Chemotherapy-induced nausea and vomiting (CINV) is estimated to affect more than 60% of patients with cancer,1 and has negative consequences for patients’ quality of life, treatment adherence, and outcomes.2,3 The most important risk factor for CINV is the emetogenic potential of the chemotherapy agent used4; consequently, recommendations of antiemetic guidelines are based on the emetogenicity of the chemotherapy agents being considered.5-7 Patient characteristics, such as intensive previous chemotherapy treatment, a history of nausea or vomiting, female sex, and age <50 years, have also been associated with increased risk for CINV.3,7-9
Combination antiemetic regimens that target multiple emesis-related pathways involving neurotransmitters such as dopamine, serotonin (5-hydroxytryptamine-3; 5-HT3), and substance P are the standard of care for CINV prophylaxis.5-7 At present, the main agents used for CINV prophylaxis are 5-HT3 receptor antagonists, neurokinin-1 (NK-1) receptor antagonists, and corticosteroids such as dexamethasone.5-7 Additional agents in the clinician’s armamentarium include medications with a longer duration of action, such as rolapitant or netupitant (NK-1 receptor antagonists),5-7 and the second-generation 5-HT3 receptor antagonist palonosetron.10-12 Netupitant is only available in combination with palonosetron, not as a single agent.7
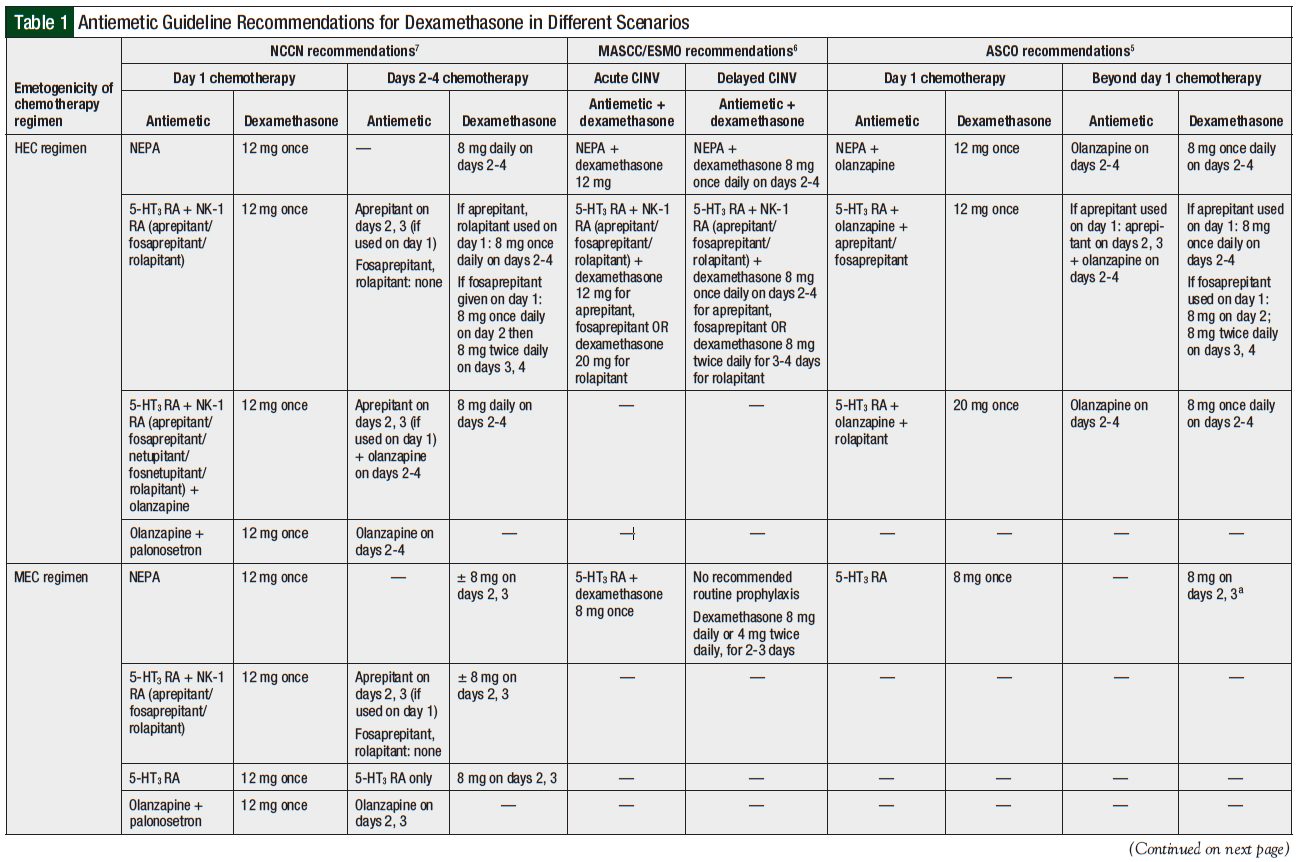
Corticosteroids are known to have antiemetic efficacy in acute and delayed settings13,14 and have been used since the 1980s as single agents or in combination with other antiemetics.15,16 International guidelines usually recommend dexamethasone in combination with other classes of antiemetic agents for prophylaxis against moderately emetogenic chemotherapy (MEC) and/or highly emetogenic chemotherapy (HEC) from day 1 of chemotherapy administration.5-7 With regimens likely to cause significant delayed nausea and/or vomiting, dexamethasone may be continued for up to 2, 3, or 4 more days (Table 1).5-7,13
Dexamethasone-sparing or dose-modification approaches may be applicable when a patient’s exposure to corticosteroids must be limited (eg, the presence of comorbidities or patient preference). Early clinical trials of dexamethasone as an antiemetic agent reported that relatively low doses of dexamethasone showed antiemetic efficacy, without a discernible dose-response relationship.14-17 Recent studies involving 5-HT3 and/or NK-1 receptor antagonist combinations also support the use of dexamethasone-sparing approaches (eg, dexamethasone coadministration on day 1 only), without loss of protection against delayed or acute CINV.18-24
Among antiemetic options for HEC and MEC regimens listed in international guidelines,5-7 an oral fixed-dose combination of 2 long-acting agents, netupitant and palonosetron (NEPA; Akynzeo), is now available in several countries.25,26 These 2 agents have longer elimination half-lives (ie, netupitant, 88 hours; palonosetron, 44-50 hours) than their older counterparts (ie, aprepitant, 9-13 hours; granisetron, 10 hours; ondansetron, 4 hours).26-28 This simplifies the dosing schedules and offers additional convenience of using 1 capsule of the combination therapy instead of multiple tablets of the older agents over several days.5,7
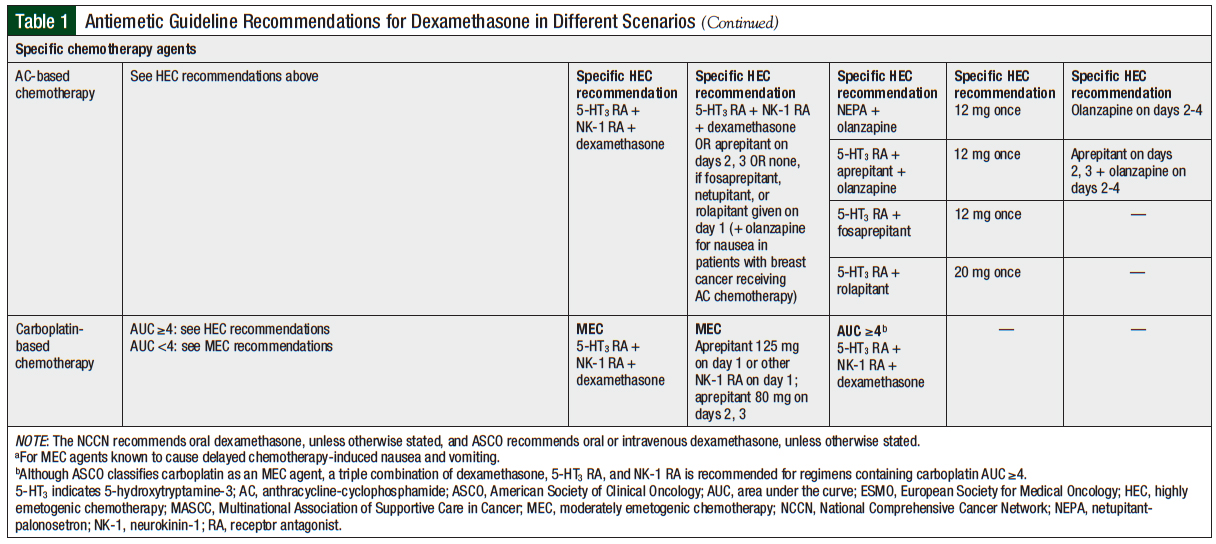
