Trastuzumab is a human epidermal growth factor receptor (HER)-2/neu inhibitor approved by the US Food and Drug Administration for the treatment of HER-2–positive breast cancer. Although acneiform rash, also known as papulopustular eruption, is a common cutaneous toxicity of epidermal growth factor receptor (EGFR) inhibitors,1 it has not been shown to be associated with trastuzumab therapy. In this article, we report on a patient who developed grade 3 acneiform rash (based on grading criteria by National Cancer Institute-Common Terminology Criteria for Adverse Events) after receiving trastuzumab therapy.
Case Report
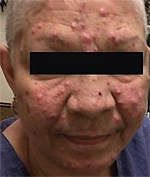 A 51-year-old woman with stage I estrogen receptor– weakly positive, progesterone receptor–negative, HER-2–positive breast cancer underwent a bilateral modified radical mastectomy, followed by a regimen of docetaxel/carboplatin/trastuzumab (TCH). At the time of treatment, she was not taking any other medications or herbal supplements. After receiving the first cycle of TCH, she presented with acneiform eruptions, predominantly on the face and scalp, which became more severe and extensive after the second cycle. Compared with the classic presentations of EGFR inhibitor–associated acneiform rash, the papules and pustules were larger in size (>0.5 to 1 cm; Figure).
A 51-year-old woman with stage I estrogen receptor– weakly positive, progesterone receptor–negative, HER-2–positive breast cancer underwent a bilateral modified radical mastectomy, followed by a regimen of docetaxel/carboplatin/trastuzumab (TCH). At the time of treatment, she was not taking any other medications or herbal supplements. After receiving the first cycle of TCH, she presented with acneiform eruptions, predominantly on the face and scalp, which became more severe and extensive after the second cycle. Compared with the classic presentations of EGFR inhibitor–associated acneiform rash, the papules and pustules were larger in size (>0.5 to 1 cm; Figure).
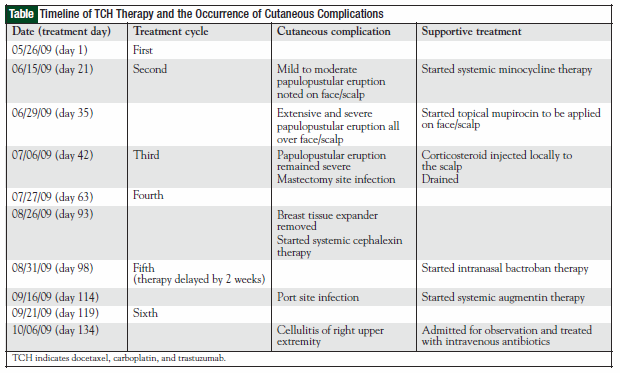
She was treated with oral minocycline and topical mupirocin cream, both of which produced only a modest response. She was then given a local corticosteroid injection to the scalp lesions and showed a marked improvement. Despite the improvement of the skin lesions, however, she experienced three separate episodes of bacterial skin infections in the setting of nonneutropenia (leukocyte counts: range, 5300- 7400/μL), all of which occurred at 3- to 4-week intervals (Table). The first infectious episode occurred 1.5 months after the initiation of chemotherapy at the left mastectomy site, followed by an infection at the port site, then a cellulitis of the right upper extremity.
Despite the severe cutaneous complications, the patient has successfully completed all six cycles of TCH at full weight-based dose (Table), and is currently being treated with intravenous trastuzumab (6 mg/kg every 3 weeks) as single-agent therapy. She experienced complete resolution of acneiform eruption, but with residual facial hyperpigmentations.
