High-dose methotrexate is defined as a methotrexate dose of >500 mg/m2 and is used in the treatment of central nervous system (CNS) lymphomas and osteosarcomas, high-risk leukemias, and other types of lymphoma.1,2 Methotrexate is cleared primarily by the kidneys via glomerular filtration and tubular secretion.2-4 The safe administration of high-dose methotrexate requires aggressive hydration to maintain urine output, urinary alkalinization to prevent methotrexate crystallization and associated renal damage, close monitoring of methotrexate serum levels, and leucovorin rescue to avoid possible adverse events related to prolonged methotrexate exposure, including renal injury, gastrointestinal tract sloughing, and myelosuppression.1,5
The renal excretion of methotrexate results in high urinary concentrations that are insoluble at physiologic urine pH, which can lead to methotrexate crystallization in the nephrons and cause acute kidney injury.1 An acute kidney injury can result in decreased methotrexate excretion leading to sustained and elevated methotrexate plasma levels resulting in sequelae such as renal failure, which may require dialysis.1,3 To prevent the renal crystallization of methotrexate, urinary alkalinization is used to maintain a goal urine pH of ≥7.1,2
Coadministration of medications that inhibit or compete with methotrexate for elimination should be avoided, because it can cause renal injury by delaying the elimination of methotrexate.3,6 Medications that may interfere with normal methotrexate clearance by causing additional kidney injury, or by competing for renal excretion, include aminoglycosides, beta-lactam antibiotics, fluoroquinolones, vancomycin, nonsteroidal anti-inflammatory agents (NSAIDs), proton pump inhibitors, radiographic contrast dyes, and trimethoprim plus sulfamethoxazole.1,3,6-8
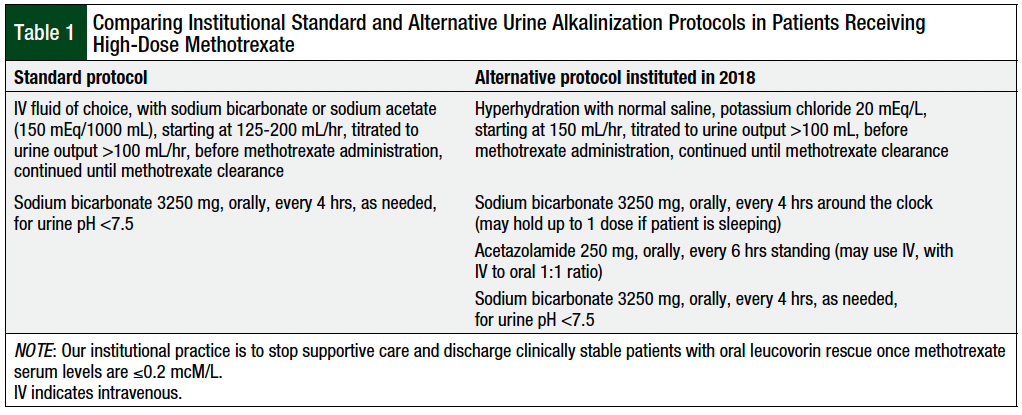
The standard-of-care urinary alkalinization strategy incorporates the use of intravenous (IV) sodium bicarbonate–containing fluids before, during, and after methotrexate administration until the serum methotrexate levels reach an established threshold per institutional laboratory assays.1 Recent drug shortages in the past 5 years that have affected the supply of IV sodium bicarbonate have prompted the development of alternative urinary alkalinization strategies for patients who are receiving high-dose methotrexate, as detailed in previous literature.9-13
Our study was conducted during the IV sodium bicarbonate shortage beginning in the fall of 2018, during which our institutional urinary alkalinization protocol for patients receiving high-dose methotrexate was adapted to accommodate for that drug shortage.
We evaluated the effect of an alternative urine alkalinization protocol that was not based on IV sodium bicarbonate in patients with cancer who were receiving high-dose methotrexate at our institution, with the goal of confirming that the time to methotrexate clearance was similar between our alternative protocol and the standard urine alkalinization protocol.