For example, if aprepitant is selected as prophylaxis for delayed emesis from anthracycline and cyclophosphamide (AC)-based regimens, such regimens are used on days 1, 2, and 3, and olanzapine is added on days 2 through 4; by contrast, NEPA-based prophylaxis requires a single dose on day 1, and olanzapine is added on days 2 through 4 (Table 1).5-7
These advantages are especially relevant in light of antiemetic guideline recommendations for MEC and HEC regimens, namely 2- to 4-drug combinations of 5-HT3 receptor antagonists and other agents, which are dosed up to 4 days (Table 1).5-7 It is important to consider reducing the dexamethasone dose when administering it with netupitant, a cytochrome (CY) P3A4 inhibitor that increases systemic exposure to dexamethasone, a CYP3A4 substrate. Nonetheless, NEPA’s prolonged action could help to mitigate the concerns of polypharmacy and medication nonadherence.29
In addition to dosing convenience, NEPA provides good control of acute- and delayed-phase CINV, comparing favorably with other 5-HT3 plus NK-1 receptor antagonist combinations.30-34 Compared with other triple-drug combinations of a 5-HT3 receptor antagonist (ondansetron or granisetron) plus aprepitant and dexamethasone,33,34 the combination of NEPA plus dexamethasone was associated with significantly higher complete response rates in the delayed phase of all cycles compared with the combination of ondansetron, aprepitant, and dexamethasone (86%-98% vs 64%-69%, respectively),33 and significantly lower rates of CINV and rescue medication use over 5 days compared with the combination of granisetron, aprepitant, and dexamethasone (8% vs 13%-15%, respectively).34
A similar trend favoring NEPA was observed by Gralla and colleagues, with higher complete response rates with NEPA plus dexamethasone for HEC (79%-91%) and MEC (80%-93%) regimens compared with aprepitant plus palonosetron and dexamethasone for HEC (58%-86%) and MEC (82%-89%) regimens.31
Uncontrolled CINV in the first chemotherapy cycle markedly increases the likelihood of anticipatory nausea in subsequent cycles.35 Effective prophylaxis upfront, therefore, represents a better strategy than antiemetic escalation therapy for CINV-free treatment across the duration of chemotherapy administration. The efficacy of NEPA for CINV prophylaxis was shown to be sustained during multiple cycles of chemotherapy.30,32-34,36
Of note, 2 studies in patients with breast cancer who received multiple cycles of AC chemotherapy indicated that the use of NEPA permitted flexibility with dexamethasone administration (ie, only day 1 of each cycle, or from days 1 to 3) without the loss of CINV protection.33,36
As a triple antiemetic combination for HEC and MEC, the European and the US prescribing information for NEPA recommend the coadministration of dexamethasone with NEPA on day 1 (Table 2).25,26 Dexamethasone may be continued on days 2 through 4 for HEC regimens.26 Dexamethasone is widely used with 5-HT3 receptor antagonists and NK-1 receptor antagonists for CINV prophylaxis.5-7
With a wider range of antiemetic agents currently available, guidance is needed for the use of appropriate, individualized dexamethasone-sparing regimens for CINV prophylaxis. We report a set of 6 patient cases that illustrate the individualization of dexamethasone dosing plus NEPA in patients who received MEC and/or HEC regimens, while maintaining good control of nausea and vomiting.
