In the United States, colorectal cancer (CRC) is the third most common cancer type and the second most common cause of cancer mortality.1,2 The lethal nature of CRC is attributable to being largely asymptomatic until advanced stages and the lack of curative treatment options for patients with advanced-stage disease.3 CRC screening in the average-risk population has clear, early detection and mortality benefits.1-3 Recently, not being up to date with screening for CRC was associated with an approximate 3-fold risk for CRC-related mortality.4
Although US public health officials have invested considerable resources in raising the awareness of CRC and encouraging Americans to participate in CRC screening, a population-based annual survey in 2012 of more than 200,000 Americans showed that only approximately 65% of US adults aged 50 to 75 years received the guideline-recommended testing for CRC.5 In an effort to close the screening gap, the National Colorectal Cancer Roundtable, a national coalition of public, private, and voluntary organizations, launched a campaign to increase CRC screening rates to 80%.6,7
In 2016, the US Preventive Services Task Force (USPSTF) updated its CRC screening recommendations to highlight the importance of improving the use of CRC screening, regardless of strategy.1 Several additional screening guidelines have been developed, including those from the National Comprehensive Cancer Network,8 which are similar to those from the USPSTF in recommending screening beginning at age 50, and the American Cancer Society, which moved the age to start screening for CRC to 45 years.9 The new US Food and Drug Administration (FDA)-approved options listed in the USPSTF guidelines include blood-based screening to detect circulating methylated Septin 9 (SEPT9) DNA (Epi proColon) and a multianalyte stool test combining fecal immunochemical testing (FIT) and fecal DNA (Cologuard; FIT/stool DNA).1
SEPT9 DNA (Epi proColon) is FDA approved for persons who are at an average risk for CRC and have chosen not to undergo screening by existing guideline-recommended methods. In a prospective study, an early version of the test had a sensitivity of approximately 48%.10 With further improvement, the commercially available FDA-approved version of SEPT9 DNA had a sensitivity of 68% to 72% (specificity, 80%) in an average-risk population and in direct comparison with FIT, and identified CRC more frequently at earlier stages than the original assay.1,11,12 The FIT/stool DNA (Cologuard) test was approved by the FDA for screening in 2014 for those at average risk, and has a reported sensitivity of approximately 92% (specificity, approximately 87%).13
New blood- and fecal-based CRC screening procedures will likely play an important role in increasing screening participation among the approximate 35% of screeningeligible Americans who are not receiving screening. For example, a study of patients who twice refused screening with traditional colonoscopy and take-home stool screening tests for CRC had a 99.5% participation rate for a blood-based screening test.14 Similarly, in a German study of patients who refused colonoscopy, 83% accepted the SEPT9 blood test and 15% selected a stool test, which in combination resulted in a 97% overall screening rate.15
Accordingly, the objective of this study was to quantify the clinical and fiscal impacts on health plans of expanding CRC screening participation from today’s level of 65% up to 80%. We focused on the 50- to 64-year-old screening-eligible population because of its relevance to commercial insurers and because this population has the largest screening gap.
Methods
We developed a simulation model in Microsoft Excel (version 15.1, Redmond, WA) to project the 3-year aggregate health outcomes and costs for CRC screening among average-risk persons who are eligible for screening according to the USPSTF recommendations (age 50-75 years)1 and have no evidence of previous screening. Table A1 in the Appendix provides all the model input values, uncertainty ranges, and data sources used in the study. Our objective was to model the population-level clinical and fiscal impacts of raising CRC screening participation from 65% to 80%. We compared a no-screening scenario and 3 common and commercially available noninvasive scenarios of SEPT9 blood-based screening, FIT fecalbased screening, and stool FIT/stool DNA testing. As is recommended for budget impact analyses, we projected the outcomes and costs for a 3-year planning period.16
Our approach synthesizes evidence from the validation studies for blood- and fecal-based CRC screening tests, as well as the US census and other sources in the peer-reviewed literature. The costs included those for CRC screening tests and procedures and associated primary care visits, as well as diagnostic workup (imaging and/or colonoscopy, pathology). We also estimated the CRC treatment cost and end-of-life care cost. The expenditure outcomes (total and per-enrollee per-month) were calculated from the payer perspective and are reported in 2018 US dollars.
