Acute graft-versus-host disease (GVHD) is a common multi-organ complication that occurs in 20% to 80% of patients undergoing allogeneic hematopoietic stem-cell transplant (HSCT) and causes significant morbidity and mortality.1 Methotrexate, a dihydrofolate reductase inhibitor, is one of the backbone agents used for GVHD prophylaxis together with other agents, such as calcineurin inhibitors, and has been linked to delayed engraftment, hepatotoxicity, and mucositis.2-4
Treatment-induced oral mucositis is one of the most debilitating adverse events from a patient’s perspective, reportedly occurring in 75% to 100% of patients receiving allogeneic HSCT, and is associated with longer periods of hospitalization and significant health and financial costs that profoundly influence patients’ quality of life (QOL).5 Some risk factors for oral mucositis, albeit controversial, include older age, female gender, low body mass index, low baseline glutamine plasma levels, altered baseline oral microbiota, elevated baseline inflammatory markers or milieu, altered salivary function, high-intensity conditioning regimens, and 5, 10-methylenetetrahydrofolate reductase +TT genotype.5,6 Oral mucositis often requires pain management with opioids, nutritional supplementation, and methotrexate dose reduction or omission.3,7
Two studies have reported that ≤69% of patients received all 4 doses of short-course methotrexate (15 mg/m2 on day 1, followed by 10 mg/m2 on days 3, 6, and 11).8,9 Although ambiguity exists around the necessity of the fourth dose of methotrexate in the absence of toxicities, omission of day 11 methotrexate dosing has been associated with decreased overall survival and increased incidence of grade 3 or 4 oral mucositis, which may be a surrogate for worse non–relapse-related mortality.8,9
To optimize the administration of all 4 prophylactic methotrexate doses and potentially decrease the incidence and severity of methotrexate-induced oral mucositis, implementation of folinic acid rescue after each methotrexate dose has been proposed.10-14 Folinic acid, also known as leucovorin, is a reduced form of folate that ultimately enables the return to homeostatic nucleotide synthesis when used as a rescue medication after methotrexate administration.11
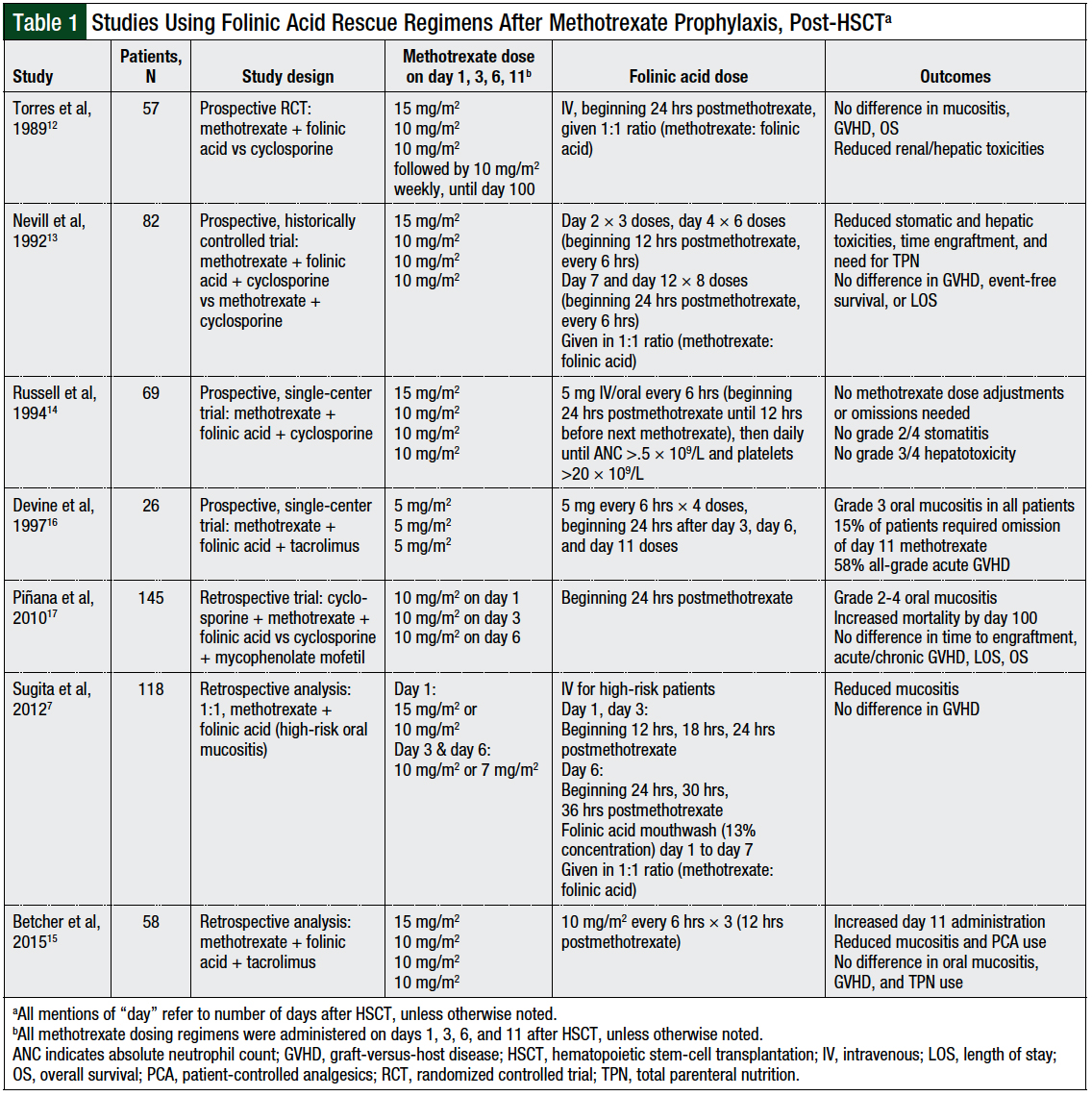
Folinic acid has significantly reduced methotrexate-associated toxicities, improved survival, and increased the tolerability of prophylactic methotrexate, while not reducing methotrexate’s ability to prevent GVHD in patients undergoing HSCT (Table 1).7,12-17
From a pharmacokinetic perspective, murine data have shown that methotrexate-induced complete suppression of blastogenesis occurs within 6 to 8 hours of methotrexate administration.18 Therefore, if folinic acid is administered ≥12 hours after methotrexate, it should not affect methotrexate’s immunosuppressive potential. Despite these findings, the use of folinic acid rescue after methotrexate was reported as standard practice in only 33% to 44% of surveyed adult transplant centers in 2 countries and in a minority of US transplant centers, based on anecdotal findings.4