Case 1. Epirubicin and Cyclophosphamide in a Patient with Early-Stage TNBC
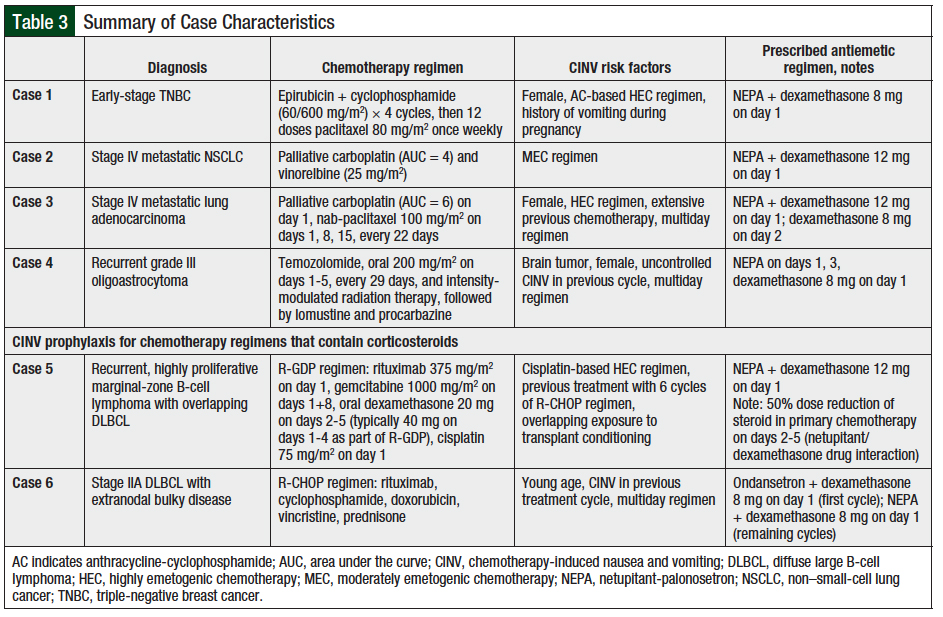
A 56-year-old woman with early-stage (pT1pN0) grade 3 triple-negative breast cancer (TNBC) had undergone breast-conserving therapy and subsequently received adjuvant chemoradiotherapy. She received initial treatment with 4 cycles of chemotherapy with the AC regimen of epirubicin 60 mg/m2 and cyclophosphamide 600 mg/m2, which is classified as a HEC regimen, followed by 12 doses of paclitaxel 80 mg/m2 once weekly.
The patient did not have any comorbidities, but she reported a history of vomiting during pregnancy. Her other risk factors for CINV were female sex and receiving an AC-based chemotherapy regimen (Table 3).
For CINV prophylaxis, the patient received NEPA plus dexamethasone 8 mg, administered on day 1 of each of the epirubicin plus cyclophosphamide chemotherapy cycles. Dexamethasone was omitted after day 1, which is according to the Multinational Association of Supportive Care in Cancer (MASCC)/European Society for Medical Oncology (ESMO) guideline recommendations6 and the prescribing information for NEPA.26
The National Comprehensive Cancer Network (NCCN) guidelines, however, recommend the use of dexamethasone 8 mg from day 2 through day 4 for HEC regimens,7 whereas the American Society of Clinical Oncology (ASCO) guidelines recommend a 4-drug combination of an NK-1 receptor antagonist, a 5-HT3 receptor antagonist, dexamethasone, and olanzapine; olanzapine should be continued from day 2 through day 4 for AC-based HEC regimens.5
The patient reported no vomiting and only mild (grade 1) nausea, which was controlled with metoclopramide.
