Oral chemotherapies continue to be a favorable treatment modality, with more than 100 agents currently approved by the US Food and Drug Administration (FDA).1-3 The prevalence of prescribing oral therapies is likely to increase, and it is currently estimated that 25% to 30% of all new anticancer agents are administered orally.3 Oral chemotherapy encompasses cytotoxic agents and targeted therapies.4
Although targeted therapy is generally regarded less toxic by drug developers and healthcare providers compared with intravenous (IV) chemotherapy, targeted agents still have a potential for life-threatening adverse events and other organ toxicities.4
Patient adherence to oral chemotherapy has been shown to be an ongoing challenge and can further complicate treatment.5 Previous research has identified many patient-related, treatment-related, and healthcare-related factors that can affect patient adherence.5 Avoidance of side effects, pill burden, mood, and forgetfulness have been identified as a few of the patient-related factors.5 These side effects can also be exacerbated by drug–drug interactions, such as those involving cytochrome P450 enzyme pathways.6 Drug–drug interaction management is one of several means by which pharmacists can improve the safety of chemotherapy use.6
Guidance for oral chemotherapy practices in the inpatient setting is limited. The American Society of Health-System Pharmacists guidelines on preventing medication errors recommend independent double-checks of IV chemotherapy by 2 qualified individuals to reduce errors in the inpatient and the outpatient settings.7 Although this process is widely accepted for IV chemotherapy, it is unclear if these same processes can improve the safety of oral chemotherapy use.
The 2018 Hematology/Oncology Pharmacist Association Best Practices for the Management of Oral Oncolytic Therapy: Pharmacy Practice Standard provides a detailed list of best practices based on a comprehensive review of the literature, but this standard does not specifically address inpatient use of oral chemotherapy.8
In addition, the 2016 Updated American Society of Clinical Oncology/Oncology Nursing Society Chemotherapy Administration Safety Standards includes few specific practice guidelines for oral chemotherapy, and these guidelines focus on education and prescription requirements.9
Whereas IV chemotherapy has well-established guidance for safety practices, available medical literature is limited on continuing oral chemotherapy during acute hospital admission, leaving an opportunity to identify safety concerns and establish an evidence-based approach to inpatient treatment to inform oral chemotherapy protocols.
The primary objective of this study was to determine the rate of incidence of oral chemotherapy treatment deviations among the 10 most often ordered oral chemotherapy classes for patients admitted to the University of Kansas Hospital. The secondary outcome was to describe the nature of treatment deviations and identify risk factors associated with treatment deviations.
Methods
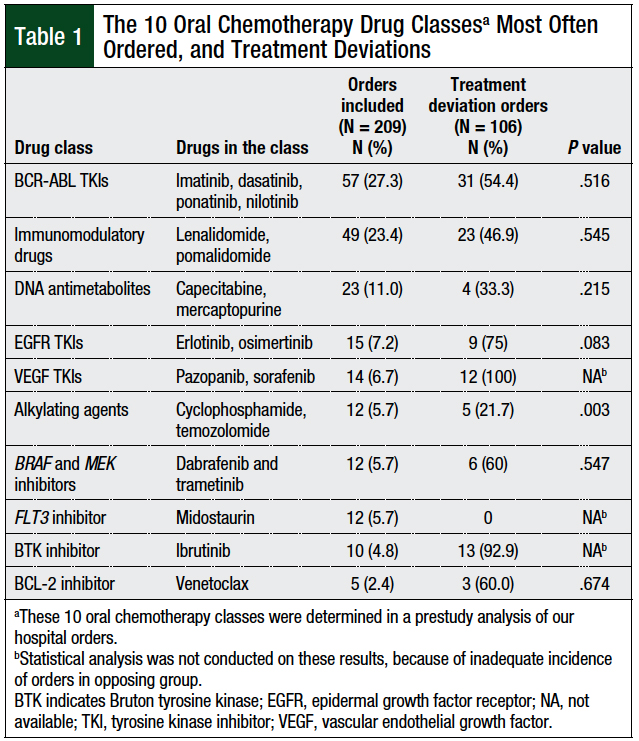
In this single-center, institutional board-approved, retrospective chart review, we evaluated inpatient orders for oral chemotherapy at the University of Kansas Hospital between December 2016 and June 2018. We performed a prestudy evaluation to determine the 10 most often administered oral chemotherapy classes at the University of Kansas Hospital to be included in the study (Table 1).
