High doses of chemotherapy in patients who undergo autologous hematopoietic stem-cell transplantation (HSCT) can result in serious complications, including delayed or incomplete engraftment and end-organ damage. Specifically, patients with prolonged neutropenia are at high risk for the development of fevers or infections.1,2 Reducing the duration of neutropenia with stem-cell rescue and growth-factor support is crucial for decreasing morbidity and mortality in patients who receive conditioning chemotherapy for autologous HSCT.3-5
Granulocyte colony-stimulating factors (G-CSFs), also known as myeloid growth factors, are key regulators of granulocyte production released from the bone marrow, and are potent activators of mature neutrophils.6 G-CSFs are administered after transplantation to accelerate engraftment, which decreases the duration of neutropenia, the risk for infection, and length of hospital stay in patients who undergo autologous HSCT.5 Although filgrastim and pegfilgrastim have been studied,5,7-16 consensus is lacking on the optimal parameters for administering G-CSFs in the setting of autologous HSCT. According to the most recent guidelines of the American Society of Clinical Oncology, G-CSFs are recommended for use on day +1 following autologous HSCT.6 However, immediately after transplantation, late committed neutrophilic progenitors have not yet been formed, so the administration of a G-CSF before this time may have little, if any, effect.17
Although studies have been conducted to assess the optimal timing of G-CSF after autologous HSCT, study populations and results have been variable.5,7-17 Most studies of early versus delayed initiation of G-CSFs have used filgrastim, and did not demonstrate meaningful differences in the time to neutrophil recovery between administration on days +0, +4, and +5.7 Moreover, delaying filgrastim even further (to day +7) produced results similar to those of day +1 initiation, with respect to platelet recovery, neutropenic fever, and antibiotic use.17 In pediatric recipients of autologous HSCT, delayed administration of filgrastim, starting on day +6, produced results similar to those obtained with day +1 initiation with regard to duration of neutropenia, time to platelet engraftment, incidence of bloodstream infections, and duration of fevers.16
Later initiation of filgrastim post-HSCT may have cost benefits, in addition to similar clinical outcomes.16,17 In 2010, our institution implemented a practice change, from initiating filgrastim on day +5 to day +10 after autologous HSCT. The purpose of this study was to compare results for patients who began receiving filgrastim on day +5 versus day +10 following autologous HSCT.
Methods
This single-center, retrospective study was conducted at a tertiary care academic medical center (Rush University Medical Center, Chicago, IL). The study was approved by the Institutional Review Board before data collection. A database of records for patients who underwent stem-cell transplantation was used to identify those who received peripheral blood autologous HSCT with CD34+ stem cells between January 1, 2007, and June 30, 2013. Patients were included if they began receiving filgrastim on day +5 (+1) or day +10 (+1) after autologous HSCT. Those who did not receive filgrastim on day +5 (+1) or day +10 (+1) were excluded. Patients were matched based on age (+5 years) and type of malignancy.
Using the medical center's electronic medical record system and the stem-cell transplantation database, the following data were collected: age, sex, weight, height, allergies, type of malignancy requiring transplant, date of autologous HSCT, and chemotherapy conditioning regimen. Medication data were reviewed to identify the initiation and duration of administration of filgrastim and antibiotics. Other data collection points included temperatures, blood and urine cultures, length of hospital stay, and survival end points.
The primary end point was time to engraftment, which was defined as absolute neutrophil count (ANC) recovery to ≥500 cells/mm3 sustained for 48 hours, or ANC ≥1000 cells/mm3 sustained for 24 hours. Secondary end points included length of hospitalization, total febrile days during neutropenia and hospitalization, number of days on antibiotic(s), and rates of infection (assessed by results of urine and/or blood cultures). Length of hospital stay was defined as the number of days from admission until discharge. Fever was defined as a single temperature of ≥101.0°F (≥38.3°C), or a temperature of ≥100.4°F (≥38.0°C) sustained for 1 hour. The number of filgrastim doses administered was compared in the 2 groups for cost-analysis purposes. Per our institutional policy, patients who weighed ≤70kg received 300µg/d until engraftment, and those who weighed >70kg received 480µg/d until engraftment.
Continuous data were analyzed using a t-test or Mann-Whitney U test, as appropriate. Categorical variables were analyzed using a chi-squared or Fisher exact test. P values <.05 were considered significant. The cost analysis was based on the average cost of filgrastim per patient. SPSS Software (Armonk, NY), version 19, was used to analyze the data.