The primary rationale given by centers not administering folinic acid is the theoretical concern that folinic acid may reduce the therapeutic efficacy of methotrexate.4,13 Such concerns of leucovorin (or folinic acid) selectively rescuing malignant cells and leading to increased risk for relapsed disease and graft rejection have been reported in pediatric clinical trials using higher doses and earlier administration schedules of leucovorin.19,20 These findings, however, have been largely contradicted by other studies, in the setting of significantly lower prophylactic doses of methotrexate and associated lower rescue leucovorin doses.11-13
The European Group for Blood and Marrow Transplantation and the European LeukemiaNet working group proposed the use of folinic acid rescue for adult allogeneic HSCT recipients receiving short-course methotrexate for GVHD prophylaxis21; however, adherence to this recommendation remains low, likely because of the lack of consistent efficacy and safety data supporting the use of folinic acid rescue for this indication.
Our institution predominantly utilizes mini-dose methotrexate (5 mg/m2 on days 1, 3, 6, and 11), without the implementation of folinic acid rescue, coupled with tacrolimus (0.03 mg/kg daily continuous infusion every 24 hours, starting day 1), for GVHD prophylaxis, after myeloablative conditioning and reduced-intensity conditioning allogeneic HSCTs. Given the low doses of methotrexate used and the anticipated low serum blood levels, our institution does not measure methotrexate levels when it is given prophylactically for GVHD.
Given the reported positive, but overall lack of robust evidence supporting the use of folinic acid rescue after administration of mini-dose methotrexate in adults, we conducted this hypothesis-generating study to evaluate the safety profile of mini-dose methotrexate by day 100 and determine the benefit of implementing a folinic acid rescue protocol in our institution.
Methods
The Mount Sinai Hospital, in New York, NY, is a 1144-bed tertiary-care teaching facility affiliated with the Tisch Cancer Institute, a National Cancer Institute (NCI)-designated center that provides robust adult and pediatric autologous and allogeneic HSCT programs. This single-center retrospective chart review was conducted on adult patients who received mini-dose methotrexate for GVHD prophylaxis, after peripheral blood and bone marrow allogeneic HSCTs, from July 1, 2013, to June 30, 2015. We identified patients from our institutional electronic health record system. The study received expedited Institutional Review Board approval.