Separating oral chemotherapies by drug class helped to evaluate adverse events and to identify oral chemotherapy classes that are potentially at increased risk for treatment deviations. Each distinct oral chemotherapy agent was included in the study once per patient hospital encounter. Orders were excluded from the analysis if the patient was younger than age 18 years, or if the therapy was indicated for a nononcologic condition.
We defined treatment deviations as consisting of at least 1 of the 4 following conditions related to inpatient oral chemotherapy treatment continuation in the setting of (1) abnormal treatment parameters, as defined by laboratory values and vital signs outside the recommended ranges described in the drug’s prescribing information; (2) potential oral chemotherapy–related adverse event(s), defined by the FDA’s boxed warnings and the warnings and precautions section in Lexicomp10 as of December 2018; (3) clinically relevant category D or X drug–drug interaction(s) among oral chemotherapy and medications started and stopped at hospital discharge, as defined by the Lexicomp online application; and (4) dosing error(s) compared with the before admission prescription. Laboratory values and vital signs that were not collected within 24 hours of the oral chemotherapy order being placed were considered treatment deviations. Intentional treatment deviations were not considered deviations for the purpose of this study.
Treatment parameters are set to the threshold value at which a dose modification or treatment withholding would be recommended by the drug’s prescribing information. For example, it is recommended in the prescribing information of capecitabine that the dose be reduced to 75% of the usual dose for an estimated creatinine clearance <50 mL/min; therefore, a treatment parameter for capecitabine is creatinine clearance >50 mL/min. Thus, it would be deemed abnormal in this study to continue treatment at full dose in a patient with a creatinine clearance <50 mL/min.11
Because of the many medications administered during inpatient stays, including one-time doses, it was not feasible to evaluate every medication for drug–drug interactions with the oral chemotherapy agent during the patient’s admission. Rather, we prioritized evaluating medication changes at discharge, because these were believed to be more likely to reflect long-term changes in therapy that would include drug–drug interactions of concern.
To evaluate the secondary objective and prioritize institutional quality improvement efforts, we performed bivariate and multivariate regression analyses. A bivariate analysis may detect differences in the incidence of treatment deviations within oral chemotherapy classes and the selected risk factors. Complementing the results of the bivariate analysis, a multivariate regression analysis may detect risk factors for treatment deviations when considering all factors simultaneously.
The process of prescribing and verifying IV chemotherapy at the University of Kansas Hospital is explained in detail in our health system policy. In that policy, “authorized prescribers” who have medical privileges granted for the treatment of malignancy are defined. Furthermore, the process of independent double-check completed by 2 oncology-trained pharmacists is mandated before administration of all IV chemotherapy; this was not required for oral chemotherapy administration at the time of this study. During the study period, any physician, including medical residents, could place orders for inpatient continuation of oral chemotherapy that was on the before admission medication list.
Results
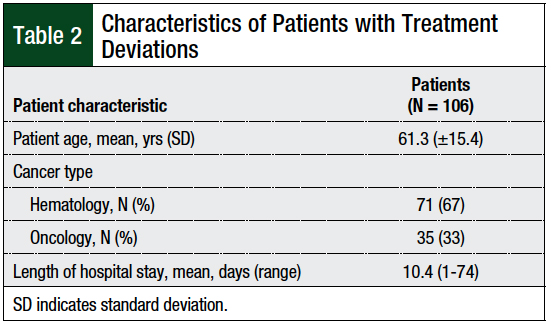
In total, 209 oral chemotherapy orders were identified in chart review, of which 106 (50.7%) orders contained at least 1 treatment deviation, and 43 (20.6%) orders contained 2 or more treatment deviations. We did not perform statistical analysis on oral chemotherapy classes with inadequate incidence of orders in opposing groups. Characteristics of patients with treatment deviations are described in Table 2.