Results
Patient Demographics
Between January 1, 2007, and June 30, 2013, 473 patients received autologous HSCT at our institution, 158 of whom met the inclusion criteria to be matched. The timing of filgrastim initiation was determined by each physician, which resulted in some patients not meeting the inclusion criteria. A total of 79 matched pairs were included in our analyses of primary and secondary end points. Baseline characteristics were well-balanced between the 2 study groups (Table 1).
Efficacy
Results are summarized in Table 2. Neutrophil engraftment was significantly faster in patients who began receiving filgrastim on day +5 after transplantation (average, 8.27 days vs 8.96 days for the +10 group; P = .006; median, 9 days for both groups; range, 5-14 days vs 6-13 days, respectively). The average length of hospital stay was 20 days for both groups (P = .745). The number of febrile days during neutropenia differed significantly: 2 days for filgrastim initiation on day +5 versus 1 day for initiation on day +10 (P = .02). The total number of febrile days during hospitalization was similar for both groups (P = .68). Rates of infection were higher for the day +5 group, as indicated by the number of positive blood cultures (16 vs 11; P = .291) and the number of positive urine cultures (19 vs 10; P = .64). The number of days of antibiotic use was similar: 9.2 for the day +5 group, and 8.3 for the day +10 group; P = .156. The average number of doses per patient was significantly higher for the day +5 group than the day +10 group (7 vs 5; P < .01). No patient died during the study period in either group.
Cost
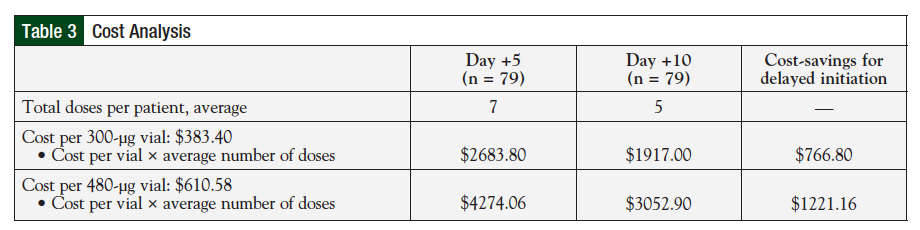
A cost analysis was performed using the average wholesale price per vial for filgrastim: $383.40 for a 300-µg vial, and $610.58 for a 480-µg vial. In the day +5 group, 27 patients required the 300-µg dose, and 52 required the 480-µg dose. Similarly, in the day +10 group, 25 patients received a dose of 300 µg, and 54 received a dose of 480 µg. The average number of doses was 7 in the day +5 group, and only 5 in the day +10 group. The average cost-savings per patient for delaying initiation of filgrastim to day +10 (+1) was $766.80 for the 300-µg dose, and $1221.16 for the 480-µg dose. Results of the cost analysis are summarized in Table 3.
Discussion
G-CSFs are routinely used to optimize supportive care in patients who undergo autologous HSCT, from stem-cell mobilization through neutrophil recovery. Relative to placebo, neutrophil engraftment is accelerated with G-CSFs.5,18-26 Many studies have shown that reducing the duration of neutropenia results in decreased treatment-related mortality, length of hospital stay, number of infections, and/or number of febrile days.5,7,18-25,27 Although the use of G-CSFs after autologous HSCT is widely supported, consensus is lacking on the optimal timing of their initiation.
Advances in stem-cell mobilization include expedited neutrophil engraftment with the use of peripheral blood stem cells (PBSCs) as an autologous HSCT source.27 The increased use of PBSCs as the source of stem cells led researchers to investigate whether delayed administration of G-CSFs in the autologous HSCT setting would lead to similar outcomes for neutrophil recovery.5,7,16,17,21,27,28 Vey and colleagues21 and Faucher and colleagues28 concluded that delaying the administration of filgrastim until day +6 produces the same clinical benefit as administration on day +1. Although our study showed a numerical difference in time to engraftment favoring day +5 filgrastim initiation, the numbers were essentially the same, allowing us to conclude clinical insignificance. These results were similar to those of Vey and colleagues, and Faucher and colleagues. Bence-Bruckler and colleagues17 and Khwaja and colleagues26 suggested that filgrastim administration could be delayed until 7 or 8 days, respectively, after autologous HSCT without compromising efficacy, and with an accompanying reduction in treatment costs compared with day +1 administration-which is similar to our findings. The results of these studies suggest that initiating filgrastim at a later time following autologous HSCT does not delay engraftment.