Our institution’s alternative urinary alkalinization protocol included hyperhydration with normal saline containing potassium chloride, standing oral or IV acetazolamide, standing oral sodium bicarbonate, and supplementary oral sodium bicarbonate, as needed for urine pH of <7.5 (Table 1). This regimen was modified from an oral sodium bicarbonate–based urinary alkalinization protocol that our institution used during a previous drug shortage in 201711 to utilize around-the-clock acetazolamide and oral sodium bicarbonate, in the hopes of decreasing oral pill burden, increasing adherence, and thereby improving the chance of achieving the goal urine pH of ≥7.
We did not use acetazolamide monotherapy because of the potential for serum bicarbonate depletion within 48 hours if additional bicarbonate supplementation is not given concomitantly.14 And because IV sodium acetate was also intermittently on shortage during that IV sodium bicarbonate shortage, it was not possible to use it routinely as an alternative treatment. Patients who could not tolerate oral sodium bicarbonate were switched to standard IV sodium bicarbonate or to IV sodium acetate protocol, depending on drug availability.
Methods
In this single-center retrospective cohort study, we compared the safety and efficacy of an alternative urinary alkalinization strategy with the standard urinary alkalinization with IV sodium bicarbonate in adults with cancer who were receiving high-dose methotrexate at a large academic medical center. This study was approved by, and was conducted in compliance with, Institutional Review Board requirements.
The patients were identified using a drug administration report that was generated by the electronic medical record (EMR). Patients aged 18 years or older who received high-dose methotrexate were divided into 2 groups: those who received standard IV sodium bicarbonate as their urine alkalinization protocol (ie, the control group) and those who received the alternative alkalinization protocol (ie, the intervention group). The control group received high-dose methotrexate doses between October 18, 2017, and October 17, 2018, whereas the intervention group received high-dose methotrexate doses between October 26, 2018, and September 25, 2019. Patients who received methotrexate doses ≤3500 mg/m2 and those who received outpatient high-dose methotrexate were excluded from the study.
The data we collected from the EMR included the dose of methotrexate; date and time of the initiation and completion of methotrexate administration; the time to methotrexate clearance (defined as a drug level <0.1 μM, discharge, or the start of the next chemotherapy cycle, whichever occurred earliest); urinary alkalinization strategy; concomitant interacting medications (defined as any of the following medications administered within 24 hours before starting methotrexate or during methotrexate clearance as defined above: aminoglycosides, penicillins and cephalosporins, vancomycin, NSAIDs, proton pump inhibitors, radiographic contrast dyes, amphotericin, probenecid, salicylates, gemfibrozil, and/or trimethoprim plus sulfamethoxazole1,3,6,15); and in the alternative protocol group, the number of administered doses of acetazolamide and bicarbonate, to calculate adherence to the protocol.
The duration of urine output of <100 mL/hr, occurrence of urine pH <7, and the duration of urine pH <7 were also collected. Urine pH was collected with each void, and the duration of urine pH >7 was calculated as the time between the first urine pH measurement of <7 and the next urine pH of ≥7. The presence of third-spacing conditions (ie, accumulation of fluids within extravascular space, such as pleural effusions, ascites, or fluid collections) and mucositis were collected, as well as the baseline, peak, and nadir of the following laboratory values: alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, total bilirubin, serum creatinine (SCr), serum carbon dioxide (CO2), absolute neutrophil count (ANC), platelet count, and hemoglobin. Baseline laboratory values were documented on the day of methotrexate administration. Nadir and peak values were recorded if the value occurred between the day after chemotherapy and the start of the next cycle. Creatinine clearance (CrCl) was calculated using the Cockcroft-Gault equation.
