Venous thromboembolism (VTE) is a major complication of hematologic malignancies and is associated with significant morbidity and mortality.1,2 Patients with multiple myeloma are especially at risk for VTEs based on the underlying disease pathophysiology and the treatments used for this patient population, specifically immunomodulatory drugs (IMiDs).1,2 The risk for VTE among patients ranges from 26% to 67% for those with newly diagnosed multiple myeloma,3,4 and from 11% to 15% for patients with relapsed or refractory multiple myeloma.5,6 The use of IMiDs further increases the risk for thromboembolic complications, particularly within the first 6 months of therapy, although this risk decreases over time.1,2
Currently, the 3 IMiDs that are indicated for the treatment of patients with multiple myeloma are thalidomide, lenalidomide, and pomalidomide. Lenalidomide was the most frequently used IMiD at our institution as a result of its tolerability and physician preference. Lenalidomide is a second-generation IMiD and is often used in the front-line setting for the treatment of patients with newly diagnosed multiple myeloma, usually in combination with other agents (ie, steroids, proteasome inhibitors). Although lenalidomide-based regimens are generally well-tolerated, there is a significant VTE risk associated with lenalidomide-based therapy.1,3,4
In a study by Rajkumar and colleagues, when used in combination with high-dose (≥480 mg monthly) dexamethasone, lenalidomide was associated with VTE rates of 26% compared with 12% in patients who received low-dose dexamethasone plus lenalidomide.3 High-dose dexamethasone is no longer recommended by the National Comprehensive Cancer Network (NCCN) guidelines.7 In the study by Rajkumar and colleagues, the use of high-dose dexamethasone did not result in superior time to progression or longer progression-free or overall survival compared with low-dose dexamethasone in patients with newly diagnosed multiple myeloma.3 In addition, treatment with low-dose dexamethasone had lower toxicity than high-dose dexamethasone when added to lenalidomide treatment.3
Given the increased risk for VTE among patients with newly diagnosed multiple myeloma who received IMiD- and steroid-based induction therapy, thromboprophylaxis is a necessity during treatment for multiple myeloma, especially in the front-line setting. The NCCN guidelines provide a risk-adapted approach for thromboprophylaxis in patients with multiple myeloma based on recommendations outlined by the International Myeloma Working Group.7,8
According to the NCCN guidelines, the risk factors for VTE include the use of certain medications (eg, erythropoiesis-stimulating agents; oral, transdermal patch, or vaginal ring contraceptives; hormone replacement therapy); diabetes; congestive heart failure; obesity (ie, body mass index ≥30 kg/m2); chronic kidney disease (ie, decreased glomerular filtration rate of <60 mL/min/1.73 m2 for ≥3 months); previous VTE; immobilization; recent major surgery; central venous access device or pacemaker placement; trauma; and blood clotting disorders.7,9
Furthermore, according to the NCCN guidelines, treatment with aspirin is recommended for patients with ≤1 individual patient–related or multiple myeloma–related risk factor.7 Prophylactic low-molecular-weight heparin (LMWH) or full-dose warfarin is recommended for patients with ≥2 risk factors and for all patients who are receiving an IMiD plus high-dose (≥480 mg monthly) dexamethasone, doxorubicin, or multiagent chemotherapy, independent of the presence of additional risk factors. The current guidelines suggest using caution in the routine use of direct oral anticoagulants in patients with cancer,7 although there is increasing evidence to support the use of direct oral anticoagulants as routine thromboembolic prophylaxis in patients with multiple myeloma.10,11 Direct oral anticoagulants are increasingly being used for the treatment of patients with multiple myeloma, because they are easy to administer and there is no need for routine monitoring of patients.10
Although different strategies for thromboprophylaxis have been effective in decreasing the risk for VTE, the comparative efficacy of different thromboprophylactic agents in patients with additional individual risk factors is not well-established. A randomized, open-label phase 3 study of 667 patients with newly diagnosed multiple myeloma reported that aspirin and fixed low-dose warfarin were as effective as LMWH in reducing the incidence of serious thrombotic events associated with thalidomide-based regimens.12 Another study reported that aspirin is an effective and safe alternative to LMWH in patients newly diagnosed with multiple myeloma who have a low individual risk for thrombosis and received lenalidomide and low-dose dexamethasone as induction therapy.1
Our study’s objectives were to assess clinicians’ adherence to thromboprophylaxis recommendations in the NCCN Clinical Practice Guidelines in Oncology: Cancer-Associated Venous Thromboembolic Disease in patients with newly diagnosed multiple myeloma receiving lenalidomide-based induction therapy, and to assess the incidence of VTE among different risk groups using various thromboprophylactic strategies.7
Methods
In this retrospective, single-center study at Yale New Haven Health, we evaluated patients with newly diagnosed multiple myeloma who received lenalidomide-based induction therapies. Patients who had a lenalidomide-based treatment plan between February 1, 2013, and May 31, 2017, were retrieved from our electronic medical records. Only patients who received lenalidomide-based induction therapy were selected for the study. Patients with active VTE before starting induction therapy, or those who did not have follow-up for at least 12 months after the initiation of therapy, were excluded from the study.