Data collected included patients’ baseline characteristics, such as age, gender, race, height, actual body weight, body surface area, malignancy diagnosis, most recent chemotherapy or conditioning regimens, stem-cell harvest source, total body irradiation dose, human leukocyte antigen (HLA) mismatch, renal or hepatic function, antimicrobial regimen, GVHD prophylactic regimen, and length of hospital stay.
Additional documented data points included methotrexate administration; oral mucositis grade (using the NCI Common Terminology Criteria for Adverse Events, version 4.0); initiation and duration of total parenteral nutrition, use of patient-controlled analgesics and oral opioids; filgrastim and palifermin use; incidence and severity of GVHD; and the length of time to engraftment (defined as a sustained absolute neutrophil count >500/mm3 for ≥3 days, and sustained platelet count >20/mcL for ≥7 consecutive days, respectively).
Our standard reduced-intensity conditioning regimens during the data collection period included fludarabine 30 mg/m2 intravenous (IV) daily from day 6 to day 2 before HSCT and IV melphalan 70 mg/m2 daily from day 3 to day 2 before HSCT for acute myeloid leukemia (AML), myelofibrosis, and lymphoma; IV fludarabine 30 mg/m2 daily from day 7 to day 3 before HSCT and IV busulfan 0.8 mg/kg every 6 hours from day 4 to day 3 before HSCT for myelodysplastic syndrome (MDS) and AML; IV busulfan 0.8 mg/kg every 6 hours from day 7 to day 5 before HSCT and IV fludarabine 30 mg/m2 daily from day 7 to day 2 before HSCT for myelofibrosis; and total body irradiation 150 cGy twice daily from day 8 to day 6 before HSCT and IV cyclophosphamide 50 mg/kg daily from day 5 to day 2 before HSCT for severe aplastic anemia and myelofibrosis.
The regularly used myeloablative conditioning regimens during the data collection period included IV busulfan 0.5 mg/kg every 6 hours from day 7 to day 4 before HSCT and IV etoposide 60 mg/kg on day 3 before HSCT for MDS, AML, and acute lymphoblastic leukemia (ALL); total body irradiation 150 cGy twice daily from day 7 to day 4 before HSCT and IV cyclophosphamide 60 mg/kg daily from day 3 to day 2 before HSCT for MDS, AML, and ALL; total body irradiation 1000 to 1600 cGy divided twice daily from day 7 to day 4 before HSCT and etoposide 60 mg/kg on day 3 before HSCT for AML and ALL; and IV cyclophosphamide 60 mg/kg daily from day 7 to day 6 before HSCT and IV busulfan 0.8 mg/kg every 6 hours from day 5 to day 2 before HSCT for MDS, AML, and sickle-cell anemia.
Cryotherapy is our standard of care—before, during, and immediately after melphalan administration—to reduce the incidence of oral mucositis. At the time of data collection, we administered palifermin to all patients receiving total body irradiation and myeloablative conditioning regimens before allogeneic HSCTs. As time progressed, however, our practice favored using palifermin primarily in patients receiving total body irradiation and on a select basis for other indications.
The primary outcome of our study was the safety of methotrexate by day 100 after HSCT, with a focus on the incidence and severity of oral mucositis (surrogate markers included the use of total parenteral nutrition and patient-controlled analgesics). Secondary outcomes were the incidence of acute GVHD and overall survival, as well as changes in renal and hepatic function by day 30 after HSCT. All patients were followed until day 100 after HSCT or death, whichever came first.
Microsoft Excel 2000 and SAS version 9.4 (SAS Institute; Cary, NC) were used to compute the statistical analyses.