During the study period, the Siemens Syva Emit Methotrexate Assay was used to test the serum methotrexate levels. Urine pH was tested by urinalysis with each urinary void. The 650-mg sodium bicarbonate oral tablets were assumed to yield 7.7 mEq of bicarbonate, with a calculated oral bioavailability of 1:1.16 The study data were collected and managed using REDCap electronic data capture tools.17
The primary outcome of the study was the time to methotrexate clearance in hours, which was calculated from the start of methotrexate infusion to methotrexate clearance, as was defined above. The time to methotrexate clearance was chosen as a primary end point because increased exposure to methotrexate and lower methotrexate clearance have been associated with increased methotrexate toxicities.15,18 The secondary outcomes of the study included the occurrence of urine pH <7 at any recorded point, the discrete number of times urine pH <7, the total hours of urine pH <7, the rates of adverse events, and protocol compliance for patients who received the alternative protocol.
The safety end points included hepatoxicity, neutropenia, thrombocytopenia, mucositis, SCr increase, and acute kidney injury. Hepatotoxicity was defined as an increase in ALT, AST, or total bilirubin. Acute kidney injury was defined based on the Kidney Disease: Improving Global Outcomes clinical practice guidelines as either an increase in SCr by ≥0.3 mg/dL within 48 hours, an increase in SCr to ≥1.5 times above baseline within the past 7 days, or urine volume ≤0.5 mL/kg per hour, for 6 hours.19 The adverse events were graded using the Common Terminology Criteria for Adverse Events version 5.0.20
Statistical Analysis