The primary end point was to confirm clinicians’ adherence to the NCCN cancer-associated venous thromboembolic disease guidelines.7 Adherence was determined based on the number of VTE risk factors and the thromboprophylactic agent the patients received, as outlined by the NCCN guidelines.7 The patients with ≤1 individual patient–related or multiple myeloma–related risk factor were classified as low risk, and patients with ≥2 risk factors were classified as high risk. Of note, patients with a diagnosis of multiple myeloma automatically had a risk factor score of 1.
The secondary end points included the incidence of patients with a first episode of deep-vein thrombosis (DVT); pulmonary embolism; arterial thrombosis; an acute cardiovascular event (eg, acute myocardial infarction or stroke); or sudden, otherwise unexplained death (presumably related to pulmonary embolism, acute myocardial infarction, or stroke based on physicians’ notes in the electronic medical record) within the first 12 months of starting induction therapy.
Other end points included the time to the first episode of VTE and the incidence of major or minor bleeding events. Major bleeding was defined as fatal bleeding, symptomatic bleeding in a crucial area or organ, or bleeding that caused a reduction in hemoglobin concentration of ≥2 g/dL or that necessitated a transfusion of ≥2 units of whole blood or red blood cells.13 The incidence of minor bleeding events comprised all bleeding events that did not meet the criteria for major bleeding.
The data points that were collected included age, sex, Karnofsky Performance Status score, chemotherapy regimen, cytogenetic risk per the Mayo Algorithmic Approach for Stratification of Myeloma and Risk-Adapted Therapy Report criteria, standard prognostic parameters (ie, serum levels of creatinine, β2-microglobulin, and serum albumin), stage of disease (according to the Revised Multiple Myeloma International Staging System), comorbidities (eg, diabetes, cardiovascular disease, chronic kidney disease), previous thromboembolism, immobilization, recent surgery, recent trauma, blood clot disorders, and the concomitant use of recombinant human erythropoietin, oral and nonoral contraceptives, or hormone replacement therapy (ie, estrogen, progesterone, testosterone). Descriptive statistics were used to analyze the data in this study.
Results
A total of 100 patients met the inclusion criteria for evaluation. The induction regimens included lenalidomide plus bortezomib and low-dose dexamethasone (N = 86), low-dose dexamethasone (N = 12), carfilzomib plus low-dose dexamethasone (N = 1), and daratumumab plus low-dose dexamethasone (N = 1). All patients received thromboprophylaxis with either oral aspirin 81 mg to 325 mg once daily (median dose, 81 mg once daily), subcutaneous LMWH (enoxaparin 40 mg once daily or 30 mg twice daily), oral warfarin (international normalized ratio 2-3), or a direct oral anticoagulant (apixaban 5 mg twice daily or rivaroxaban 20 mg once daily, after loading dose).
The baseline characteristics of the patients are shown in Table 1. In all, 46% of our population was classified as high risk for thromboembolic events. The majority of patients had at least 1 cardiovascular risk factor (63%), including hypertension (52%), coronary arterial disease (8%), or previous myocardial infarction (3%). In the high-risk group, 7 patients had a history of VTE, including upper-extremity DVT (N = 1), lower-extremity DVT (N = 2), and pulmonary embolism (N = 3); in addition, 1 patient had DVT and pulmonary embolism.