The objective of our study was to compare delayed administration of filgrastim (day +5) with very delayed administration (day +10). We hypothesized that the effect on engraftment would be similar with both times of initiation. Although the difference in the primary end point (time to engraftment) was significant, favoring filgrastim initiation on day +5, we believe that this finding does not represent a clinically meaningful difference in neutrophil engraftment, particularly because the results for the secondary end points were similar. The average difference in neutrophil engraftment time between the study groups was <1 day. Length of hospital stay, rates of infection, and total febrile days were similar, which has been reported previously. Future studies are warranted to examine other secondary end points, including platelet recovery and platelet transfusions. The current literature is conflicting as to the best time to initiate filgrastim after autologous HSCT. However, our results for time to engraftment and reduction in costs are similar to those of other studies.21,28
In addition to delayed engraftment, infectious complications, and prolonged hospitalization, autologous HSCT is associated with high costs.6 Our study shows that delaying initiation of filgrastim from day +5 to day +10 has a positive impact on cost-savings. On average, 75 autologous HSCTs are performed annually in our institution. Considering the average number of doses administered in each group, the potential savings associated with very delayed initiation would be $57,510 to $91,587 annually (at current per-vial costs). To analyze the true cost impact of very delayed administration of filgrastim, further investigation is warranted to determine and evaluate other factors that contribute to costs.
Few retrospective and prospective studies on delayed filgrastim administration have been conducted.17,21,26,28 To our knowledge, this is the first retrospective study in which the impact of very delayed administration (day +10) was examined. Strengths of this analysis include matching patient demographics (including age and malignancy), computerized documentation for all end points, and a highly standardized transplantation program.
Limitations
Limitations of the study include the retrospective design, the relatively small sample size, and the inclusion of only one study center. Because filgrastim was not initiated consistently on day +5 or day +10 after HSCT, the inclusion criteria were expanded to day +5 (+1) and day +10 (+1), which resulted in some variation among the matched pairs. Thus, comparisons were made between patients who had initiation on day +4, day +5, or day +6 and those who had initiation on day +9, day +10, or day +11. The findings for precisely matched pairs may have differed.
Conclusion
It appears that delaying filgrastim administration until day +10 after autologous HSCT does not result in a clinically meaningful delay in neutrophil engraftment compared with filgrastim initiation on day +5. This is supported by the similar lengths of hospitalization, rates of infection, and total febrile days observed in our study groups. For institutions with filgrastim on the inpatient formulary for autologous HSCT, delaying administration until day +10 may be considered a safe and economical option. However, given that this was a retrospective, single-center study, the optimal timing of filgrastim administration is still unknown, and a prospective, randomized controlled trial is needed to confirm the results.
Author Disclosure Statement
The authors reported no conflicts of interest.
References
1. Bearman SI, Appelbaum FR, Back A, et al. Regimen related toxicity and early post-transplant survival in patients undergoing marrow transplantation for lymphoma. J Clin Oncol. 1989;7:1288-1294.
2. Mossad SB, Longworth DL, Goormastic M, et al. Early infectious complications in autologous bone marrow transplantation: a review of 219 patients. Bone Marrow Transplant. 1996;18:265-271.
3. Smith LA, Wright-Kanuth MS. Complications and risks in hematopoietic stem cell transplant patients. Clin Lab Sci. 2001;14:118-124.
4. Junghanss C, Marr KA. Infectious risks and outcomes after stem cell transplantation: are nonmyeloablative transplants changing the picture? Curr Opin Infect Dis. 2002;15:347-353.
5. Trivedi M, Martinez S, Corringham S, et al. Optimal use of G-CSF administration after hematopoietic SCT. Bone Marrow Transplant. 2009;43:895-908.
6. Smith TJ, Khatcheressian J, Lyman GH, et al. 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol. 2006;24:3187-3205.
7. Khot A, Dickinson M, Stokes K, et al. A risk-adapted protocol for delayed administration of filgrastim after high-dose chemotherapy and autologous stem cell transplantation. Clin Lymphoma Myeloma Leuk. 2013;13:42-47.
8. Sebban C, Lefranc A, Perrier L, et al. A randomized phase II study of the efficacy, safety, and cost-effectiveness of pegfilgrastim and filgrastim after autologous stem cell transplant for lymphoma and myeloma (PALM study). Eur J Cancer. 2012;48:713-720.
9. Wannesson L, Luthi F, Zucca E, et al. Pegfilgrastim to accelerate neutrophil engraftment following peripheral blood stem cell transplant and reduce the duration of neutropenia, hospitalization, and use of intravenous antibiotics: a phase II study in multiple myeloma and lymphoma and comparison with filgrastim-treated matched controls. Leuk Lymphoma. 2011;52:436-443.
10. Samaras P, Buset EM, Siciliano RD, et al. Equivalence of pegfilgrastim and filgrastim in lymphoma patients treated with BEAM followed by autologous stem cell transplantation. Oncology. 2010;79:93-97.