Case 2. Carboplatin and Vinorelbine in a Patient with Stage IV NSCLC
This was an 86-year-old man with stage IV contralateral metastatic non–small-cell lung cancer (NSCLC). The patient’s Eastern Cooperative Oncology Group (ECOG) performance status score was 1. He also had type 2 diabetes mellitus and was taking oral antidiabetic medication. He received palliative chemotherapy consisting of 3 cycles of carboplatin (for target area under the curve [AUC] of 4) plus vinorelbine 25 mg/m2 (Table 3). The only risk factor for CINV in this patient was the receipt of carboplatin for target AUC of 4.
The NCCN guidelines define carboplatin dose used for a target AUC ≥4 as HEC and AUC <4 as MEC.7 Although the MASCC/ESMO guidelines do not differentiate emetic risk with carboplatin by AUC,6 and the ASCO guidelines regard carboplatin AUC ≥4 as moderately emetogenic,5 all 3 guidelines recommend a triple antiemetic combination of a 5-HT3 receptor antagonist, an NK-1 receptor antagonist, and a steroid or dexamethasone. The NCCN guidelines also recommend continuing dexamethasone therapy beyond day 1.7
Although the use of dexamethasone therapy for CINV arising from carboplatin-based regimens is not explicitly addressed in the prescribing information of NEPA,26 the combination of single-dose NEPA plus dexamethasone may provide good control of nausea and/or vomiting, according to a post-hoc analysis of patients who received carboplatin-based chemotherapy in a clinical trial of NEPA plus dexamethasone versus aprepitant plus palonosetron and dexamethasone.31
More recent analyses also support the antiemetic efficacy of NEPA in subsets of patients who received carboplatin therapy for lung cancer and gynecologic cancers.37
In accordance with most of the recommendations above, the patient received NEPA plus dexamethasone 12 mg on day 1 of each treatment cycle. Dexamethasone was omitted after day 1. This approach was justified, considering that the use of corticosteroids is not advisable in patients with diabetes mellitus, because these agents are known to reduce insulin sensitivity.7,23,38 The patient reported no nausea or vomiting during the 3 cycles of chemotherapy.
Case 3. Carboplatin and Nab-Paclitaxel in a Heavily Pretreated Patient with Stage IV NSCLC
Our third case also involved carboplatin treatment in a 54-year-old woman with stage IV NSCLC (adenocarcinoma) who presented with mediastinal lymph nodes, lung metastases, and pleural effusions. The clinical management of this patient was complicated, because of her history of TNBC, for which she had received 4 previous lines of treatment, including endocrine and palliative chemotherapies.
The patient’s ECOG performance status score was 1 to 2, and she received 3 cycles of carboplatin (AUC = 6) on day 1 and nab-paclitaxel 100 mg/m2 on days 1, 8, and 15, every 22 days (Table 3). As described in Case 2, for carboplatin-containing regimens for target AUC ≥4, current guidelines recommend a triple-drug antiemetic regimen of a 5-HT3 receptor antagonist, an NK-1 receptor antagonist, and dexamethasone.5-7
The patient’s main CINV risk factors included female sex and receipt of carboplatin for target AUC ≥4. Moreover, her poor performance status and previous exposure to several palliative chemotherapy regimens presented a challenging clinical situation. For CINV prophylaxis, NEPA plus dexamethasone 12 mg was administered on day 1 of each treatment cycle, in accordance with international guideline recommendations and NEPA prescribing information.5,7,25,26
In addition, because this patient was heavily pretreated and was receiving fifth-line therapy, it was decided to administer dexamethasone 8 mg on day 2 of each chemotherapy cycle, as a precaution and consistent with the NCCN and ASCO guidelines.5,7 Emesis was well-controlled, and the patient only complained of mild (grade 1) nausea during the 3 treatment cycles.
Case 4. Temozolomide Therapy in a Patient with Grade III Oligoastrocytoma
Case 4 was a 54-year-old woman with recurrent oligoastrocytoma (World Health Organization grade III). Her ECOG performance status score was 0 to 1, and her comorbidities included arterial hypertension and recurrent seizures that were well-controlled. She started temozolomide therapy in 2012, and in 2014 she underwent intensity-modulated radiation therapy, followed by lomustine and procarbazine in 2015.
During her course of temozolomide (oral 200 mg/m2 on days 1-5 every 29 days), which is classified as a MEC regimen, she received ondansetron as antiemetic prophylaxis in the earlier cycles, but her emesis control remained unsatisfactory (Table 3). In addition, the patient expressed a preference for limiting exposure to steroid therapy.
Therefore, she was prescribed NEPA, which was dosed twice weekly on days 1 and 3 in subsequent chemotherapy cycles. This alternate-day antiemetic dosing interval was based on the elimination half-life of netupitant and palonosetron in patients with cancer (ie, 88 hours and 48 ± 19 hours, respectively)25; a similar strategy was reported to provide good control of CINV in patients receiving multiday chemotherapy.39,40 Dexamethasone 8 mg was prescribed on day 1 only in all cycles, despite her situation, which would have warranted dexamethasone dosing for the duration of temozolomide treatment.
Brain tumors are known to cause nausea41,42; hence, the risk factors in this patient included her oligoastrocytoma diagnosis, female sex, and uncontrolled CINV with ondansetron in previous chemotherapy cycles.35 After the introduction of NEPA plus dexamethasone for emesis control, the patient only complained of mild (grade 1) nausea. This case illustrates the efficacy of multiple-day dosing of NEPA as part of a dexamethasone-sparing regimen that improves nausea control.