The overall incidence of VTE was 9% (Table 2). Among the 46 patients in the high-risk group, the thromboprophylaxis regimens included aspirin (N = 34), direct oral anticoagulants (N = 7), warfarin (N = 4), and enoxaparin (N = 1). The overall rate of VTE among the 46 high-risk patients was 13%. Of these 46 patients, 4 (11.7%) of the 34 patients who received aspirin, 1 (25%) of the 4 who received warfarin, 1 (14%) of the 7 who received direct oral anticoagulants, and none of the patients who received enoxaparin had VTE.
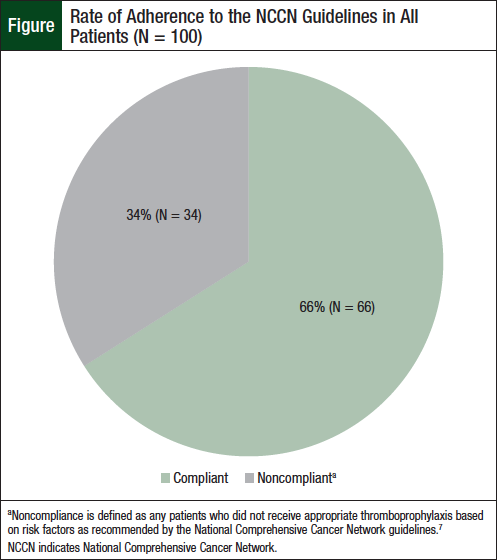
Of the 34 high-risk patients who received aspirin, 24 (70.5%) had 2 risk factors for VTE, 9 (26.4%) had 3 risk factors, and 2 (5.8%) had 4 risk factors. The rate of adherence to the NCCN guidelines among this high-risk group was 26%. Among the 12 patients who received appropriate thromboprophylaxis per guideline recommendations, 2 had a VTE. Among the 34 patients who did not adhere to the guidelines, 4 patients had a VTE. All 54 patients with a low VTE risk received thromboprophylaxis with aspirin. The rate of adherence to the NCCN guidelines among this low-risk group was 100%. The total adherence rate to the NCCN guidelines for all risk level groups was 66% (Figure).
Pulmonary embolism was only observed in 1 (5.6%) high-risk patient who received aspirin (Table 2). DVT was observed in 3 (8.8%) high-risk and 3 (5.6%) low-risk patients who received aspirin, 1 (14%) high-risk patient who received direct oral anticoagulants, 1 (25%) high-risk patient who received warfarin, and in none of the patients who received enoxaparin. There were no cases of arterial thrombosis, acute cardiovascular events, or death associated with VTE. Overall, most thromboembolic events occurred after the first 6 months of induction therapy. The median time to VTE was 7 months (range, 3-8 months). Only 1 patient had VTE at 3 months, and 8 patients had VTE after 6 months of induction therapy.
There was only 1 subdural hematoma major bleeding episode, which was in the aspirin group, although this event resolved spontaneously. There was 1 minor bleeding episode in the warfarin group. No bleeding complications were detected during thromboprophylaxis with the use of LMWHs or direct oral anticoagulants.
Discussion
In this retrospective chart review, we assessed adherence to thromboprophylaxis recommendations by the NCCN guidelines for patients with newly diagnosed multiple myeloma who are receiving lenalidomide-based induction therapies.7 The overall rate of compliance to the guidelines was 66%. All the low-risk patients were adherent to the NCCN guidelines; however, less than 50% of the high-risk patients received optimal thromboprophylaxis. A majority (N = 34) of the high-risk patients received VTE prophylaxis with aspirin.
Of these patients, 28 (80%) had cardiovascular disease, 12 (34%) were obese, 8 (22%) had immobility for more than 5 days, 7 (20%) had surgery within 3 months of induction therapy, 6 (17%) had chronic kidney disease, and 6 (17%) had diabetes. The majority of patients had the combined risk factors of obesity and hypertension. Several patients who received VTE prophylaxis with direct oral anticoagulants, LMWH, or warfarin had underlying comorbidities, for which these agents would be specifically indicated (eg, atrial fibrillation) versus unlabeled indications, including combined risk factors (eg, obesity and hypertension).