Bivariate analyses were performed on continuous and categorical variables to compare urine alkalinization. The time to methotrexate clearance was analyzed using an analysis of covariance (ANCOVA) using baseline CrCl,3,4,15 methotrexate dose,3,4,21 hours with urine pH <7,3,15 and hours with urine output <100 mL/hr (per our institution standard goal for threshold urine output). The value of P <.05 was considered significant. Alternative alkalinization protocol compliance data were analyzed using descriptive statistics. Compliance to the protocol was calculated by dividing the total number of administered medication doses by the total number of doses ordered.
IBM SPSS Statistics version 25.0 (IBM; Armonk, NY) was used to conduct the statistical analyses.
Results
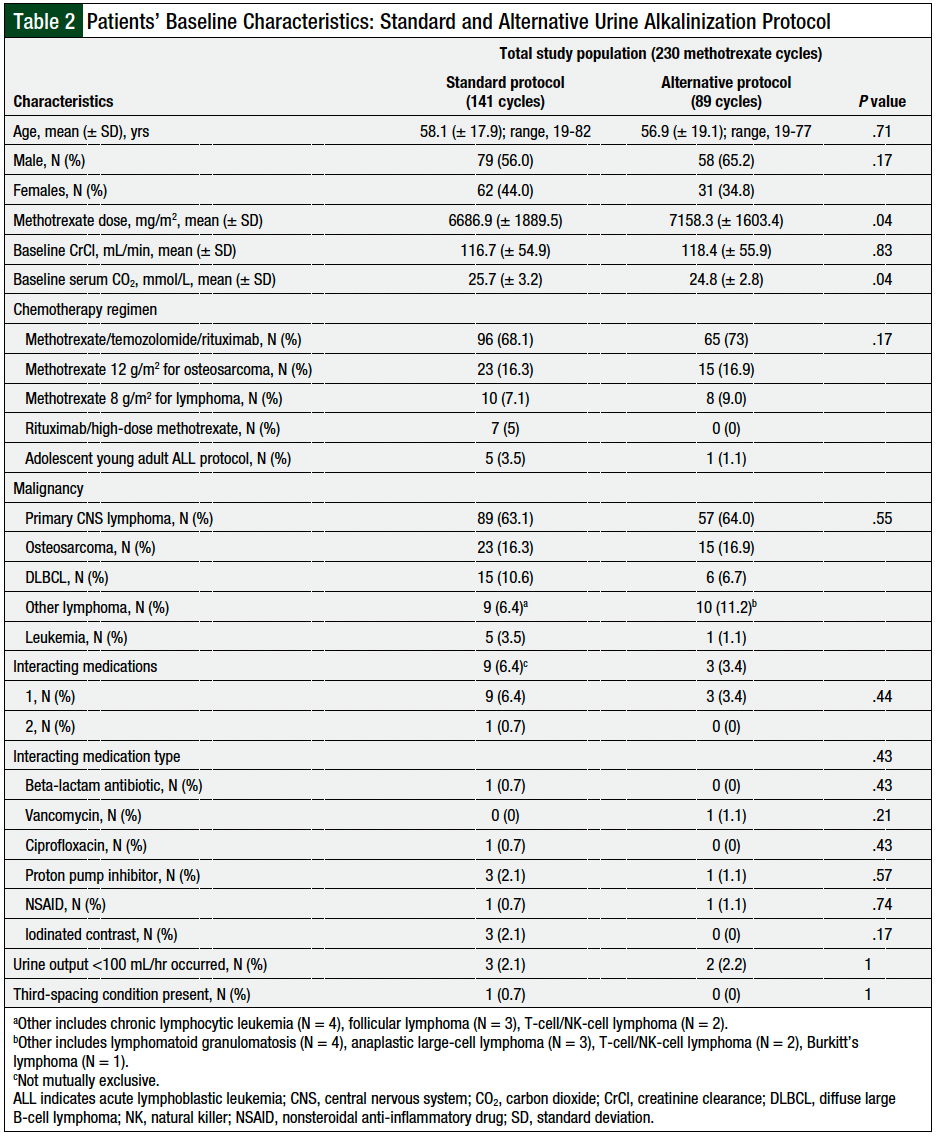
This study included 57 patients who received 230 total cycles of high-dose methotrexate, including 141 cycles using the standard protocol and 89 cycles using the alternative protocol. The most common cancer was primary CNS lymphoma, followed by osteosarcomas. The patient characteristics were similar between the 2 groups, including age, sex, baseline CrCl, and baseline CO2, except the methotrexate dose that was administered was significantly higher in the alternative protocol group, by 7% (7158.3 mg/m2 [± 1603.4 mg/m2] vs 6686.9 mg/m2 [± 1889.5 mg/m2]; P = .04). The factors that affected methotrexate clearance, including interacting medications, urine output <100 mL/hr, and the presence of third-spacing conditions, were also similar between the 2 groups (Table 2).
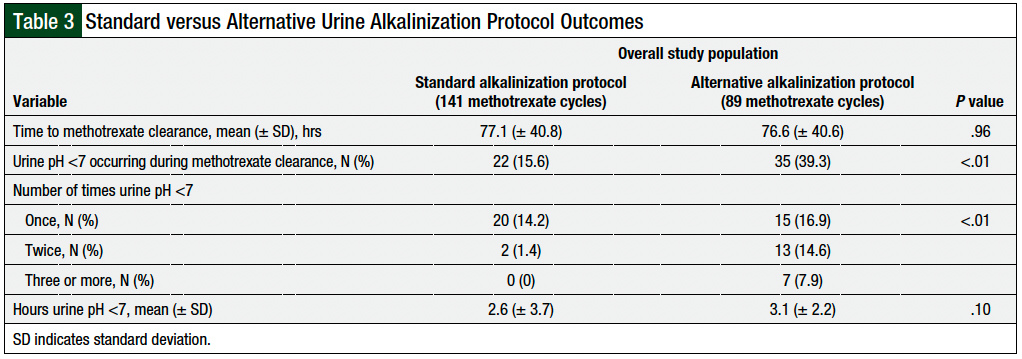
In the analysis of all 230 high-dose methotrexate cycles, the mean time to methotrexate clearance for the standard protocol was 77.1 hours (± 40.8 hours) versus 76.6 hours (± 40.6 hours) for the alternative protocol (P = .96; Table 3). Urine pH decreased to <7 in 15.6% and 39.3% of cycles in the standard and alternative protocol groups, respectively (P <.01), and the number of times that urine pH decreased to <7 was higher in the alternative protocol group (P <.01; Table 3). The time with a urine pH of <7 was 2.6 hours (± 3.7 hours) and 3.1 hours (± 2.2 hours) between the 2 groups, respectively (P = .1; Table 3).