11. Samaras P, Blickenstorfer M, Siciliano RD, et al. Pegfilgrastim reduces the length of hospitalization and the time to engraftment in multiple myeloma patients treated with melphalan 200 and auto-SCT compared with filgrastim. Ann Hematol. 2011;90:89-94.
12. Rifkin R, Spitzer G, Orloff G, et al. Pegfilgrastim appears equivalent to daily dosing of filgrastim to treat neutropenia after autologous peripheral blood stem cell transplantation in patients with non-Hodgkin lymphoma. Clin Lymphoma Myeloma Leuk. 2010;10:186-191.
13. Mathew S, Adel N, Rice RD, et al. Retrospective comparison of the effects of filgrastim and pegfilgrastim on the pace of engraftment in auto-SCT patients. Bone Marrow Transplant. 2010;45:1522-1527.
14. Gerds A, Fox-Geiman M, Dawravoo K, et al. Randomized phase III trial of pegfilgrastim versus filgrastim after autologous peripheral blood stem cell transplantation. Biol Blood Marrow Transplant. 2010;16:678-685.
15. Castagna L, Bramanti S, Levis A, et al. Pegfilgrastim versus filgrastim after high-dose chemotherapy and autologous peripheral blood stem cell support. Ann Oncol. 2010;21:1482-1485.
16. Pai V, Fernandez SA, Laudick M, et al. Delayed administration of filgrastim (G-CSF) following autologous peripheral blood stem cell transplantation (APBSCT) in pediatric patients does not change time to neutrophil engraftment and reduces use of G-CSF. Pediatr Blood Cancer. 2010;54:728-733.
17. Bence-Bruckler I, Bredeson C, Atkins H, et al. A randomized trial of granulocyte colony-stimulating factor (Neupogen) starting day 1 vs day 7 post-autologous stem cell transplantation. Bone Marrow Transplant. 1998;22:965-969.
18. Sheridan WP, Morstyn G, Wolf M, et al. Granulocyte colony-stimulating factor and neutrophil recovery after high-dose chemotherapy and autologous bone marrow transplantation. Lancet. 1989;2:891-895.
19. Taylor KM, Jagannath S, Spitzer G, et al. Recombinant human granulocyte colony-stimulating factor hastens granulocyte recovery after high-dose chemotherapy and autologous bone marrow transplantation in Hodgkin's disease. J Clin Oncol. 1989;7:1791-1799.
20. Stahel RA, Jost LA, Cerny T, et al. Randomized study of recombinant human granulocyte colony-stimulating factor after high-dose chemotherapy and autologous bone marrow transplantation for high-risk lymphoid malignancies. J Clin Oncol. 1994;12:1931-1938.
21. Vey N, Molnar S, Faucher C, et al. Delayed administration of granulocyte colony-stimulating factor after autologous bone marrow transplantation: effect on granulocyte recovery. Bone Marrow Transplant. 1994;14:779-782.
22. Klumpp TR, Mangan KF, Goldberg, SL, et al. Granulocyte colony-stimulating factor accelerates neutrophil engraftment following peripheral-blood stem cell transplantation: a prospective randomized trial. J Clin Oncol. 1995;13:1323-1327.
23. Shimazaki C, Oku N, Uchiyama H, et al. Effect of granulocyte colony-stimulating factor on hematopoietic recovery after peripheral blood progenitor cell transplantation. Bone Marrow Transplant. 1994;13:271-275.
24. McQuaker IG, Hunter AE, Pacey S, et al. Low-dose filgrastim significantly enhances neutrophil recovery following autologous peripheral-blood stem-cell transplantation in patients with lymphoproliferative disorders: evidence for clinical and economic benefit. J Clin Oncol. 1997;15:451-457.
25. Schmitz N, Dreger P, Zander AR, et al. Results of a randomized, controlled, multicentre study of recombinant human granulocyte colony-stimulating factor (filgrastim) in patients with Hodgkin's disease and non-Hodgkin's lymphoma undergoing autologous bone marrow transplantation. Bone Marrow Transplant. 1995;15:261-266.
26. Lewis A. Autologous stem cells derived from the peripheral blood compared to standard bone marrow transplant; time to engraftment: a systemic review. Int J Nurs Stud. 2005;42:589-596.
27. Khwaja A, Mills W, Leveridge K, et al. Efficacy of delayed granulocyte colony-stimulating factor after autologous BMT. Bone Marrow Transplant. 1993;11:479-482.
28. Faucher C, Le Corroller AG, Chabannon C, et al. Administration of G-CSF can be delayed after transplantation of autologous G-CSF-primed blood cells: a randomized study. Bone Marrow Transplant. 1996;17:533-536.