In our study, the overall incidence of VTE in patients who received optimal thromboprophylaxis was 7.6%, which is consistent with published literature.11,14 Our findings show that aspirin prophylaxis in high-risk patients with newly diagnosed multiple myeloma is associated with high rates of VTE (11.7%) compared with low-risk patients (5.6%). The rates of VTE in our study were higher than the reported rate in a prospective, randomized, open-label trial with overall incidence of VTE of approximately 2.3% in patients with newly diagnosed multiple myeloma who received induction therapy with lenalidomide plus low-dose dexamethasone.1 Of note, that study excluded patients with a history of VTE.1
The higher VTE rate in our study may result from more than 50% of the high-risk patients receiving suboptimal thromboprophylaxis with aspirin, and because we included patients with a history of VTE. In addition, our study had a follow-up of 12 months compared with 6-month follow-up in published literature.1 The majority of the VTE events in our study were seen after 6 months, as opposed to the first month of induction therapy as in a prospective clinical trial.1 Similar to our study, that clinical trial had no reports of acute cardiovascular events or sudden deaths. Also, as in our study, there was no evidence of early or late deaths that were determined to be related to VTE during treatment with this induction regimen.1
In our study, there was 1 pulmonary embolism event in a high-risk patient who received aspirin, whereas no pulmonary embolisms were reported among patients who received LMWH, warfarin, or direct oral anticoagulants. This is consistent with the findings in an article by Schulman and colleagues.13 Palumbo and colleagues evaluated VTE thromboprophylaxis in patients with newly diagnosed multiple myeloma who received thalidomide-based regimens, and no pulmonary embolisms were detected in the LMWH group.12 In another study by Mathew and colleagues, none of the 26 patients with multiple myeloma had a thrombotic event during 6 months of treatment with direct oral anticoagulants, including apixaban and rivaroxaban.10 However, 4 patients did eventually have a recurrent VTE after 6 months. Of note, 3 patients had their oral anticoagulants discontinued before the recurrent VTE episode, and 1 patient had a VTE after being switched to warfarin.10
In our study, DVT was observed in 6 patients who received aspirin, 1 patient who received warfarin, and 1 patient who received direct oral anticoagulants. The incidence of VTE in our study is inconsistent with other studies1,10; the rates of DVT are higher in our study, which may be in part attributed to our small sample of patients receiving various thromboprophylactic agents. This further limited our ability to assess the incidence of VTE based on individual patient-specific risk factors.
A recent study by Storrar and colleagues evaluated the rates of thrombosis in 70 patients with multiple myeloma who received IMiD-containing regimens and thromboprophylaxis with apixaban 2.5 mg twice daily.11 The rates of thrombosis were very low, with no cases of VTE identified, and only 2 cases of arterial thrombosis. Although 1 patient with coexistent thrombocytopenia had an episode of major bleeding, there was no other record of a clinically relevant bleeding episode.11 Based on these findings, apixaban is an attractive and safe option for patients with multiple myeloma who are receiving an IMiD-containing regimen. However, that study was limited by its retrospective design and small sample size.11 Prospective studies are needed to further validate these data.
In our study, 1 major bleeding event episode was associated with the use of high-dose aspirin (325 mg daily). Of note, the patient had thrombocytopenia at the time of the event, which might have contributed to the major bleeding. One patient who received warfarin had a minor bleeding episode. Otherwise, there were no major or minor episodes of bleeding reported in patients who received LMWH or direct oral anticoagulants. The rates of minor and major bleeding episodes in patients who received appropriate thromboprophylaxis were 2.17% and 1.8%, respectively.
In a randomized, open-label, multicenter phase 3 clinical trial comparing treatment with aspirin or fixed low-dose warfarin versus LMWH as thromboprophylaxis in 667 patients with multiple myeloma who received a thalidomide-based regimen, 6 (2.7%) patients in the aspirin group, 1 (0.5%) patient in the warfarin group, and 3 (1.4%) patients in the LMWH group had minor bleeding episodes.12 Our findings are consistent with what has been reported in the literature, which confirms that the incidence of major and minor bleeding episodes in patients with multiple myeloma who receive thromboprophylaxis is low.11
Clinical pharmacists play a major role in ensuring that patients receive appropriate thromboprophylaxis while improving medication adherence through patient counseling. Pharmacists may implement VTE risk assessments to provide appropriate thromboprophylaxis in patients with newly diagnosed multiple myeloma. By identifying barriers to medication adherence (eg, cost, drug-related toxicities, lack of education or awareness, polypharmacy), pharmacists can increase patient adherence to treatment regimens by recommending the utilization of a pill dispenser, maintaining an accurate medication list, finding alternative cost-saving medications, and minimizing polypharmacy.
