In 2019, approximately 174,650 new cases of prostate cancer were reported, representing 9.9% of all new cancers in the United States. Prostate cancer currently ranks as the fifth leading cause of death from cancer, comprising 5.5% of all cancer deaths.1 Castration-resistant prostate cancer (CRPC) occurs when prostate cancer continues to progress despite maintaining castrate testosterone concentrations through chemical or surgical castration.2 Patients with CRPC may have distant metastatic disease, or they may have no distant metastases but have increasing levels of prostate-specific antigen (PSA), despite having castrate testosterone levels (ie, nonmetastatic CRPC).2
CRPC results from different mechanisms, including amplified expression of androgen receptors.3 Previously, the National Comprehensive Cancer Network (NCCN) guidelines recommended observation or the addition of hormonal therapy for patients with nonmetastatic CRPC.2 However, this recommendation was updated after the approval of 2 agents, enzalutamide and apalutamide, with a new indication for nonmetastatic CRPC.2,4,5 For metastatic CRPC, the NCCN recommends enzalutamide as a Category 1 first-line therapy, along with other agents, including docetaxel and abiraterone plus prednisone. Enzalutamide has also been demonstrated to extend survival for patients with metastatic CRPC who had disease progression while receiving docetaxel.2,4,6
Enzalutamide is a potent androgen receptor inhibitor that competitively inhibits androgen binding to its receptor, subsequently hindering androgen-receptor nuclear translocation and DNA binding.4 This second-generation antiandrogen agent demonstrated greater affinity to androgen receptors in comparison with first-generation antiandrogens, such as bicalutamide.3
The PROSPER study was an international, randomized phase 3 clinical trial that evaluated enzalutamide versus placebo in more than 1400 men with nonmetastatic CRPC.7 The study showed significantly prolonged metastasis-free survival for the patients who received enzalutamide compared with patients in the placebo group—36.6 months versus 14.7 months, respectively. In addition, patients in the enzalutamide group demonstrated median time to PSA progression of 37.2 months compared with only 3.9 months with placebo.7 This significant extension of the metastasis-free period using enzalutamide upfront in the nonmetastatic setting helps to delay future complications that are known to arise in patients who present with metastatic disease.
The use of enzalutamide has not been evaluated in patients with renal dysfunction and a creatinine clearance (CrCl) of less than 30 mL/min at baseline.4 Previously, 2 cases have been reported about the use of enzalutamide for patients undergoing hemodialysis, and neither of the cases demonstrated serious adverse effects.8,9
Our case represents a novel use of enzalutamide for a patient with severe renal dysfunction who was not undergoing hemodialysis. This case report demonstrates the efficacy and safety of enzalutamide therapy in a patient with severely impaired renal function.
Case Report
This case report describes a patient diagnosed with prostate cancer who started enzalutamide therapy on September 6, 2018. We followed the patient until December 1, 2018, and the patient continued to receive enzalutamide even after the observed study period.
This patient, a 74-year-old Hispanic man, had a medical history of stage 4 chronic kidney disease, with a diagnosis of nonmetastatic CRPC, and with a PSA doubling time of 1 month. The patient had a baseline serum creatinine of 2.5 mg/dL and a calculated CrCl of 28 mL/min, using the Cockcroft-Gault formula. Consistent with the current NCCN guidelines,2 the decision was to manage the patient with the use of enzalutamide.
Because of the lack of robust evidence of the safety of enzalutamide use in patients with renal impairment, a conservative dose of enzalutamide 80 mg was chosen to initiate the treatment, with an intent to increase to a full dose of enzalutamide 160 mg as tolerated by the patient.