Results
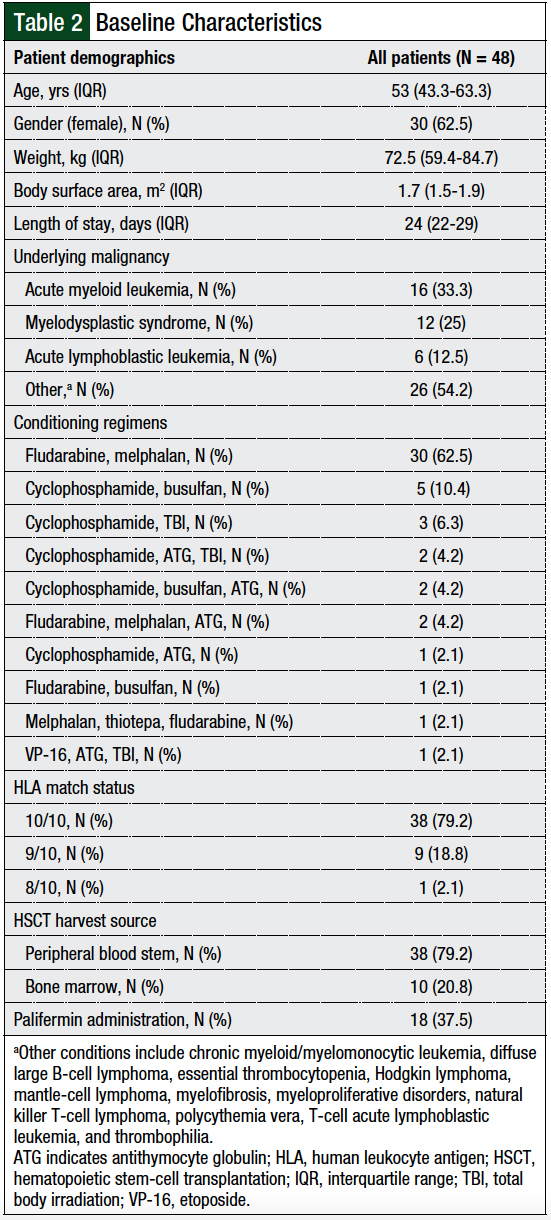
A total of 78 allogeneic HSCT recipients were identified during the specified time frame. Of those patients, 30 were excluded for one of several reasons, including receipt of an alternative GVHD prophylactic regimen (N = 24), short-course methotrexate (N = 5), or receipt of an umbilical cord HSCT (N = 1). A total of 48 patients were qualified for study inclusion and evaluation (Table 2).
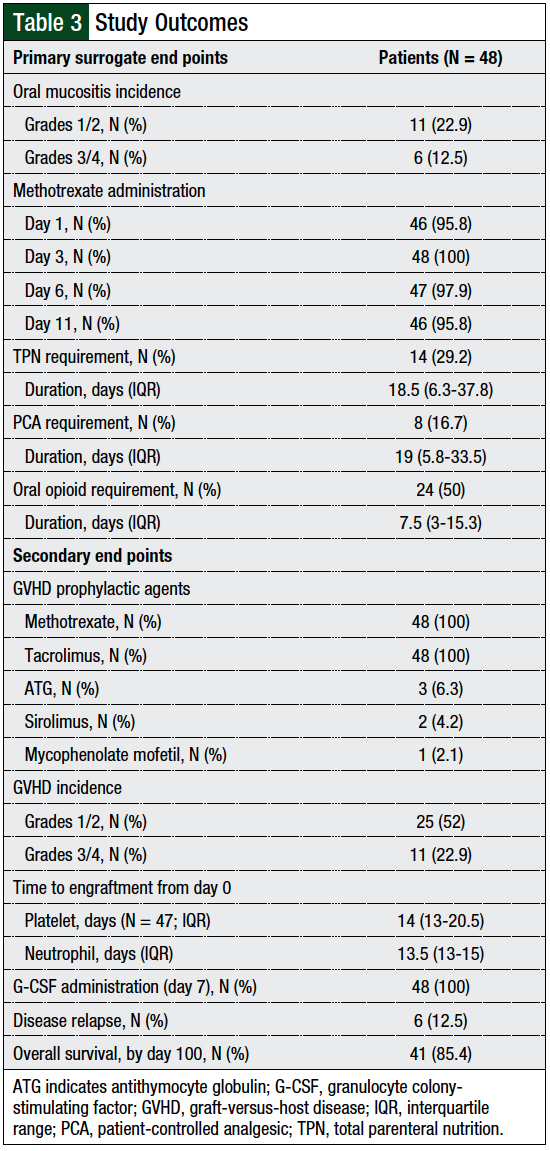
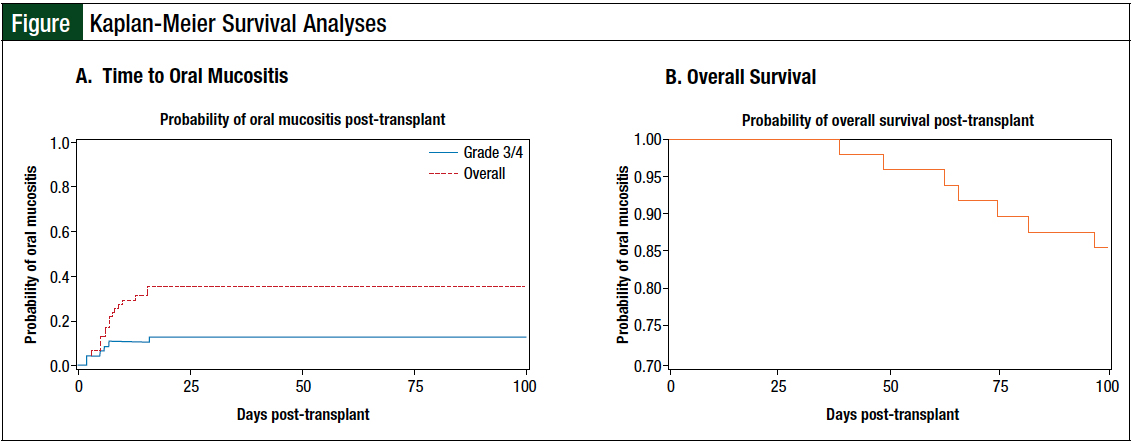
Grade 1 or 2 oral mucositis occurred in 11 (22.9%) patients, and grade 3 or 4 oral mucositis was noted in 6 (12.5%) patients. The use of total parenteral nutrition, patient-controlled analgesics, and oral opioids were required by 29.2%, 16.7%, and 50% of the patients, respectively (Table 3). The first incidence of oral mucositis was reported by day 2 after HSCT and plateaued by day 16 after HSCT (Figure A).
Time to oral mucositis was not affected by the number of methotrexate doses administered (ie, 4 methotrexate doses vs less than 4 methotrexate doses; log-rank, P = .93). Two (4.2%) patients required the day 11 methotrexate dose to be omitted: 1 of them had grade 4 oral mucositis and acute kidney injury, and the other patients had acute kidney injury. Of the 18 patients who received palifermin, 11 had all-grade oral mucositis, 3 of whom received total body irradiation and had grades 1, 3, and 4 oral mucositis, respectively. GVHD and engraftment findings are detailed in Table 3.
We did not find any statistical difference in time to neutrophil engraftment between patients who had oral mucositis and those who did not (log-rank, P = .76). The rate of relapsed disease by day 30 was 12.5%, and the rate of overall survival by day 100 was 85.4% (Figure B). No statistical differences were observed in renal and hepatic function between baseline and day 30.
Discussion
The results of our study compare favorably to clinical trials and consensus recommendations focusing on day 11 methotrexate administration rates; in our study, 95.8% of the patients received the day 11 dose.8,15,21 Furthermore, 35.4% of our patients had all-grade oral mucositis, with grade 1 or 2 occurring in 22.9% of the patients. This compares favorably with safety data from Przepiorka and colleagues, who reported that their entire study cohort had grade 1 (23%) or grade 2 (77%) oral mucositis.3