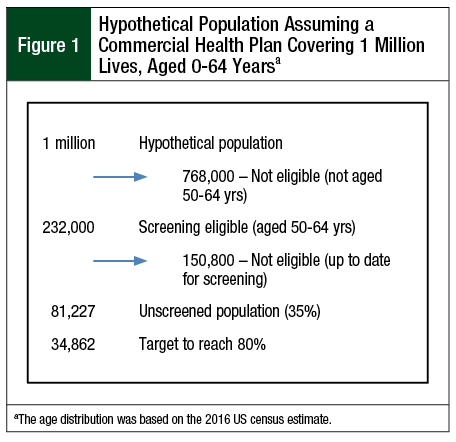
We modeled a hypothetical population of 1 million members of a commercial health plan aged 50 to 64 years. Age and sex distributions were based on a 2016 projection of the 2010 US Census.17 The distribution of CRC-screening eligible people is shown in Figure 1. We assumed that 81,227 (35%) of eligible but unscreened members were nonadherent, which was defined as patients who were otherwise eligible for CRC screening but who had not had screening in the relevant time window (ie, colonoscopy in past 10 years, FIT in past year, or FIT/stool DNA in the past 3 years).
For the unscreened members, the detection of CRC was estimated as the incidence rate, which was determined from the Surveillance, Epidemiology, and End Results (SEER) Program database. The stage distribution of CRC at diagnosis in the status quo scenario was based on a previous study of screening for CRC in nonadherent individuals.18,19
The health plan–wide screening approaches that were modeled include SEPT9 blood testing, FIT, and FIT/stool DNA. For all approaches, we estimated the cost of achieving 80% participation over a 3-year period. As outlined in Figure 1, achieving 80% screening (185,600 members) required an additional 34,800 members over the baseline adherence. We assumed that for each method, the screening rates increased incrementally by 5% of the total population (or approximately 16.5% of the unscreened population) each year for 3 years.
Based on previous population-wide studies, we assumed that the overall rate of positive screening tests was 20% for blood-based testing with SEPT9,11 approximately 7% with fecal-based testing with FIT,13 and approximately 14% with FIT/stool DNA testing.13 The proportion of positive screening tests resulting in a diagnosis of CRC was based on the average prevalence estimates for CRC (71 per 10,000 life-years) from 2 large studies (PRESEPT11 and DEEP-C,13 with 16,846 screening colonoscopies combined) of CRC screening outcomes (weighted by sample size)10,13 and test sensitivity values derived from the validation studies. The CRC sensitivity rates for SEPT9, FIT, and FIT/stool DNA screening were 72.2% (95% confidence interval [CI], 62.5%-80.1%),12 68% (95% CI, 58.2%-76.5%),12 and 92.3% (95% CI, 83%-97.5%),13 respectively.
The stage distribution of patients with CRC in the cohort was also estimated from the combined colonoscopy outcomes of the PRESEPT and DEEP-C studies. To estimate the stage distribution of cases of CRC detected for each screening method, the population prevalence estimates were multiplied by the reported stage-specific detection rates.12,13 The projected distributions for SEPT9 were 34.2% for stage I, 35.8% for stage II, 18.6% for stage III, and 11.4% for stage IV; for FIT, 36.7% for stage I, 34.7% for stage II, 21.7% for stage III, and 6.9% for stage IV; and for FIT/stool DNA, 44.1% for stage I, 32.8% for stage II, 15.6% for stage III, and 6.3% for stage IV. We used the SEER incidence rate to estimate the proportion of nonadherent patients and false-negative patients who were diagnosed with CRC after having symptoms. These symptom-detected individuals have a more advanced-stage distribution (18% for stage I, 34% for stage II, 23% for stage III, and 25% for stage IV).19
We used an average (weighted by sample size) of the prevalence estimates for advanced adenomas (863 per 10,000 life-years) and nonadvanced adenomas (3164 per 10,000 life-years) that were reported in 2 large studies of CRC screening outcomes as the foundation for the calculation of adenoma outcomes.10,13 For SEPT9, we estimated the sensitivity for advanced and nonadvanced adenomas based on a validation study12 to be 14.6% and 7.7%, respectively. Based on a large study, for FIT we estimated the sensitivity values for advanced and nonadvanced adenomas,20 which were 7.7% for each. For FIT/stool DNA, the estimates for advanced and nonadvanced adenomas were based on the performance evaluation study,13 and were 42.4% and 17.2%, respectively.