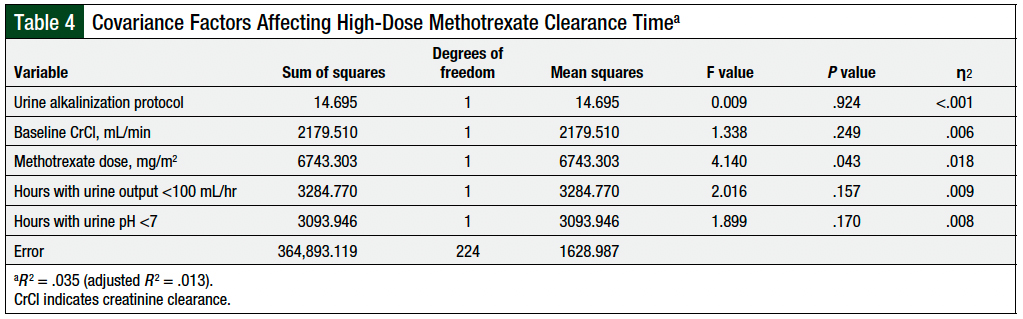
The only variable that affected the time to methotrexate clearance in the ANCOVA analysis was the methotrexate dose (P = .043); the urine alkalinization protocol did not affect the time to high-dose methotrexate clearance (P = .924; Table 4).
In the alternative protocol group, in 23 (25.8%) of the 89 cycles, the patients did not tolerate the alternative protocol and were switched to the standard protocol of IV sodium acetate (N = 15; 65.2%) or IV sodium bicarbonate (N = 8; 34.8%). In patients who received the alternative protocol, the mean acetazolamide compliance rate was 97.2%, and the mean compliance rate with around-the-clock oral sodium bicarbonate was 90.16%. The median number of oral sodium bicarbonate as-needed doses received was 2 (interquartile range, 1-4).
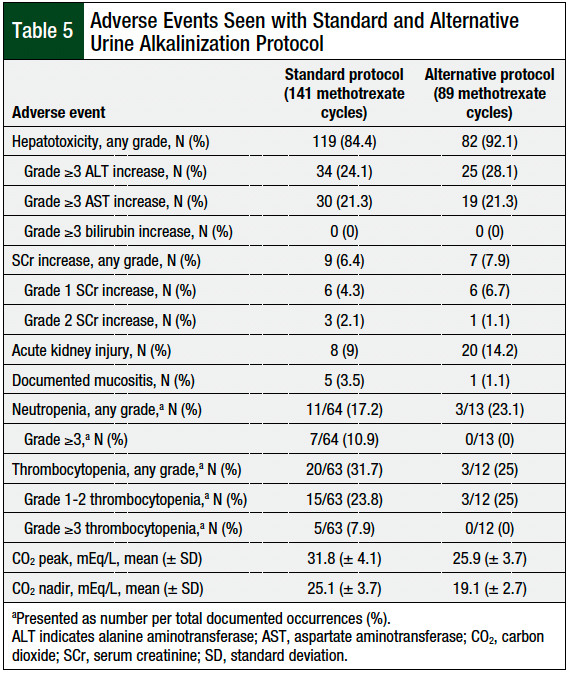
The adverse events were similar between both groups (Table 5), except for the CO2 nadir and peak; the CO2 nadir was lower in the alternative protocol group (19.1 [± 2.7] mmol/L vs 25.1 [± 3.7] mmol/L), and the CO2 peak was higher in the standard protocol group (31.8 [± 4.1] mmol/L vs 25.9 [± 3.7] mmol/L).