Our study differed, however, in having a higher rate (12.5%) of grade 3 or grade 4 oral mucositis than reported in other studies.3,13 This clinically significant increase in the proportion of patients with grade 3 or 4 oral mucositis is more consistent with, but still lower than, the increased higher-grade mucositis rate found in another study.7,15
The use of total parenteral nutrition, patient-controlled analgesics, and oral opioids reported in our evaluation highlighted the clinical severity of oral mucositis in our patient population and the likely decrease in QOL seen in our patients with this adverse event.
A total of 75% of patients in our study had all-grade GVHD, with 52% of the patients having grade 1 or 2 and 22.9% of patients having grade 3 or 4 GVHD; these values compare favorably with several studies,7,14 although other studies have reported better rates.3,12,13,16,17 This difference could be attributed to the conditioning intensity, use of total body irradiation, graft source, GVHD prophylactic regimen, reporting method of GVHD incidence, and frequency of omitting day 11 methotrexate dose, in addition to other factors.17 The rates of engraftment, disease relapse, and renal or hepatic impairment were lower in our study than in the control groups in other studies.3,13
Our findings of grade 3 or 4 oral mucositis, the use of total parenteral nutrition, patient-controlled analgesics, and hospital length of stay highlight the need for consideration of alternative management strategies for oral mucositis. Strategies that have been implemented to mitigate or prevent the development of oral mucositis after allogeneic HSCT include maintaining adequate oral hygiene, glutamine supplementation, as well as the use of palifermin, topical and systemic growth factors, interleukin-11, and bacterial decontamination of the oral cavity.22
The other options noted have not been widely accepted as standard of care. In addition, despite the encouraging evidence supporting the efficacy of folinic acid in preventing oral mucositis, the medical community has largely not adopted this approach.