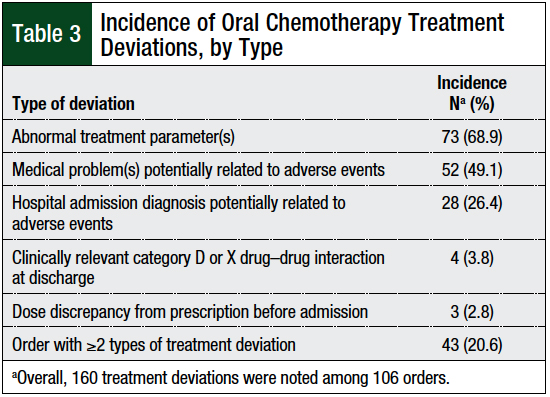
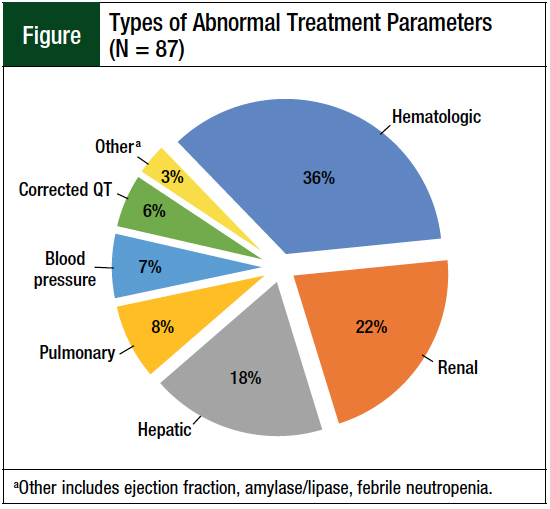
The most common type of treatment deviation was an abnormal treatment parameter, which occurred in 73 (68.9%) of oral chemotherapy orders (Table 3). Among these 73 orders, 87 distinct treatment deviations occurred (Figure). Continuation of oral chemotherapy in the inpatient setting in a patient whose admitting diagnosis was potentially related to oral chemotherapy adverse events occurred in 28 (26.4%) orders. Continuation of oral chemotherapy in the inpatient setting in a patient with an active medical problem that was potentially related to oral chemotherapy adverse events occurred in 52 (49.1%) orders. A drug–drug interaction was present on the discharge medication list of 4 (3.8%) orders. Dose discrepancies occurred in 3 (2.8%) orders.
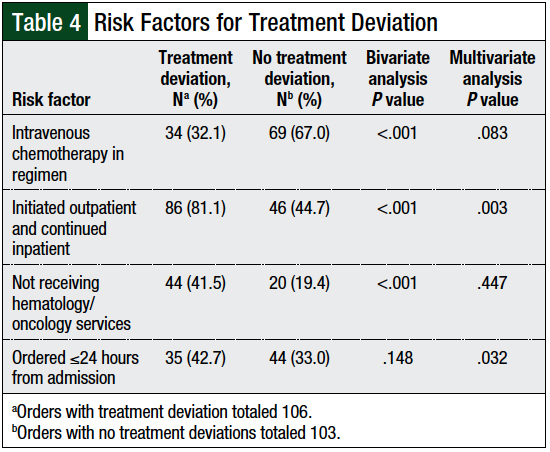
The bivariate analysis showed that fewer treatment deviations occurred when oral chemotherapy was co-administered with IV chemotherapy than when oral chemotherapy was used alone (32.1% vs 67.0%, respectively; P <.001) and when patients received treatment on a hematology/oncology service than when they did not (58.5% vs 80.6%, respectively; P <.001).