The primary efficacy was measured by the patient’s PSA levels. We measured primary safety outcomes by screening for signs of drug interactions and patient-reported therapy-related adverse effects. Signs of drug interactions were closely monitored, because of the unknown clearance of enzalutamide in patients with chronic kidney disease, resulting in delayed clearance. In addition, blood pressure and peripheral edema were measured closely, because these are known adverse effects of enzalutamide.4
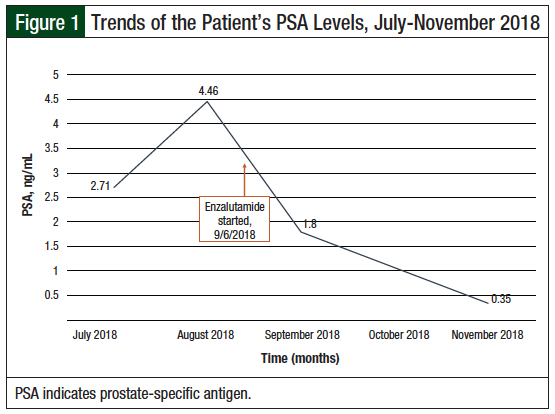
Before the initiation of enzalutamide in September 2018, the patient’s castration levels of testosterone were confirmed (<50 ng/dL), and the PSA was 4.46 ng/mL (Figure 1). The patient continued to receive androgen-deprivation therapy with leuprolide throughout the observed period.
Enzalutamide 80-mg therapy was initiated, and 1 month later, in October 2018, we observed a significant drop in PSA, to 1.8 ng/mL. Further evaluation of the PSA in November 2018 showed an even further decline in PSA level, to 0.35 ng/mL, with the patient maintaining enzalutamide dosing at 80 mg daily (Figure 1).
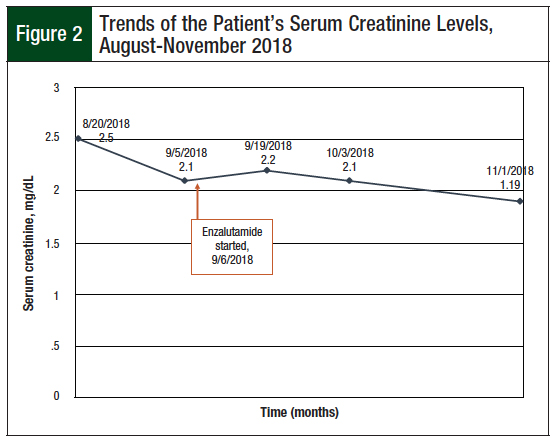
The patient’s renal function remained relatively constant over the entirety of his treatment. His serum creatinine levels slightly improved, to a new baseline of approximately 2.1 mg/dL (Figure 2).
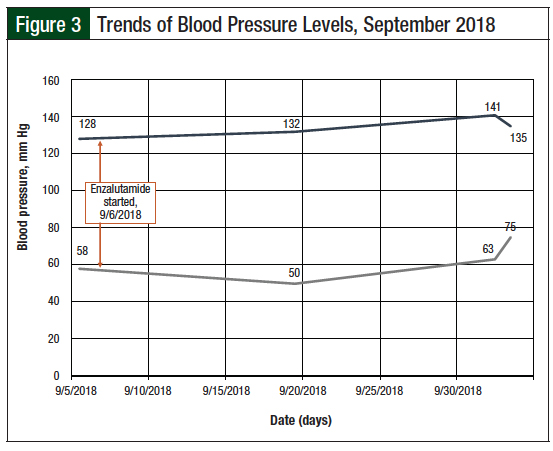
The only noted adverse event after initiation of treatment with enzalutamide was a mild increase in blood pressure, which is a known adverse effect of enzalutamide.4 The patient’s blood pressure before treatment initiation was 128/58 mm Hg, and in September 2018, during follow-up visits, it ranged from 132 to 141/50 to 75 mm Hg (Figure 3).
The patient was asymptomatic with these blood pressure elevations. With regard to other known adverse effects of enzalutamide,4 the patient denied having dry skin, pruritus, constipation, diarrhea, nausea/vomiting, decrease in appetite, arthralgia, fatigue, dizziness, headache, dyspnea, unusual bleeding or bruising, blurry vision, hot flashes, fever, and seizures. The patient had peripheral edema in his right foot before the start of enzalutamide treatment that improved slightly after the initiation of treatment.