The next 2 cases illustrate some principles for use of dexamethasone with NEPA after day 1 in corticosteroid-containing regimens that are often used in patients with hemato-oncology diagnoses.
Case 5. R-GDP Regimen in Marginal-Zone B-Cell Lymphoma
This case describes steroid dose reduction related to the interaction between dexamethasone and netupitant. A 77-year-old man received treatment for proliferative marginal-zone B-cell lymphoma with overlapping diffuse large B-cell lymphoma (DLBCL). In 2012, he received 6 cycles of the rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) regimen after presenting with stage IV disease involving the bone marrow and pleura.
His ECOG performance status score was 1, and his arterial hypertension was the only comorbidity of note. After disease recurrence, the patient received 3 cycles of the rituximab, gemcitabine, dexamethasone, and cisplatin (R-GDP) regimen. This included rituximab 375 mg/m2 on day 1, gemcitabine 1000 mg/m2 on days 1 and 8, oral dexamethasone 20 mg on days 2 through 5 (different from the typical dexamethasone 40 mg on days 1-4 as part of R-GDP), and cisplatin 75 mg/m2 on day 1. He then had autologous stem-cell transplant.
This patient’s main risk factor was treatment with cisplatin-based HEC (Table 3). For CINV prophylaxis, NEPA plus dexamethasone 12 mg was administered on day 1 of each R-GDP cycle. Although current guidelines and the prescribing information for NEPA recommend continuing dexamethasone beyond day 1 for HEC regimens, additional dexamethasone dosing was omitted on day 2 through day 4, because the patient received dexamethasone 20 mg on day 2 through day 5 as part of the R-GDP regimen.
Of note, the dexamethasone dose within the R-GDP regimen was reduced from 40 mg to 20 mg after day 1, because of the drug interaction of dexamethasone and netupitant. This is in accordance with the prescribing information for NEPA, which recommends an approximately 50% dose reduction of dexamethasone in this setting.26 The patient reported only mild (grade 1) nausea.
Case 6. R-CHOP Regimen in Stage IIA Diffuse Large B-Cell Lymphoma
A 25-year-old man was diagnosed with early-stage (stage IIA) DLBCL, with extranodal bulky disease infiltrating the mesentery and ileum, leading to gastrointestinal bleeding. His ECOG performance status score was 0, with no other comorbidities. The patient received standard therapy with 3 weekly dosing of the R-CHOP regimen over 6 cycles, as well as adjuvant radiotherapy for the bulky disease.
This patient’s main risk factors were his young age, the incidence of CINV in a previous treatment cycle, and a multiday chemotherapy regimen (Table 3). Of note, although the R-CHOP regimen contains doxorubicin and cyclophosphamide, unlike the AC combination in Case 1, it is not considered a classic HEC regimen because of the dose differences between R-CHOP and standard AC combinations. However, the results of a Japanese registry survey by Yoshida and colleagues indicated that despite 5-day administration of high-dose prednisone with the CHOP regimen, triple-drug antiemetic therapy may provide better CINV control than dual-drug antiemetic therapy in patients receiving CHOP-like regimens for hematologic malignancies.43
This finding by Yoshida and colleagues corroborated with our patient’s response to CINV prophylaxis with triple-drug antiemetic therapy as compared with dual-drug antiemetic therapy. For the initial chemotherapy cycle, the patient received prophylaxis with ondansetron and dexamethasone; his CINV was inadequately controlled, resulting in grade 2 nausea and vomiting for 3 to 4 days.
After the first cycle, the patient was switched to NEPA, with dexamethasone 8 mg added only on day 1, in accordance with the MASCC/ESMO guidelines for MEC regimens and NEPA prescribing information.6,26 CINV control clearly improved after the initiation of this combination. He had only mild (grade 1) nausea for 2 to 3 days.
The patient received no other steroids, because he was already receiving oral prednisone 100 mg (on days 2-5) as part of the R-CHOP regimen.
Discussion
Among these 6 cases of patients receiving MEC or HEC regimens, prophylaxis based on the combination of a 5-HT3 receptor antagonist plus an NK-1 receptor antagonist, the use of NEPA (plus additional dexamethasone as a precaution in 1 case) provided good protection against acute and delayed emesis. In these cases, when patients had nausea, it was limited to mild levels (grade 1).
Combination antiemetic regimens are accepted as the standard of care for CINV prophylaxis, but recommendations on individual components vary across guidelines, especially for MEC regimens. For example, the MASCC/ESMO and ASCO guidelines recommend a dual combination of a 5-HT3 receptor antagonist and dexamethasone, whereas the NCCN guidelines recommend a variety of options, ranging from dual- to triple-drug combination of antiemetic agents on day 1 (Table 1).5-7 The recommendations for CINV prophylaxis for HEC regimens are more standardized across guidelines. Triple- or quadruple-drug combinations of a 5-HT3 receptor antagonist, an NK-1 receptor antagonist, and dexamethasone, with or without olanzapine, are recommended for day 1 through day 4.5-7
NEPA, combined with dexamethasone, has been shown to provide good control of delayed and acute emesis.30-34 The use of long-acting palonosetron and netupitant provides added flexibility to adjust the dexamethasone dosing according to needs, by using lower doses or administering it on day 1 only.7,18,20
These patient cases illustrate various adjustments to dexamethasone plus NEPA, including limiting steroid exposure when necessary or desirable (Case 2 and Case 4), or appropriate steroid continuation and dosing to manage higher emetic risk (Case 3).