Limitations
This study has several limitations, including its single-center, retrospective nature, as well as the small sample size.
In addition, the small number of patients within each subgroup who received a specific thromboprophylactic agent limited our analysis to compare the incidence of VTE rates among the different prophylactic strategies.
Finally, adherence was defined as patients receiving appropriate thromboprophylaxis based on the NCCN guidelines. However, we were unable to assess patient adherence to the prophylactic agents. This is a potential confounding factor that we could not control for in the analysis of our data, but may represent more realistic clinical outcomes.
Conclusion
Our findings show that although all patients received thromboprophylaxis, several high-risk patients received suboptimal prophylaxis with aspirin, which might have contributed to higher rates of VTE in our study. Providing optimal thromboprophylaxis in patients with newly diagnosed multiple myeloma is imperative to reducing the early complications of the disease. VTE risk assessments are essential when initiating thromboprophylaxis in patients with newly diagnosed multiple myeloma who are receiving lenalidomide-based induction therapies.
Newer and more effective thromboprophylactic agents, such as direct oral anticoagulants, are associated with fewer drug interactions and do not require laboratory monitoring. Prospective clinical studies are warranted to compare the efficacy and safety of direct oral anticoagulants, such as apixaban, rivaroxaban, edoxaban, and dabigatran etexilate, in patients with newly diagnosed multiple myeloma who are receiving lenalidomide plus steroid-based induction therapy.
Author Disclosure Statement
Dr Chew, Dr Afifi, Dr Nguyen, and Dr Merl have no conflicts of interest to report.
References
- Larocca A, Cavallo F, Bringhen S, et al. Aspirin or enoxaparin thromboprophylaxis for patients with newly diagnosed multiple myeloma treated with lenalidomide. Blood. 2012;119:933-939; quiz 1093.
- Wun T, White RH. Epidemiology of cancer-related venous thromboembolism. Best Pract Res Clin Haematol. 2009;22:9-23.
- Rajkumar SV, Jacobus S, Callander NS, et al; for the Eastern Cooperative Oncology Group. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: an open-label randomised controlled trial. Lancet Oncol. 2010;11:29-37. Erratum in: Lancet Oncol. 2010;11:14.
- Zonder JA, Crowley J, Hussein MA, et al. Lenalidomide and high-dose dexamethasone compared with dexamethasone as initial therapy for multiple myeloma: a randomized Southwest Oncology Group trial (S0232). Blood. 2010;116:5838-5841.
- Weber DM, Chen C, Niesvizky R, et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med. 2007;357:2133-2142.
- Dimopoulos M, Spencer A, Attal M, et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med. 2007;357:2123-2132. Erratum in: N Engl J Med. 2009;361:544.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Cancer-Associated Venous Thromboembolic Disease. Version 1.2019. February 28, 2019. www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf. Accessed August 1, 2019.
- Palumbo A, Rajkumar SV, Dimopoulos MA, et al. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia. 2008;22:414-423.
- Chapter 1: Definition and classification of CKD. Kidney Int Suppl (2011). 2013;3:19-62.
- Mathew S, Howlett C, Richter J, Jain R. Safety and efficacy of direct oral anticoagulants in multiple myeloma patients. Abstract presented at Eastern States Conference for Pharmacy Residents and Preceptors annual meeting; April 30-May 3, 2017; Hershey, PA.
- Storrar NPF, Mathur A, Johnson PRE, Roddie PH. Safety and efficacy of apixaban for routine thromboprophylaxis in myeloma patients treated with thalidomide- and lenalidomide-containing regimens. Br J Haematol. 2019;185:142-144.
- Palumbo A, Cavo M, Bringhen S, et al. Aspirin, warfarin, or enoxaparin thromboprophylaxis in patients with multiple myeloma treated with thalidomide: a phase III, open-label, randomized trial. J Clin Oncol. 2011;29:986-993.
- Schulman S, Kearon C; for the Subcommittee on Control of Anticoagulation of the Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. 2005;3:692-694.
- Cesarman-Maus G, Braggio E, Fonseca R. Thrombosis in multiple myeloma (MM). Hematology. 2012;17(suppl 1):S177-S180.