The cost associated with blood-based screening with SEPT9 (Current Procedural Terminology [CPT] code 81327, $192) was based on pricing from the Centers for Medicare & Medicaid Services (CMS). The cost of fecal-based CRC screening was based on 2018 CMS reimbursement for FIT (CPTcode 82274, $19.64). The cost of FIT/stool DNA testing was based on published CMS reimbursement (CPTcode 81528, $508.87). Our calculations assume that the full cost of the blood- and fecal-based screening tests for CRC will be covered by insurers (without cost-sharing) in accordance with the terms of the Affordable Care Act regarding the coverage of screening tests with USPSTF grade A ratings,21 and the CMS national coverage determination for CRC screening.1
In the base case, we assumed that 65% of patients with a positive blood- or fecal-based screening test for CRC would advance to receive a diagnostic colonoscopy, and that those with a finding of CRC, advanced adenoma, or nonadvanced adenoma would also have a pathology follow-up. We estimated the costs for diagnostic colonoscopy to be $1500 (CPTcode 45378) and for the associated pathology to be $77 (CPTcode 88305).22
We applied the 1-year stage-specific CRC care costs from a previous analysis of disease-attributable cost using the SEER-Medicare–linked database to the fraction with a diagnosis of CRC during the 1-year model time horizon.23,24
Sensitivity Analyses
We evaluated the outcome uncertainty using one-way sensitivity analysis.25 To do so, low- and high-value estimates for each model parameter were propagated through the model framework, and the resulting ranges of incremental early-stage (stage I-II) CRC diagnoses and expenditures (vs the status quo scenario) were recorded. We focused on the incremental outcome of early-stage CRC detection because that is the primary clinical benefit of CRC screening programs (vs detection at advanced stage). The results are presented in the form of tornado diagrams (Appendix Figures A1-A3).
Results
From the population of 1 million commercial health plan members, an estimated 232,000 members are screening eligible and an estimated 150,800 (65%) are up to date for screening (Figure 1). We assessed the screening of an additional 34,862 eligible members over 3 years to reach the goal of 80% (185,662) screening adherence in the population.
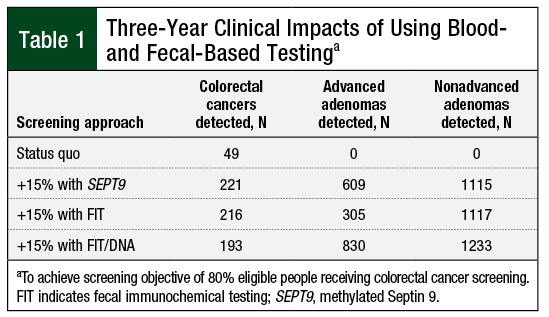
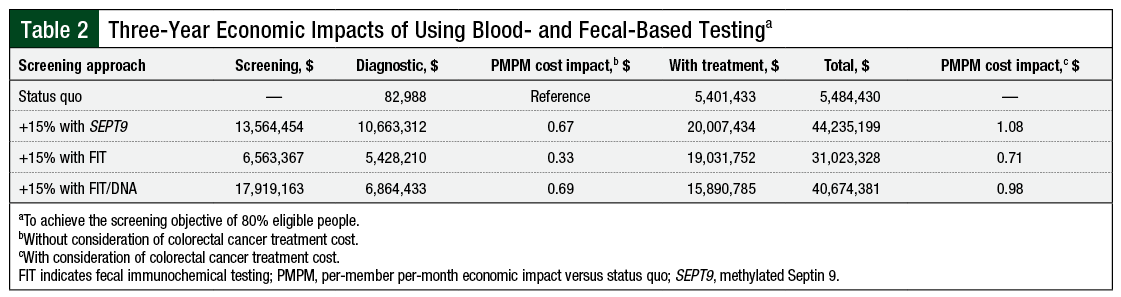
Using SEPT9 testing, screening an additional 15% of members resulted in 61 cases of CRC detected over 1 year and 221 cases detected over 3 years (Table 1). For advanced adenomas, 120 and 609 cases were detected in 1- and 3-year projections, respectively. For nonadvanced adenomas or polyps, 402 and 2048 cases, respectively, were detected in 1- and 3-year projections. The estimated per-member per-month (PMPM) cost impact (vs no screening) for the 1- and 3-year projections were $0.40 and $0.67, respectively (Table 2); including the treatment costs for CRC, these values were $0.72 and $1.08, respectively.