For high emetic risk, multiple-dose NEPA has been explored in patients with hematologic malignancies receiving multiday or high-dose chemotherapy, with and without dexamethasone39,40; a similar approach proved successful in Case 4.
Such strategies demand attention to the interplay of several factors, including the pharmacology of the chemotherapy and the antiemetic agents, as well as patient factors (eg, medical history, comorbidities, and concurrent medications). As integral members of multidisciplinary care teams, pharmacists can support tailored CINV management to maintain an acceptable quality of life for patients receiving chemotherapy.
Medication review by an oncology pharmacist provides opportunities to flag issues or advise on alternatives by weighing all relevant factors. This is confirmed by the findings of a recent survey that revealed the value of pharmacist interventions in reducing inappropriate prescriptions and polypharmacy among patients with cancer by identifying potential medication interactions or duplication of pharmacological effects.44
Among our cases, examples of consultation or intervention opportunities include proactive management of corticosteroid exposure in view of the patient’s comorbidities (as in Case 2, in which corticosteroids were contraindicated because of diabetes), and dexamethasone omission or dose reduction when NEPA is chosen for antiemetic prophylaxis (Case 5), because netupitant increases dexamethasone exposure.26
Certain multiday combination chemotherapies, such as the R-CHOP and R-GDP regimens for hematologic cancers, already contain corticosteroids. Case 5 and Case 6 illustrate the adjustment of dexamethasone in 2 such scenarios, complementing the guidance in the NEPA prescribing information, which contains no specific information on dexamethasone co-prescription beyond day 1 in corticosteroid-containing regimens.26
The marked improvement with optimized antiemesis after cycle 1 in Case 4 and Case 6 points to the benefits of optimizing antiemetic regimens upfront to prevent CINV in early cycles, rather than stepwise escalation of antiemetics after a CINV event. If multiple-dose NEPA may be considered to improve CINV control, a pharmacist could advise on suitable dosing intervals based on the pharmacokinetic profiles of netupitant and palonosetron.
Pharmacists could also help to assess the suitability of selected antiemetic agents or regimens based on other considerations, including cost, efficacy, and the potential for patient nonadherence.45 Beyond medication management, pharmacists are also well-positioned to advise about the selection of antiemetic medications based on the hospital formulary, and to counsel patients on the importance of adherence,46 a key determinant of successful CINV prevention.
The use of “smart” prescribing decision-support tools programmed to draw data, such as medical history, risk factors, and past prescriptions, from electronic medical records could guide physicians and pharmacists in designing suitable antiemetic regimens.47 Similarly, awareness of the evolving oncology treatment landscape and guideline updates can empower pharmacists to play active roles in optimizing CINV prophylaxis for patients.
These 6 cases show that the combination of NEPA and dexamethasone provides dosing convenience, ease of administration, and improved overall CINV control in multiday and multicycle chemotherapy, consistent with published studies.30,32,48,49
Conclusion
This case series highlights the possibility of applying dexamethasone-sparing strategies for emesis control in patients receiving MEC or HEC regimens. Furthermore, these cases show that some individualization of dexamethasone is required when used in combination with NEPA and in patients who require limited exposure to corticosteroids.
A multidimensional approach that considers guideline recommendations and patient characteristics, such as individual risk factors, comorbidities, and preferences, is necessary for effective prophylaxis of CINV.
Acknowledgment
Wei Yi Kwok and Geraldine Toh, of Tech Observer Asia Pacific, provided medical writing and editorial support for this article.
Funding Source
This study was funded by Mundipharma Singapore Holding Pte, Singapore, which is licensed by Helsinn, the manufacturer of Akynzeo, to market the drug in select countries outside of the United States.
Author Disclosure Statement
Dr Böhme has received honorarium from Mundipharma group of companies; Miss Yeoh is an employee of Mundipharma Singapore Holding Pte.
References
- Sommariva S, Pongiglione B, Tarricone R. Impact of chemotherapy-induced nausea and vomiting on health-related quality of life and resource utilization: a systematic review. Crit Rev Oncol Hematol. 2016;99:13-36.
- Inoue M, Shoji M, Shindo N, et al. Cohort study of consistency between the compliance with guidelines for chemotherapy-induced nausea and vomiting and patient outcome. BMC Pharmacol Toxicol. 2015;16:5.
- Rao KV, Faso A. Chemotherapy-induced nausea and vomiting: optimizing prevention and management. Am Health Drug Benefits. 2012;5(4):232-240.
- Hesketh PJ, Kris MG, Grunberg SM, et al. Proposal for classifying the acute emetogenicity of cancer chemotherapy. J Clin Oncol. 1997;15:103-109.
- Hesketh PJ, Kris MG, Basch E, et al. Antiemetics: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2017;35:3240-3261. Erratum in: J Clin Oncol. 2018;36:1459.
- Molassiotis A, Aapro M, Herrstedt J, et al. MASCC/ESMO antiemetic guidelines: introduction to the 2016 guideline update. Support Care Cancer. 2017;25:267-269.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Antiemesis. Version 2.2020. April 23, 2020. www.nccn.org/professionals/physician_gls/pdf/antiemesis.pdf. Accessed August 28, 2020.