Discussion
This case report shows the safety and efficacy of enzalutamide use in a patient with chronic kidney disease and prostate cancer. Despite only giving half of the dose approved by the US Food and Drug Administration for enzalutamide, the patient had close to a 12-fold improvement in his PSA values within 3 months. Aside from a mild elevation in blood pressure, which is a known adverse event in approximately 6% to 14% of patients receiving enzalutamide,4 the patient did not have any concerning adverse events that warranted discontinuation of the medication.
The efficacy and safety of enzalutamide have been evaluated in several clinical trials, including the AFFIRM, PREVAIL, and PROSPER studies.6,7,10 However, the drug’s effects in patients with severe renal dysfunction have not been formally studied before. A pharmacokinetics study identified 2 major metabolites of enzalutamide in human plasma—active metabolite N-desmethyl enzalutamide and inactive metabolite carboxylic acid derivative.11
Enzalutamide is hepatically metabolized, with cytochrome P2C8 primarily responsible for the formation of N-desmethyl enzalutamide.4,10 Renal excretion accounts for approximately 71% of the elimination, largely for the inactive metabolite, carboxylic acid derivative.4,11 Enzalutamide and its active metabolite represent an insignificant amount in urine. Fecal elimination accounts for approximately 14%.4,11
Based on these pharmacokinetic characteristics, the main concern of using enzalutamide in patients with severe renal dysfunction is the potential accumulation of the carboxylic acid derivative. Currently, this metabolite is considered to have no pharmacologic effects.11
The average half-life of enzalutamide after a single oral dose is 5.8 days, and steady state would be achieved by day 28.4 N-desmethyl enzalutamide demonstrated an average half-life of 7.8 to 8.6 days.4 Well after reaching steady state, the patient experienced no adverse effects or drug interactions related to enzalutamide. Based on this case, enzalutamide in patients with severe renal dysfunction (CrCl <30 mL/min) may be reasonable. However, a conservative dose titration and close monitoring are recommended to monitor efficacy and safety.
Conclusion
This case report demonstrates the efficacy and safety of the use of reduced-dose (80 mg daily) enzalutamide in a 75-year-old patient with severely impaired renal function and a diagnosis of CRPC. Future studies should examine the use of the full recommended dose of enzalutamide (160 mg daily) and the outcomes associated with the duration of use beyond 3 months.
Author Disclosure Statement
Dr Uhm and Dr Malamakal have no conflicts of interest to report.
References
- National Cancer Institute SEER Program. Cancer stat facts: prostate cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed September 15, 2020.
- National Comprehensive Cancer Network. Clinical Practice Guidelines in Oncology: Prostate Cancer. (NCCN Guidelines): Prostate Cancer. Version 2.2020. May 21, 2020. www.nccn.org/professionals/physician_gls/pdf/prostate.pdf. Accessed September 1, 2020.
- Tran C, Ouk S, Clegg NJ, et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009;324:787-790.
- Xtandi (enzalutamide) capsules/tablets, for oral use [prescribing information]. Northbrook, IL: Astellas Pharma; August 2020.
- Erleada (apalutamide) tablets [prescribing information]. Horsham, PA: Janssen Products, LP; 2020.
- Scher HI, Fizazi K, Saad F, et al; for the AFFIRM investigators. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187-1197.
- Hussain M, Fizazi K, Saad F, et al. Enzalutamide in men with nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2018;378:2465-2474.
- Tsang ES, de Haan M, Eigl BJ. A case report of enzalutamide administration in a dialysis-dependent patient with castration-resistant prostate cancer. J Oncol Pharm Pract. 2018;24:143-145.
- Simões J, Augusto I, Meireles S, et al. Metastatic castration-resistant prostate cancer and the challenge of a patient with chronic kidney disease in hemodialysis. Autops Case Rep. 2018;8:e2018011.
- Beer TM, Armstrong AJ, Rathkopf DE, et al; for the PREVAIL investigators. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424-433.
- Gibbons JA, Ouatas T, Krauwinkel W, et al. Clinical pharmacokinetic studies of enzalutamide. Clin Pharmacokinet. 2015;54:1043-1055.