Using FIT testing, screening an additional 15% of members resulted in 59 cancers detected in the 1-year projection and 216 cases in the 3-year projection (Table 1). For advanced adenomas, 58 and 305 cases were detected in the 1- and 3-year projections, respectively. For nonadvanced adenomas, 212 and 1117 cases were detected in the 1- and 3-year projections, respectively. The estimated PMPM cost impact (vs no screening) for the 1- and 3-year projections were $0.19 and $0.33, respectively (Table 2); including treatment costs, these values were $0.48 and $0.71, respectively.
Using FIT/stool DNA testing, screening an additional 15% of members resulted in an additional 74 cancers detected in the 1-year simulation and 193 cancers detected in the 3-year simulation (Table 1). For advanced adenomas, 319 and 830 were detected in the 1- and 3-year projections, respectively. For nonadvanced adenomas, 474 and 1233 were detected in the 1- and 3-year projections, respectively. The estimated PMPM cost impact (vs no screening) for the 1- and 3-year projections were $0.79 and $0.69, respectively (Table 2); including treatment costs, these values were $1.14 and $0.98, respectively.
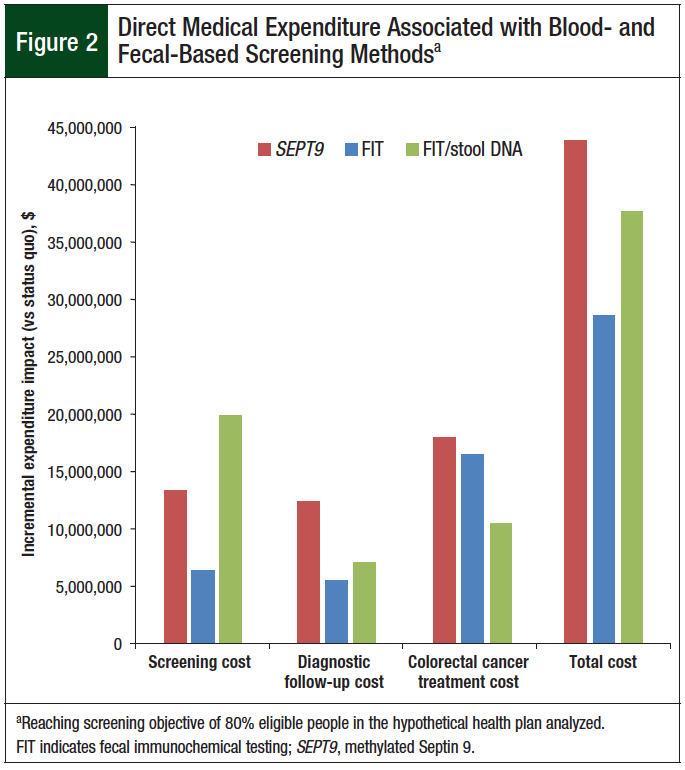
As illustrated in Figure 2, the costs for screening, diagnosis, and treatments varied for each screening strategy. For SEPT9 and FIT, the screening and diagnostic costs were relatively similar. For stool DNA, the screening costs were higher than diagnostic costs and were similar to the treatment costs.
