study. Lancet Haematol. 2019;6:e48-e57.
- Davis EJ, Salem JE, Young A, et al. Hematologic complications of immune checkpoint inhibitors. Oncologist. 2019;24:584-588.
- Ferrari SM, Fallahi P, Elia G, et al. Autoimmune endocrine dysfunctions associated with cancer immunotherapies. Int J Mol Sci. 2019;20:2560.
- Nair R, Gheith S, Nair SG. Immunotherapy-associated hemolytic anemia with pure red-cell aplasia. N Engl J Med. 2016;374:1096-1097.
- Olson DJ, Rajagopal P, Tjota MY, et al. A case of dual-mechanism immune-related anaemia in a patient with metastatic melanoma treated with nivolumab and ipilimumab. J Immunother Cancer. 2020;8:e000380.
- Liu M, Liu T, Meng WT, et al. Role of gammadelta T cells in pathogenesis of acquired pure red cell aplastic anemia. Zhongguo Shi Yan Xue YE Xue Za Zhi. 2007;15:142-146.
- Aye LL, Harris JB, Siddiqi I, Hagiya A. Bone marrow findings of immune-mediated pure red cell aplasia following anti-programmed cell death receptor-1 therapy: a report of two cases and review of literature. J Hematol. 2019;8:71-78.
- Hall M, Meti N, Liontos L, et al. Refractory autoimmune hemolytic anemia and pure red cell aplasia secondary to immunotherapy requiring prolonged immunosuppression. JCO Oncol Pract. 2020;16:699-700.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Management of Immunotherapy-Related Toxicities. Version 3.2021. May 14, 2021. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. Accessed September 13, 2021.
- Friedman CF, Proverbs-Singh TA, Postow MA. Treatment of the immune-related adverse effects of immune checkpoint inhibitors: a review. JAMA Oncol. 2016;2:1346-1353.
- Sawada K, Fujishima N, Hirokawa M. Acquired pure red cell aplasia: updated review of treatment. Br J Haematol. 2008;142:505-514.
- Gordon IO, Wade T, Chin K, et al. Immune-mediated red cell aplasia after anti-CTLA-4 immunotherapy for metastatic melanoma. Cancer Immunol Immunother. 2009;58:1351-1353.
- Means RT Jr. Pure red cell aplasia. Blood. 2016;128:2504-2509.
- Alrifai T, Ali FS, Saleem S, et al. Immune checkpoint inhibitor induced diabetes mellitus treated with insulin and metformin: evolution of diabetes management in the era of immunotherapy. Case Rep Oncol Med. 2019;2019:8781347. doi: 10.1155/2019/8781347.
- Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy—immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13:195-207.
- Zaied AA, Akturk HK, Joseph RW, Lee AS. New-onset insulin-dependent diabetes due to nivolumab. Endocrinol Diabetes Metab Case Rep. 2018;2018:17-0174. doi:10.1530/EDM-17-0174.