- Dranitsaris G, Molassiotis A, Clemons M, et al. The development of a prediction tool to identify cancer patients at high risk for chemotherapy-induced nausea and vomiting. Ann Oncol. 2017;28:1260-1267.
- Van Ryckeghem F. Corticosteroids, the oldest agent in the prevention of chemotherapy-induced nausea and vomiting: what about the guidelines? J Transl Int Med. 2016;4:46-51.
- Gralla R, Lichinitser M, Van der Vegt S, et al. Palonosetron improves prevention of chemotherapy-induced nausea and vomiting following moderately emetogenic chemotherapy: results of a double-blind randomized phase III trial comparing single doses of palonosetron with ondansetron. Ann Oncol. 2003;14:1570-1577.
- Aapro MS, Grunberg SM, Manikhas GM, et al. A phase III, double-blind, randomized trial of palonosetron compared with ondansetron in preventing chemotherapy-induced nausea and vomiting following highly emetogenic chemotherapy. Ann Oncol. 2006;17:1441-1449.
- Saito M, Aogi K, Sekine I, et al. Palonosetron plus dexamethasone versus granisetron plus dexamethasone for prevention of nausea and vomiting during chemotherapy: a double-blind, double-dummy, randomised, comparative phase III trial. Lancet Oncol. 2009;10:115-124. Erratum in: Lancet Oncol. 2010;11:226.
- Grunberg SM. Antiemetic activity of corticosteroids in patients receiving cancer chemotherapy: dosing, efficacy, and tolerability analysis. Ann Oncol. 2007;18:233-240.
- Ioannidis JPA, Hesketh PJ, Lau J. Contribution of dexamethasone to control of chemotherapy-induced nausea and vomiting: a meta-analysis of randomized evidence. J Clin Oncol. 2000;18:3409-3422.
- Navari RM, Aapro M. Antiemetic prophylaxis for chemotherapy-induced nausea and vomiting. N Engl J Med. 2016;374:1356-1367.
- Aapro MS, Alberts DS. High-dose dexamethasone for prevention of cis-platin-induced vomiting. Cancer Chemother Pharmacol. 1981;7:11-14.
- Münstedt K, Müller H, Blauth-Eckmeyer E, et al. Role of dexamethasone dosage in combination with 5-HT3 antagonists for prophylaxis of acute chemotherapy-induced nausea and vomiting. Br J Cancer. 1999;79:637-639.
- Celio L, Frustaci S, Denaro A, et al; for the Italian Trials in Medical Oncology Group. Palonosetron in combination with 1-day versus 3-day dexamethasone for prevention of nausea and vomiting following moderately emetogenic chemotherapy: a randomized, multicenter, phase III trial. Support Care Cancer. 2011;19:1217-1225.
- Damian S, Celio L, De Benedictis E, et al. Is a dexamethasone-sparing strategy capable of preventing acute and delayed emesis caused by combined doxorubicin and paclitaxel for breast cancer? Analysis of a phase II trial. Oncology. 2013;84:371-377.
- Aapro M, Fabi A, Nolè F, et al. Double-blind, randomised, controlled study of the efficacy and tolerability of palonosetron plus dexamethasone for 1 day with or without dexamethasone on days 2 and 3 in the prevention of nausea and vomiting induced by moderately emetogenic chemotherapy. Ann Oncol. 2010;21:1083-1088.
- Ito Y, Tsuda T, Minatogawa H, et al. Placebo-controlled, double-blinded phase III study comparing dexamethasone on day 1 with dexamethasone on days 1 to 3 with combined neurokinin-1 receptor antagonist and palonosetron in high-emetogenic chemotherapy. J Clin Oncol. 2018;36:1000-1006.
- Komatsu Y, Okita K, Yuki S, et al. Open-label, randomized, comparative, phase III study on effects of reducing steroid use in combination with palonosetron. Cancer Sci. 2015;106:891-895.
- Celio L, Bonizzoni E, Zattarin E, et al. Impact of dexamethasone-sparing regimens on delayed nausea caused by moderately or highly emetogenic chemotherapy: a meta-analysis of randomised evidence. BMC Cancer. 2019;19:1268.
- Gu YL, Xie JM, Ren J, et al. Dexamethasone-sparing regimen is an effective and safe alternative in overall antiemetic protection: a systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e17364.
- European Medicines Agency. Akynzeo: netupitant/palonosetron hard capsules. European public assessment report-product information. Updated June 29, 2020. www.ema.europa.eu/en/documents/product-information/akynzeo-epar-product-information_en.pdf. Accessed September 10, 2020.
- Akynzeo (netupitant and palonosetron) capsules, for oral use [prescribing information]. Iselin, NJ: Helsinn Therapeutics; June 2020.
- Blower P, Aapro M. Granisetron vs ondansetron: is it a question of duration of 5-HT3 receptor blockade? Br J Cancer. 2002;86:1662-1663; author reply 1664.
- Janicki PK. Management of acute and delayed chemotherapy-induced nausea and vomiting: role of netupitant–palonosetron combination. Ther Clin Risk Manag. 2016;12:693-699.
- Gilmore J, Bernareggi A. Complementary pharmacokinetic profiles of netupitant and palonosetron support the rationale for their oral fixed combination for the prevention of chemotherapy-induced nausea and vomiting. J Clin Pharmacol. 2019;59:472-487.