In one-way sensitivity analyses, the most significant parameters that influenced the total expenditures were the cost of diagnostic colonoscopy, the proportion adherent to diagnostic colonoscopy after a positive screening SEPT9 or FIT test, and the sensitivity of SEPT9 and FIT in the additional use of the SEPT9 scenario. The complete results of the one-way sensitivity analysis are presented in Appendix Figure A1 and Appendix Figure A2.
Discussion
Increasing adherence to screening is critical to improving CRC outcomes in the United States. A key component of any strategy to increase adherence involves recruiting individuals who have been nonadherent to screening over time. Accordingly, we used simulation modeling informed with data from the literature and nationally representative databases to evaluate the clinical and fiscal impacts of using noninvasive testing to improve CRC screening participation among eligible commercial health plan members who have not been screened.
Assuming that all noninvasive strategies can be applied to improve CRC screening adherence from 65% to 80% participation over 3 years, this analysis estimates the health plan cost and outcomes of each strategy. SEPT9 and FIT achieved similar 3-year outcomes in terms of the number of CRC cases detected (221 and 216 cases, respectively); FIT/stool DNA detected slightly fewer cases (193 cases). Including the costs of screening, diagnostic testing, and treatment, the PMPM costs were highest for SEPT9 ($1.08), followed by FIT/stool DNA ($0.98) and FIT ($0.71), suggesting a modest tradeoff between cost and the number of cases detected among the screening strategies. Considering only the costs of screening and diagnosis, the costs for SEPT9 and FIT/stool DNA were almost the same ($0.67 and $0.69, respectively) and the cost for FIT was lower ($0.33).
FIT/stool DNA and SEPT9 identified substantially higher numbers of persons with advanced adenomas (830 and 609 patients, respectively) than FIT testing (305 patients). The increase in lesion detection by SEPT9 was a result of increased test positivity, which in turn led to higher rates of colonoscopy. The removal of advanced adenomas (often precursors to CRC) ultimately lowers the incidence of CRC and improves patient outcomes. FIT was better at avoiding the detection of nonadvanced adenomas. For the majority of these lesions, the potential for the transformation to CRC is very low, thus the costs and potential harms related to removing the lesions should be minimized.
Despite many years of effort, CRC screening rates have plateaued well below the national goal of 80% for the United States, with a considerable segment of the population remaining unscreened. The results of a recent large survey study on failure to screen in the Northern and Southern California Kaiser Permanente health systems clearly illustrate the clinical importance of CRC screening.4 In this setting, 75.9% of CRC-related mortality occurred in patients who were not up to date with screening, and the failure to screen was therefore associated with a nearly 3-fold increased risk for CRC-related death. Doubeni and colleagues concluded that further improving access to screening and the completion of follow-up diagnostics remain important areas of focus for decreasing CRC-related deaths.4
For unscreened individuals who have not received colonoscopy, which is the dominant mode of screening, a noninvasive test may be the best option to reduce the burden of CRC. With rapidly increasing care costs, early detection via increased uptake becomes even more vital. Health insurance plans that are considering how to renew their efforts to improve screening rates among their most reluctant members must weigh many factors, including the costs related to testing, adherence over time, the sensitivity and specificity of testing, and the rates of diagnostic evaluation among patients who test positive on screening. This latter issue is critical, particularly in the population that remains unscreened over time.