Discussion
Drug-induced acneiform rash and acne vulgaris share a close resemblance in appearance, but they differ pathologically and etiologically.2 Unlike acneiform rash, which is predominantly inflammatory papulopustular eruptions without the presence of comedones or any apparent infectious etiology, acne vulgaris is characterized by the presence of sebum and comedones.2-4
Like EGFR, HER-2 receptors have been shown to play a role in keratinocyte differentiations in the skin5; hence, the observed cutaneous toxicity in our patient could be associated with the deregulated process of keratinocyte differentiations, which is related to trastuzumab therapy. Although “acne” has been reported in 2% of trastuzumab-treated patients,6 clinical presentations of acne differ from the acneiform rash we observed in our patient. Likewise, although docetaxel has been associated with various types of cutaneous toxicities, this specific dermatologic toxicity—acne iform rash—has not yet been described with docetaxel therapy.7 Based on an objective causality assessment scale,8 trastuzumab is a possible cause of the cutaneous toxicity observed in our patient. The pattern of trastuzumabinduced acneiform eruption appears to resemble that of EGFR inhibitors in that the eruption subsided with multiple exposure.
Drug-induced pustules, in general, have been shown to be sterile. Nonetheless, the increased Staphylococcus carrier state and/or the im paired skin barriers in patients with severe papulopustular eruption have been suggested as the possible causes for developing secondary bacterial infections. 1 In fact, a recent study showed that >50% of cases with drug-induced pustules were carriers for Staphylococcus.9 In our patient, bacterial cultures were not obtained at the time of acneiform eruption, thus the causal relationship could not be clearly established.
Conclusion
Current management strategies for acneiform rash include alcohol-free topical emollient, oral antihistamine, topical clindamycin gel, and a tetracycline-derivative systemic antibiotic. Our patient responded well to local corticosteroid therapy, but the use of corticosteroids in acneiform rash remains controversial.
Based on our experience, vigilant surveillance for infectious complications is warranted in patients who develop severe drug-induced acneiform rash. Further - more, in light of documented cases of methicillin-resistant Staphylococcus aureus superinfection associated with acneiform rash,1 nasal sterilization has also been suggested as a preventive strategy against bacterial skin infection.1,9
Therefore, in patients who develop severe druginduced acneiform eruption, it may be deemed appropriate to obtain bacterial cultures and prescribe a course of nasal mupirocin, at the time of eruption, to eradicate Staphylococcus colonization and minimize the risk of developing secondary bacterial infections.
Disclosure Drs Kim and Adelson did not report any potential financial conflicts of interest.
References
- Agero AL, Dusza SW, Benvenuto-Andrade C, et al. Dermatologic side effects associated with the epidermal growth factor receptor inhibitors. J Am Acad Dermatol. 2006;55:657-670.
- Shah NT, Kris MG, Pao W, et al. Practical management of patients with nonsmall- cell lung cancer treated with gefitinib. J Clin Oncol. 2005;23:165-174.
- Busam KJ, Capodieci P, Motzer R, et al. Cutaneous side-effects in cancer patients treated with the antiepidermal growth factor receptor antibody C225. Br J Dermatol. 2001;144:1169-1176.
- Lacouture ME. Mechanisms of cutaneous toxicities to EGFR inhibitors. Nat Rev Cancer. 2006;6:803-812.
- Maguire HC Jr, Jaworsky C, Cohen JA, et al. Distribution of neu (c-erbB-2) protein in human skin. J Invest Dermatol. 1989;92:786-790.
- Herceptin (trastuzumab) [package insert]. San Francisco, CA: Genentech Inc; March 2009.
- Taxotere (docetaxel) [package insert]. Bridgewater, NJ: sanofi-aventis; October 2010.
- Naranjo CA, Busto U, Abel JG, Sellers EM. Empiric delineation of the probability spectrum of adverse drug reactions. Clin Pharmacol Ther. 1981;29:267-268.
- Amitay-Laish I, David M, Stemmer SM. Staphylococcus coagulase-positive skin inflammation associated with epidermal growth factor receptor-targeted therapy: an early and a late phase of papulopustular eruptions. Oncologist. 2010;15:1002-1008.