- Aapro M, Karthaus M, Schwartzberg L, et al. NEPA, a fixed oral combination of netupitant and palonosetron, improves control of chemotherapy-induced nausea and vomiting (CINV) over multiple cycles of chemotherapy: results of a randomized, double-blind, phase 3 trial versus oral palonosetron. Support Care Cancer. 2017;25:1127-1135.
- Gralla RJ, Bosnjak SM, Hontsa A, et al. A phase III study evaluating the safety and efficacy of NEPA, a fixed-dose combination of netupitant and palonosetron, for prevention of chemotherapy-induced nausea and vomiting over repeated cycles of chemotherapy. Ann Oncol. 2014;25:1333-1339.
- Rugo HS, Rossi G, Rizzi G, Aapro M. Efficacy of NEPA (netupitant/palonosetron) across multiple cycles of chemotherapy in breast cancer patients: a subanalysis from two phase III trials. Breast. 2017;33:76-82.
- Yeo W, Lau TKH, Kwok CCH, et al. NEPA efficacy and tolerability during (neo)adjuvant breast cancer chemotherapy with cyclophosphamide and doxorubicin. BMJ Support Palliat Care. 2020;0:1-7. doi.org/10.1136/bmjspcare-2019-002037.
- Zhang L, Lu S, Feng J, et al. A randomized phase III study evaluating the efficacy of single-dose NEPA, a fixed antiemetic combination of netupitant and palonosetron, versus an aprepitant regimen for prevention of chemotherapy-induced nausea and vomiting (CINV) in patients receiving highly emetogenic chemotherapy (HEC). Ann Oncol. 2018;29:452-458.
- Molassiotis A, Lee PH, Burke TA, et al. Anticipatory nausea, risk factors, and its impact on chemotherapy-induced nausea and vomiting: results from the Pan European Emesis Registry study. J Pain Symptom Manage. 2016;51:987-993.
- Caputo R, Cazzaniga ME, Sbrana A, et al. Netupitant/palonosetron (NEPA) and dexamethasone for prevention of emesis in breast cancer patients receiving adjuvant anthracycline plus cyclophosphamide: a multi-cycle, phase II study. BMC Cancer. 2020;20:232.
- Bošnjak SM, Stamatovic L, Borroni ME, et al. Efficacy and safety of oral NEPA (netupitant/palonosetron), the first fixed-combination antiemetic, in patients with gynecological cancers receiving platinum-based chemotherapy. Int J Gynecol Cancer. 2018;28:1153-1161.
- Geer EB, Islam J, Buettner C. Mechanisms of glucocorticoid-induced insulin resistance: focus on adipose tissue function and lipid metabolism. Endocrinol Metab Clin North Am. 2014;43:75-102.
- Bubalo J, Chen AI, Leong K, et al. Phase II clinical trial of NEPA (netupitant/palonosetron) for prevention of chemotherapy induced nausea and vomiting (CINV) in patient receiving the BEAM conditioning regimen. Biol Blood Marrow Transplant. 2019;25(3 suppl):Abstract 414.
- Di Renzo N, Musso M, Scimè R, et al. Efficacy and safety of multiple doses of NEPA without dexamethasone in preventing nausea and vomiting induced by multiple-day and high-dose chemotherapy in patients with non-Hodgkin’s lymphoma undergoing autologous hematopoietic stem cell transplantation. Bone Marrow Transplant. 2020 Apr 28. Epub ahead of print.
- Chandana SR, Movva S, Arora M, Singh T. Primary brain tumors in adults. Am Fam Physician. 2008;77:1423-1430.
- Comelli I, Lippi G, Campana V, et al. Clinical presentation and epidemiology of brain tumors firstly diagnosed in adults in the emergency department: a 10-year, single center retrospective study. Ann Transl Med. 2017;5:269.
- Yoshida I, Tamura K, Miyamoto T, et al. Prophylactic antiemetics for haematological malignancies: prospective nationwide survey subset analysis in Japan. In Vivo. 2019;33:1355-1362.
- Uchida M, Suzuki S, Sugawara H, et al. A nationwide survey of hospital pharmacist interventions to improve polypharmacy for patients with cancer in palliative care in Japan. J Pharm Health Care Sci. 2019;5:14.
- Barbour SY. Caring for the treatment-experienced breast cancer patient: the pharmacist’s role. Am J Health Syst Pharm. 2008;65(suppl 3):S16-S22.
- Al-Quteimat O, Tollison J, Siddiqui MA. Olanzapine for prevention or management of chemotherapy-induced nausea and vomiting: a promising option. J Hematol Oncol Pharm. 2019;9:9-15.
- Rahimi R, Moghaddasi H, Rafsanjani KA, et al. Effects of chemotherapy prescription clinical decision-support systems on the chemotherapy process: a systematic review. Int J Med Inform. 2019;122:20-26.
- Aapro M, Rugo H, Rossi G, et al. A randomized phase III study evaluating the efficacy and safety of NEPA, a fixed-dose combination of netupitant and palonosetron, for prevention of chemotherapy-induced nausea and vomiting following moderately emetogenic chemotherapy. Ann Oncol. 2014;25:1328-1333.
- Hesketh PJ, Aapro M, Jordan K, et al. A review of NEPA, a novel fixed antiemetic combination with the potential for enhancing guideline adherence and improving control of chemotherapy-induced nausea and vomiting. Biomed Res Int. 2015;2015:651879.