Among 79 oral chemotherapy orders that were placed within 24 hours of admission, 35 (44.3%) had treatment deviations. Outpatient-initiated orders showed a higher proportion of treatment deviations compared with inpatient-initiated orders (81.1% vs 44.7%, respectively; P <.001).
In the multivariate risk factor analysis, oral chemotherapy orders that were placed within 24 hours of admission (P = .032) and oral chemotherapy initiated at the outpatient setting and continued in the inpatient setting (P = .003) were 2 factors that predicted increased risk for treatment deviation (Table 4).
Discussion
This review of oral chemotherapy orders shows that BCR-ABL tyrosine kinase inhibitors (TKIs) and immunomodulatory drugs included the largest number of treatment deviations overall, although these were also the 2 most common oral chemotherapy classes represented in the study. We found the highest percentage of treatment deviations within the vascular endothelial growth factor (VEGF) TKIs and the Bruton tyrosine kinase (BTK) inhibitors classes. VEGF TKIs and BTK inhibitors may represent oral chemotherapy classes at increased risk for treatment deviation, but this could be a result of practice preferences at our institution and may not necessarily be a generalizable finding.
Oral chemotherapy was continued in the inpatient setting when the admitting diagnosis was potentially related to oral chemotherapy adverse events for 28 patients. The protocols for oral chemotherapy at the University of Kansas Hospital during the study period were still evolving. During the dates of data collection, any prescriber could order oral chemotherapy agents from the before admission medication list; no hematologist/oncologist was required to be consulted before ordering chemotherapy, which might have contributed to the incidence of oral chemotherapy treatment deviations noted.
The grade of an adverse event was not consistently documented in the electronic medical record (EMR), so treatment continuation in the setting of lower grades of adverse events might have been appropriate, but we did not capture these data in this study. The low rate of drug–drug interactions noted overall may be attributable to the EMR-integrated drug–drug interaction tool at our institution, as well as to our institution’s pharmacist-driven discharge medication review policies. Dose discrepancies were found in only 3 oral chemotherapy orders, and this, too, may be attributed to our admission medication reconciliation process.
Oral chemotherapy orders with at least 1 IV chemotherapy agent included in the regimen were associated with fewer treatment deviations, suggesting that established protocols for IV chemotherapy may be effectively applied to oral chemotherapy to reduce treatment deviations. Overall, a majority (63.2%) of oral chemotherapy orders were initiated in the outpatient setting as opposed to the inpatient setting, which is expected, because the majority of cancer care plans are established in the outpatient clinics.
Our data identified outpatient initiation of oral chemotherapy as a risk factor for treatment deviation, suggesting that transitioning to the inpatient setting poses a risk for the safety of oral chemotherapy. These data also suggest the importance of allowing appropriate time for evaluation of continuing oral chemotherapy rather than immediately ordering for continuation from the before admission medication list.
At our institution, patients with active malignancies are typically assigned to hematology/oncology services, although not exclusively. As expected, oral chemotherapy orders for patients receiving these specialty services had fewer treatment deviations. Although a minority (37.8%) of the oral chemotherapy orders overall were placed within 24 hours of admission, this group had a disproportionately high incidence of treatment deviations and represented an independent risk factor for treatment deviation (Table 4). This may be attributed to oral chemotherapy being ordered for patients before expert evaluation.
Finally, after the initial results of this study, improvements involving our oral chemotherapy process were established at the University of Kansas Hospital that mandated that oral chemotherapy be continued from the before admission medication list within the inpatient setting only under certain conditions. Oral chemotherapy may be ordered for continuation during an acute admission under the guidance of a specialist physician, and only these physicians or attending physicians of the primary medical service (if not the same) can place the order for oral chemotherapy. In addition, provider documentation of the decision to continue the oral chemotherapy must be listed in the EMR, and the orders must undergo pharmacist double-check on verification.