Several factors are causative for post-HSCT complications, such as oral mucositis; the GVHD prophylactic regimen is only one of those factors. Tacrolimus plus methotrexate has been the favored GVHD prophylactic regimen after myeloablative conditioning regimens for allogeneic HSCTs, thanks to its superior efficacy in preventing acute GVHD.23
Additional GVHD prophylactic regimens, such as cyclosporine or tacrolimus plus mycophenolate mofetil and/or sirolimus, are also effective options, with slightly better safety profiles.24 Because 83.3% of our patient population received reduced-intensity conditioning regimens, comparing our rate of oral mucositis to the safety profiles of nonmethotrexate-containing GVHD prophylactic regimens used in reduced-intensity conditioning allogeneic HSCTs may bolster the recommendation to initiate a folinic acid rescue protocol at our institution.
This is supported by a systematic review by Chaudhry and colleagues, who reported an overall 44.6% grade 0 oral mucositis incidence among patients who received non–methotrexate-based GVHD prophylaxis after reduced-intensity conditioning regimens.25 Only 1 of those evaluated trials noted a 45.2% incidence of grade 1 or 2 oral mucositis and a 24.4% incidence of grade 3 or 4 oral mucositis (in patients who received cyclosporine/mycophenolate mofetil).25 In patients receiving <45 mg/m2 cumulative dose of methotrexate for GVHD prophylaxis after reduced-intensity conditioning regimens, 17.6%, 11%, 59.7%, and 65.3% of patients had grade 0, 1, 2 to 4, and 3/4 oral mucositis, respectively.25
Our incidence of all-grade (35.4%) and grade 3 or 4 (12.5%) oral mucositis, secondary to administration of prophylactic methotrexate with tacrolimus, is less than the rates reported in that systematic analysis.25 By contrast, Kornblit and colleagues reported a 0% incidence of mucositis in each of the 2 arms dosed with either tacrolimus-mycophenolate mofetil or tacrolimus-mycophenolate mofetil-sirolimus, in patients who received nonmyeloablative conditioning regimens.26 Given the nonmyeloablative nature of the conditioning therapy used in that study, the safety results were expectedly more favorable than what we reported for our patients.25
It is important to be discerning when drawing conclusions from such interstudy similarities and discrepancies, because of the inherent differences in each study’s design, including, but not limited to, stem-cell sources, HLA matching, supportive care regimens, and patient comorbidities. Based on the results of our study, in addition to the evidence supporting the use of a folinic acid rescue protocol and the importance of patient QOL, there may be a role for evaluating the potential benefits of a folinic acid rescue regimen in our patients with hematologic malignancies who had allogeneic HSCT.
Limitations
Although our results are telling, several study limitations must be recognized. This single-center, retrospective chart review had a small sample size and, therefore, the results may not be generalizable to a larger, more diverse patient population.
Furthermore, a comparator group was not included, which would have enabled the assessment of the safety profile of mini-dose methotrexate with folinic acid rescue versus mini-dose methotrexate without folinic acid rescue.
Regarding opioid use, we only collected data for oral opioid use and patient-controlled analgesic administration, and did not collect data relating to the use of IV opioid formulations, so as to emphasize the stark contrast in the likely severity of patients’ oral mucositis. The use of total parenteral nutrition and patient-controlled analgesics was confounded by multiple indications for their use, such as generalized poor nutritional status and back pain, respectively.
The incidence of oral mucositis in our study could have been confounded by the use of palifermin, given to 37.5% of our patients. Despite the expected protective effects of palifermin, however, 11 (29.3%) patients receiving it had all-grade oral mucositis. The incidence of oral mucositis could have been underreported because of the use of palifermin, which could potentially emphasize the benefit of alternative oral mucositis prophylactic approaches at our institution.
Moreover, complications secondary to oral mucositis, such as rates of infection and febrile neutropenia, were not regularly collected and should be considered useful data points in future studies, given the risk for microbial translocation in the setting of friable mucosal linings.
Finally, because the analysis focused on recipients of allogeneic HSCT from peripheral blood or bone marrow harvest sources, the results are less translatable to recipients of donor lymphocyte infusions, CD34+-selected stem-cell boosts, and umbilical cord HSCTs.