Acute kidney injury occurred in 9% of the methotrexate cycles in the standard protocol group and in 14.2% of the cycles in the alternative protocol group, and any-grade SCr increase occurred in 6.4% and 7.9% of the standard protocol and alternative protocol groups, respectively; no grade ≥3 increased SCr levels occurred.
Hepatotoxicity of any grade occurred in 84.4% of patients in the standard protocol group and in 92.1% of patients in the alternative protocol group, with a grade ≥3 increased ALT in 24.1% versus 28.1% of the patients, respectively, and a grade ≥3 increased AST in 21.3% of patients in each group.
Of the 64 cycles with documented between-cycles complete blood counts in the standard protocol group, 17.2% had any grade neutropenia, and 10.9% had grade ≥3 neutropenia. Of the 13 cycles in the alternative group with documented nadirs, 23.1% had grade 1 or 2 neutropenia, and none of the patients had grade ≥3 neutropenia. Thrombocytopenia of any grade occurred in 31.7% of the 63 patients in the alternative protocol group with documented nadirs, as well as 7.9% grade ≥3 events. Of the 12 patients in the alternative protocol group with documented nadirs, 25% had grade 1 or 2 platelet decreases, and none had grade ≥3 events (Table 5).
Discussion
The baseline characteristics in both groups were well-matched, except for patients in the alternative protocol group who received a mean methotrexate dose that was 7% higher than the standard protocol group. Although higher methotrexate doses are associated with longer methotrexate clearance times,3,4,21 and our covariate analysis demonstrated that methotrexate dose significantly affected the methotrexate clearance time in our patients, the mean time to methotrexate clearance was similar between the standard and alternative urine alkalinization protocol groups. The type of urine alkalinization protocol did not affect significantly the time to methotrexate clearance. These results suggest that our alternative urine alkalinization protocol is feasible and can be used during IV sodium bicarbonate shortages.
The incidence of urine pH <7 was more common with the alternative protocol. The higher incidence and duration of urine pH <7 can be explained by the alternative protocol allowing a single oral bicarbonate dose to be held overnight to allow for uninterrupted sleep, which resulted in an increased risk for the urine pH level to decrease overnight below the goal.
Although the incidence of, number of, and hours with urine pH <7 were higher in the alternative protocol group, the urine pH did not have a significant effect on methotrexate clearance time in the covariate analysis, and the time to methotrexate clearance and the rates of adverse events were not affected in the 2 groups. And although urine pH <7 results in impaired methotrexate clearance and subsequent toxicity,3,22 the clinical significance of, or the threshold for, the hours or the number of times that urine pH <7 affected methotrexate clearance time and/or adverse events is unknown, and no published studies have evaluated these end points.
The incidence of adverse events in the 2 groups was similar, except that the serum CO2 levels differed between the groups. In the alternative protocol group, which received acetazolamide, the CO2 nadir was lower than in the standard protocol group, which is expected, given that the mechanism of action of acetazolamide is to increase renal tubule bicarbonate excretion, which can cause hypocarbia.12,23
Conversely, in the standard-of-care protocol group that received IV sodium bicarbonate with supplemental oral sodium bicarbonate, a higher CO2 peak was observed, which is also associated with the administration of large amounts of bicarbonate. The incidence of liver function test elevations was high and was similar across both groups; acute transient elevations of aminotransferases to 10 times the upper limit of normal or more are common after high-dose methotrexate and are usually self-resolving,24 which was the case in this study.
Although the alternative protocol had a 26% discontinuation rate, patients who tolerated the alternative protocol had adherence rates of 97% and 90% for acetazolamide and oral sodium bicarbonate, respectively. This represents an improvement from the almost 68% adherence rate from our previous urine alkalinization protocol, in which patients received scheduled oral sodium bicarbonate administered every 2 hours.11 We believe that our protocol’s improved adherence is a result of changing the frequency of scheduled medications from every 2 hours to every 4 hours and allowing the omission of a single dose while patients were sleeping, if their urine pH was ≥7.5.