A key to improving screening participation is patient acceptance of the testing method. Therefore, patient preferences should be considered in health plan decisions about promoting the use of blood- and fecal-based testing approaches to expand CRC screening participation. For example, in a 2016 US study, only 36.9% of 2000 participants completed the mailed CRC screening test, despite a cash incentive to return the test.26
Blood-based screening may have an advantage in that individuals are more willing to have a blood test than take a stool sample at home.14 A randomized trial of blood-based versus fecal-based screening for CRC showed that a significantly greater number of unscreened participants completed blood-based testing than fecal-based testing (99.5% vs 88.1%, respectively).14 Blood tests can also be completed easily as part of a standard medical office appointment for a wellness check or to take advantage of opportunities to promote screening during scheduled visits to manage chronic illnesses.
Outside of an investigational setting, future studies should characterize and compare patient preferences for different CRC screening methods and evaluate the clinical and fiscal implications of differential participation rates. There is particular need for communitybased evaluations of the uptake, acceptance, performance, and cost of blood-based screening relative to other screening methods.
Limitations
This analysis has several limitations. First, although our estimates of screening cost account for programmatic costs associated with implementing or maintaining a screening program, reliable estimates of these costs are not readily available, particularly for individuals who have been resistant to getting any type of screening, although they may be substantial and ongoing.27 For example, patients who are nonadherent to screening may require multiple reminders before electing to receive screening, and these resource needs could carry on indefinitely.
Second, the overall budget impact estimates are highly dependent on follow-up colonoscopy costs. We applied a cost of $1500, but we recognize that reimbursements vary widely across health plans in the United States.
Third, a key factor to the success of any noninvasive screening test is adherence to diagnostic follow-up among patients with a positive test. A retrospective study showed that the rate of diagnostic follow-up for positive noninvasive tests can be poor, particularly in minority and underserved populations.28 It is not known whether there would be differential follow-up of positive blood-based versus stool-based testing.
Finally, we based the costs of treatment for CRC on a published analysis that used the Truven Health MarketScan Research Databases.24 The cost of treatment for CRC related to payers not included in these databases (eg, Medicare, Medicaid) may differ in community practice.
Conclusion
In the United States, 35% of persons who are eligible for CRC screening remain unscreened, in many cases because of aversion to colonoscopy. Now, with the availability of a blood-based screening test for CRC, from a health plan perspective, it may be feasible to achieve population screening goals that have been set by national quality-setting groups, such as the USPSTF.
For patients who are unwilling to screen with colonoscopy, noninvasive blood- and stool-based methods can shift CRC detection to earlier and more curable stages of illness with similar cost. Therefore, screening promotion should consider the option that maximizes uptake, given the particular constraints and needs of local and regional care delivery. Given the similarity in clinical performance and budget impact between FIT/stool DNA and SEPT9, promoting blood-based testing may be an attractive option for health insurers in select settings. Our findings may help healthcare stakeholders to assess the clinical and fiscal tradeoffs of alternative approaches that may help to increase CRC screening participation in the United States.
Funding Source
Funding for this study was provided by Epigenomics.
Author Disclosure Statement
Dr Roth and Dr Ramsey are consultants to, and Dr deVos is an employee of, Epigenomics.
References
- US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2016;315:2564-2575. Errata in: JAMA. 2016;316:545; JAMA. 2017;317:2239.
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5-29.
- American Cancer Society. Colorectal cancer facts & figures 2014-2016. Atlanta, GA; 2014.
- Doubeni CA, Fedewa SA, Levin TR, et al. Modifiable failures in the colorectal cancer screening process and their association with risk of death. Gastroenterology. 2019;156:63-74.e6.
- Centers for Disease Control and Prevention. Vital signs: colorectal cancer screening test use—United States, 2012. MMWR Morb Mortal Wkly Rep. 2013;62:881-888.
- American Cancer Society. ColonMD: Clinicians’ Information Source. www.cancer.org/health-care-professionals/colon-md.html#NCCRT. Accessed May 20, 2019.
- Meester RGS, Doubeni CA, Zauber AG, et al. Public health impact of achieving 80% colorectal cancer screening rates in the United States by 2018. Cancer. 2015;121:2281-2285.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Colorectal Cancer Screening. Version 1.2018. March 26, 2018. www.nccn.org/professionals/physician_gls/pdf/colorectal_screening.pdf. Accessed May 20, 2019.