Although comparing our results with those from other trials may reflect positively on our practice, one must consider differences in oral mucositis grading, clinical variance in reporting GVHD, the use of different conditioning and GVHD prophylactic regimens among transplant centers, stem-cell source, type of donor, and patient-specific risk factors.
Conclusions
Given the formidable implications and continued reporting of severe methotrexate-induced oral mucositis despite the implementation of recommended supportive care strategies, evidenced in this study and in the broader literature, it may be appropriate to reassess the benefit of using folinic acid rescue, even for mini-dose methotrexate, as a result of its excellent tolerability, equivalent rates of acute GVHD, ease of administration, and low cost. Our findings lead us to believe that the adoption of a folinic acid rescue protocol may aid in collectively reducing the incidence of severe oral mucositis, the need for prolonged opioid pain management, and hospital length of stay, as well as increasing a patient’s QOL. Although the current lack of consensus regarding the specific dose, schedule, and time of initiation of folinic acid makes its use contentious, nonetheless we believe it would be clinically reasonable to administer this therapy to patients at high risk for severe oral mucositis.
Once a folinic acid protocol is implemented, we could perform a prospective study with the new folinic acid rescue regimen to compare it with the historical controls already described in this manuscript. Because the available studies have focused on the implementation of folinic acid rescue after a short-course methotrexate prophylaxis, analysis of the efficacy of a folinic acid rescue protocol after mini-dose methotrexate prophylaxis would be a nuanced addition to the current literature.
Based on the reported regimens and national anecdotal practices, we would recommend considering the initiation of folinic acid 5 mg/m2 intravenously or orally every 6 hours for 4 doses, starting 24 hours after mini-dose methotrexate administrations on days 1, 3, 6, and 11. Based on the current pharmacokinetic data, there would not be a need for serum methotrexate-guided dosing of folinic acid rescue.
Randomized controlled trials would be warranted to investigate the impact of folinic acid rescue on the incidence of oral mucositis, GVHD, disease relapse, and overall survival. In the interim, clinicians are encouraged to continue implementing evidence-based strategies, within the construct of a collaborative, interdisciplinary team, to analyze critically the utility of adopting alternative supportive care measures for our highest-risk patients who undergo an HSCT.
Acknowledgments
The authors wish to acknowledge the support of Joanne Meyer, MS, PharmD, and Sara S. Kim, PharmD, BCOP, Department of Pharmacy, Mount Sinai Hospital, as well as the Biostatistics Shared Resource Facility, Icahn School of Medicine at Mount Sinai.
Author Disclosure Statement
Dr Solodokin, Dr Steinberg, and Ms Moshier have no conflicts of interest to report.
References
- Martin PJ, Rizzo JD, Wingard JR, et al. First and second-line systemic treatment of acute graft-versus-host disease: recommendations of the American Society of Blood and Marrow Transplantation. Biol Blood Marrow Transplant. 2012;18:1150-1163.
- Chao NJ, Schmidt GM, Niland JC, et al. Cyclosporine, methotrexate, and prednisone compared with cyclosporine and prednisone for prophylaxis of acute graft-versus-host disease. N Engl J Med. 1993;329:1225-1230.
- Przepiorka D, Ippoliti C, Khouri I, et al. Tacrolimus and minidose methotrexate for prevention of acute graft-versus-host disease after matched unrelated donor marrow transplantation. 1996;88:4383-4389.
- Bhurani D, Schifter M, Kerridge I. Folinic acid administration following MTX as prophylaxis for GVHD in allogeneic HSCT centres in Australia and New Zealand. Bone Marrow Transplant. 2008;42:547-550.
- Niscola P, Romani C, Cupelli L, et al. Mucositis in patients with hematologic malignancies: an overview. Haematologica. 2007;92:222-231.
- Barasch A, Peterson DE. Risk factors for ulcerative oral mucositis in cancer patients: unanswered questions. Oral Oncol. 2003;39:91-100.
- Sugita J, Matsushita T, Kashiwazaki H, et al. Efficacy of folinic acid in preventing oral mucositis in allogeneic hematopoietic stem cell transplant patients receiving MTX as prophylaxis for GVHD. Bone Marrow Transplant. 2012;47:258-264.
- Hamilton BK, Rybicki L, Haddad H, et al. Does day 11 omission of methotrexate due to toxicity influence the outcome in myeloablative hematopoietic cell transplant? Results from a single-center retrospective cohort study. Blood Cancer J. 2015;5:e344.