Although other studies have evaluated various alternative urine alkalinization strategies in patients with cancer who received high-dose methotrexate,9-13 our study adds valuable data to support the feasibility of these alternative urine alkalinization protocols. Our study is the largest data set published to date, with a total of 230 methotrexate cycles evaluated, and the higher mean methotrexate dose (approximately 6860 mg/m2) shows that it is applicable even to patients who receive greater high-dose methotrexate doses.
Limitations
The limitations of this study include its retrospective nature, the lack of documentation for subjective adverse events, and that nadir complete blood counts were not routinely drawn as the standard of care.
Subjective adverse events relying on documentation in the EMR, such as mucositis, could be underreported if providers did not document them. Hematologic nadirs were difficult to assess, because complete blood counts were not routinely drawn at approximate nadir periods as part of the standard of care. Therefore, the incidence and severity of hematologic adverse events were likely underestimated.
Because real-time continuous urine pH monitoring was not available, the surrogate data points of urine pH level with each void were used, which may underestimate and/or overestimate the time of urine pH <7. However, a lack of documentation and the use of surrogate laboratory values would not be expected to differ between the 2 groups, and thus were not expected to bias our between-group analyses.
Adherence is still a concern with our alternative alkalinization protocol, as is demonstrated by the 26% of patients who were switched to IV alkalinization because of an inability to tolerate the pill burden associated with the number of oral sodium bicarbonate tablets.
Because other urine alkalinization studies have successfully used lower oral sodium bicarbonate doses,9,10,12,13 our protocol could be modified in the future to incorporate lower doses of oral sodium bicarbonate, especially considering that oral sodium bicarbonate and acetazolamide can effectively contribute to urine alkalinization.9,10,12-14,25
Conclusion
As a result of drug shortages, treatment recommendations and mitigation strategies that use alternative drugs and protocols are required to ensure the effective and safe delivery of care. During the IV sodium bicarbonate shortage that our institution had starting in fall 2018, an alternative urine alkalinization protocol that used oral or IV acetazolamide and oral sodium bicarbonate was implemented for patients who were receiving high-dose methotrexate. No differences were found in the time to methotrexate clearance or the rates of adverse events with our alternative urine alkalinization protocol compared with the standard protocol, although more incidences of and longer time of urine pH <7 were reported, which demonstrates that this is a feasible option during times of IV sodium bicarbonate shortage.
Acknowledgments
This work was conducted with support from Harvard Catalyst/The Harvard Clinical and Translational Science Center. The content is solely the responsibility of the authors and does not necessarily represent the official views of Harvard Catalyst, Harvard University and its affiliated academic healthcare centers, or the National Institutes of Health. We would also like to thank Ephraim Hochberg, MD, for his assistance and support.
Author Disclosure Statement
Dr Sinoimeri and Dr Monaco have no conflicts of interest to report. Dr Lei has been a Consultant to Fresenius Kabi USA, Trapelo Health, and Dedham Group. Dr Lou provided one-time consultancy to Fresenius Kabi.
References
- Howard SC, McCormick J, Pui CH, et al. Preventing and managing toxicities of high-dose methotrexate. Oncologist. 2016;21:1471-1482.
- Drost SA, Wentzell JR, Giguère P, et al. Outcomes associated with reducing the urine alkalinization threshold in patients receiving high-dose methotrexate. Pharmacotherapy. 2017;37:684-691.
- Wiczer T, Dotson E, Tuten A, et al. Evaluation of incidence and risk factors for high-dose methotrexate-induced nephrotoxicity. J Oncol Pharm Pract. 2016;22:430-436.