- Wolf AMD, Fontham ETH, Church TR, et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 2018;68:250-281.olf AMD, Fontham ETH, Church TR, et al. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 2018;68:250-281.
- Church TR, Wandell M, Lofton-Day C, et al; for the PRESEPT clinical study steering committee, investigators and study team. Prospective evaluation of methylated SEPT9 in plasma for detection of asymptomatic colorectal cancer. Gut. 2014;63:317-325.
- Potter NT, Hurban P, White MN, et al. Validation of a real-time PCR–based qualitative assay for the detection of methylated SEPT9 DNA in human plasma. Clin Chem. 2014;60:1183-1191.
- Johnson DA, Barclay RL, Mergener K, et al. Plasma Septin9 versus fecal immunochemical testing for colorectal cancer screening: a prospective multicenter study. PLoS One. 2014;9:e98238.
- Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening.N Engl J Med. 2014;370:1287-1297.
- Liles EG, Coronado GD, Perrin N, et al. Uptake of a colorectal cancer screening blood test is higher than of a fecal test offered in clinic: a randomized trial. Cancer Treat Res Commun. 2017;10:27-31.
- Adler A, Geiger S, Keil A, et al. Improving compliance to colorectal cancer screening using blood and stool based tests in patients refusing screening colonoscopy in Germany. BMC Gastroenterol. 2014;14:183.
- Sullivan SD, Mauskopf JA, Augustovski F, et al. Budget impact analysis—principles of good practice: report of the ISPOR 2012 Budget Impact Analysis Good Practice II Task Force. Value Health. 2014;17:5-14.
- Howden LM, Meyer JA. Age and sex composition: 2010. 2010 Census briefs. May 2011. www.census.gov/prod/cen2010/briefs/c2010br-03.pdf. Accessed June 21, 2016.
- Cress RD, Morris C, Ellison GL, Goodman MT. Secular changes in colorectal cancer incidence by subsite, stage at diagnosis, and race/ethnicity, 1992–2001. Cancer. 2006;107(5 suppl):1142-1152.
- Knudsen AB, Zauber AG, Rutter CM, et al. Estimation of benefits, burden, and harms of colorectal cancer screening strategies: modeling study for the US Preventive Services Task Force. JAMA. 2016;315:2595-2609.
- Jensen CD, Corley DA, Quinn VP, et al. Fecal immunochemical test program performance over 4 rounds of annual screening: a retrospective cohort study. Ann Intern Med. 2016;164:456-463.
- HealthCare.gov. Preventive care benefits for adults. www.healthcare.gov/preventive-care-adults/. Accessed June 21, 2016.
- Ladabaum U, Levin Z, Mannalithara A, et al. Colorectal testing utilization and payments in a large cohort of commercially insured US adults. Am J Gastroenterol. 2014;109:1513-1525.
- Yabroff KR, Lamont EB, Mariotto A, et al. Cost of care for elderly cancer patients in the United States. J Natl Cancer Inst. 2008;100:630-641.
- Fitch K, Pyenson B, Blumen H, et al. The value of colonoscopic colorectal cancer screening of adults aged 50 to 64 years. Am J Manag Care. 2015;21:e430-e438.
- Briggs A, Claxton K, Sculpher M. Decision Modelling for Health Economic Evaluation. New York, NY: Oxford University Press; 2006.
- Gupta S, Miller S, Koch M, et al. Financial incentives for promoting colorectal cancer screening: a randomized, comparative effectiveness trial. Am J Gastroenterol. 2016;111:1630-1636.
- Liss DT, French DD, Buchanan DR, et al. Outreach for annual colorectal cancer screening: a budget impact analysis for community health centers. Am J Prev Med. 2016;50:e54-e61.
- Issaka RB, Singh MH, Oshima SM, et al. Inadequate utilization of diagnostic colonoscopy following abnormal FIT results in an integrated safety-net system. Am J Gastroenterol. 2017;112:375-382.