- Kumar S, Wolf RC, Chen MG, et al. Omission of day +11 methotrexate after allogeneic bone marrow transplantation is associated with increased risk of severe acute graft-versus-host disease. Bone Marrow Transplant. 2002;30:161-165.
- Gratwohl AA, Bull MI, Graw RG Jr, et al. Methotrexate and citrovorum factor after histoincompatible allogeneic bone marrow transplants in dogs. Acta Haematol. 1978;60:233-242.
- Robien K, Schubert MM, Yasui Y, et al. Folic acid supplementation during methotrexate immunosuppression is not associated with early toxicity, risk of acute graft-versus-host disease or relapse following hematopoietic transplantation. Bone Marrow Transplant. 2006;37:687-692.
- Torres A, Martinez F, Gomez P, et al. Cyclosporin A versus methotrexate, followed by rescue with folinic acid as prophylaxis of acute graft-versus-host disease after bone marrow transplantation. Blut. 1989;58:63-68.
- Nevill TJ, Tirgan MH, Deeg HJ, et al. Influence of post-methotrexate folinic acid rescue on regimen-related toxicity and graft-versus-host disease after allogeneic bone marrow transplantation. Bone Marrow Transplant. 1992;9:349-354.
- Russell JA, Woodman RC, Poon MC, et al. Addition of low-dose folinic acid to a methotrexate/cyclosporin A regimen for prevention of acute graft-versus-host disease. Bone Marrow Transplant. 1994;14:397-401.
- Betcher J, Shelton T, Slack JL, et al. Folinic acid rescue after methotrexate graft versus host disease prophylaxis to reduce mucositis and improve the probability of day +11 methotrexate administration - role of the hematopoietic cell transplant pharmacist in development of program guidelines. Biol Blood Marrow Transplant. 2015;21(2 suppl):Abstract 124.
- Devine SM, Geller RB, Lin LB, et al. The outcome of unrelated donor bone marrow transplantation in patients with hematologic malignancies using tacrolimus (FK506) and low dose methotrexate for graft-versus-host disease prophylaxis. Biol Blood Marrow Transplant. 1997;3:25-33.
- Piñana JL, Valcárcel D, Fernández-Avilés F, et al. MTX or mycophenolate mofetil with CsA as GVHD prophylaxis after reduced-intensity conditioning PBSCT from HLA-identical siblings. Bone Marrow Transplant. 2010;45:1449-1456.
- Kempf RA, Mitchell MS. Effects of chemotherapeutic agents on the immune response. II. Cancer Invest. 1985;3:23-33.
- Skärby TV Ch, Anderson H, Heldrup J, et al; for the Nordic Society of Paediatric Haematology and Oncology (NOPHO). High leucovorin doses during high-dose methotrexate treatment may reduce the cure rate in childhood acute lymphoblastic leukemia. Leukemia. 2006;20:1955-1962.
- Lindqvist H, Remberger M, Harila-Saari A, et al. Folinic acid supplementation in higher doses is associated with graft rejection in pediatric hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2013;19:325-328.
- Ruutu T, Gratwohl A, de Witte T, et al. Prophylaxis and treatment of GVHD: EBMT–ELN working group recommendations for a standardized practice. Bone Marrow Transplant. 2014;49:168-173. Erratum in: Bone Marrow Transplant. 2014;49:319.
- Cutler C, Li S, Kim HT, et al. Mucositis after allogeneic hematopoietic stem cell transplantation: a cohort study of methotrexate- and non–methotrexate-containing graft-versus-host disease prophylaxis regimens. Biol Blood Marrow Transplant. 2005;11:383-388.
- Ram R, Gafter-Gvili A, Yeshurun M, et al. Prophylaxis regimens for GVHD: systematic review and meta-analysis. Bone Marrow Transplant. 2009;43:643-653.
- Cutler C, Logan B, Nakamura R, et al. Tacrolimus/sirolimus vs tacrolimus/methotrexate as GVHD prophylaxis after matched, related donor allogeneic HCT. Blood. 2014;124:1372-1377.
- Chaudhry HM, Bruce AJ, Wolf RC, et al. The incidence and severity of oral mucositis among allogeneic hematopoietic stem cell transplantation patients: a systematic review. Biol Blood Marrow Transplant. 2016;22:605-616.
- Kornblit B, Maloney DG, Storer BE, et al. A randomized phase II trial of tacrolimus, mycophenolate mofetil and sirolimus after non-myeloablative unrelated donor transplantation. Haematologica. 2014;99:1624-1631.