- Yang Y, Wang X, Tian J, Wang Z. Renal function and plasma methotrexate concentrations predict toxicities in adults receiving high-dose methotrexate. Med Sci Monit. 2018;24:7719-7726.
- Ramsey LB, Balis FM, O’Brien MM, et al. Consensus Guideline for Use of Glucarpidase in Patients with High-Dose Methotrexate Induced Acute Kidney Injury and Delayed Methotrexate Clearance. Oncologist. 2018;23:52-61.
- Joerger M, Huitema ADR, van den Bongard HJGD, et al. Determinants of the elimination of methotrexate and 7-hydroxy-methotrexate following high-dose infusional therapy to cancer patients. Br J Clin Pharmacol. 2006;62:71-80.
- Jarfaut A, Santucci R, Levêque D, Herbrecht R. Severe methotrexate toxicity due to a concomitant administration of ciprofloxacin. Med Mal Infect. 2013;43:39-41.
- Urata S, Yoshikawa N, Saito K, et al. Delayed methotrexate elimination in a patient with primary central nervous system lymphoma: a case report. J Clin Pharm Ther. 2021 May 19. Epub ahead of print.
- Kramer E, Filtz M, Pace M. Evaluation of methotrexate clearance with an enteral urine alkalinization protocol for patients receiving high-dose methotrexate. J Oncol Pharm Pract. 2021;27:26-32.
- Reed DR, Pierce EJ, Sen JM, Keng MK. A prospective study on urine alkalization with an oral regimen consisting of sodium bicarbonate and acetazolamide in patients receiving high-dose methotrexate. Cancer Manag Res. 2019;11:8065-8072.
- Roy AM, Lei M, Lou U. Safety and efficacy of a urine alkalinization protocol developed for high-dose methotrexate patients during intravenous bicarbonate shortage. J Oncol Pharm Pract. 2019;25:1860-1866.
- Visage R, Kaiser N, Williams M, Kim A. Oral methods of urinary alkalinization for high-dose methotrexate administration: alternatives to intravenous sodium bicarbonate during a critical drug shortage. J Pediatr Hematol Oncol. 2019;41:371-375.
- Whiteside H, Gandhi A, Ajebo G, Bryan LJ. When baking soda goes on shortage: urine alkalinization with acetazolamide and oral sodium bicarbonate. Ann Pharmacother. 2018;52:297-298.
- Shamash J, Earl H, Souhami R. Acetazolamide for alkalinisation of urine in patients receiving high-dose methotrexate. Cancer Chemother Pharmacol. 1991;28:150-151.
- Relling MV, Fairclough D, Ayers D, et al. Patient characteristics associated with high-risk methotrexate concentrations and toxicity. J Clin Oncol. 1994;12:1667-1672.
- Sodium bicarbonate. Lexi-Drugs. Lexicomp. Wolters Kluwer. https://online.lexi.com. Accessed July 12, 2020. [Requires subscription to access.]
- Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381.
- Kawakatsu S, Nikanjam M, Lin M, et al. Population pharmacokinetic analysis of high-dose methotrexate in pediatric and adult oncology patients. Cancer Chemother Pharmacol. 2019;84:1339-1348.
- Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120:c179-c184.
- Department of Health & Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf. Accessed July 12, 2020.
- Fukuhara K, Ikawa K, Morikawa N, Kumagai K. Population pharmacokinetics of high-dose methotrexate in Japanese adult patients with malignancies: a concurrent analysis of the serum and urine concentration data. J Clin Pharm Ther. 2008;33:677-684.
- Sand TE, Jacobsen S. Effect of urine pH and flow on renal clearance of methotrexate. Eur J Clin Pharmacol. 1981;19:453-456.
- King PD, Perry MC. Hepatotoxicity of chemotherapy. Oncologist. 2001;6:162-176.
- Ku M, Bazargan A, Tam C. Addition of low dose acetazolamide as an adjunct in patients undergoing high dose methotrexate is safe and beneficial. Intern Med J. 2020;50:357-362.