Limitations
Our results should be considered together with the study limitations. These results reflect the practices at the University of Kansas Hospital, and therefore the findings may not be generalizable to other institutions.
In addition, the study methods did not enable us to determine if the treatment deviation originated in the inpatient or the outpatient setting. In the study design, we were less concerned with where the treatment deviation originated and were rather more concerned with whether the inpatient team propagated the treatment deviation.
Furthermore, abnormal treatment parameters were the most prevalent deviations noted, but because of inconsistent documentation, we were unable to determine if the providers had intentionally continued therapy despite not meeting treatment parameters.
Finally, documentation of intentional continuation of oral chemotherapy in the setting of abnormal treatment parameter only occurred in 6 of the 73 orders. Of note, oral chemotherapy treatment deviations were not correlated directly with clinical outcomes in this study, and it is unclear if these treatment deviations affected treatment outcomes.
Conclusion
This study revealed several areas that could be updated to help optimize the safety of oral chemotherapy and helped our organization to prioritize its oral chemotherapy safety efforts. Based on our observation that oral chemotherapy co-administered with IV chemotherapy contained fewer treatment deviations, we inferred that our IV chemotherapy protocols, with a double-check by oncology-trained pharmacists, may be a method to improve oral chemotherapy safety.
When patients were admitted to the hematology/oncology services, treatment deviations occurred less frequently. As a result, our organization now mandates a hematology/oncology consultation for admitted patients before continuing oral chemotherapy from their before admission medication list. Finally, holding the use of oral chemotherapy for newly admitted patients until completing a thorough evaluation may reduce treatment deviations and improve safety.
This study illustrates the need for further quality improvement efforts to enhance the safety of oral chemotherapy. Future measures should focus on oral chemotherapy that is ordered within 24 hours of hospital admission from an outpatient-initiated prescription, because we found that transitioning to the inpatient setting is a critical point in terms of oral chemotherapy safety.
Author Disclosure Statement
Dr Cascone, Dr Hosmer, Mr Henry, and Dr Grauer have no conflicts of interest to report. Dr Mahmoudjafari has received honoraria from Juno Pharmaceuticals.
References
- Weingart SN, Flug J, Brouillard D, et al. Oral chemotherapy safety practices at US cancer centres: questionnaire survey. BMJ. 2007;334:407.
- Collins CM, Elsaid KA. Using an enhanced oral chemotherapy computerized provider order entry system to reduce prescribing errors and improve safety. Int J Qual Health Care. 2011;23:36-43.
- National Cancer Institute. A to Z list of cancer drugs. www.cancer.gov/about-cancer/treatment/drugs. Accessed November 9, 2019.
- Weingart SN, Brown E, Bach PB, et al. NCCN task force report: oral chemotherapy. J Natl Compr Canc Netw. 2008;6:S1-S16.
- Krikorian S, Pories S, Tataronis G, et al. Adherence to oral chemotherapy: challenges and opportunities. J Oncol Pharm Pract. 2019;25:1590-1598.
- Ansari JA. Drug interaction and pharmacist. J Young Pharm. 2010;2:326-331.
- Goldspiel B, Hoffman JM, Griffith NL, et al. ASHP guidelines on preventing medication errors with chemotherapy and biotherapy. Am J Health Syst Pharm. 2015;72:e6-e35.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association best practices for the management of oral oncolytic therapy: pharmacy practice standard. J Oncol Pract. 2019;15:e346-e355.
- Neuss M, Gilmore T, Belderson K, et al. 2016 Updated American Society of Clinical Oncology/Oncology Nursing Society chemotherapy administration safety standards, including standards for pediatric oncology. Oncol Nurs Forum. 2017;44:31-43.
- Lexicomp online. https://online.lexi.com/lco/action/login. [Requires subscription.]
- Xeloda (capecitabine) tablets, for oral use [prescribing information]. San Francisco, CA: Genentech; February 22, 2019.