Lung cancer is the second most common cancer in the United States.1 In 2015, an estimated 221,200 new cases of lung cancer were diagnosed, representing 13.3% of all new cancer cases.1 Furthermore, lung cancer is the leading cause of cancer mortality in men and women, responsible for 27% of all cancer deaths, and claiming more lives than breast, colon, prostate, and ovarian cancers combined.1,2 From 2007 to 2011, there was a slight decline in lung cancer incidence and deaths.3
The 2 major types of lung cancers include non–small-cell lung cancer (NSCLC) and small-cell lung cancer.4 Approximately 85% to 90% of all lung cancers are NSCLC, which comprise squamous-cell carcinoma, adenocarcinoma, and large-cell carcinoma.4,5 Less common types of NSCLC include carcinoid tumor, pleomorphic, salivary gland carcinoma, and unclassified carcinoma.5 Squamous-cell carcinomas account for 25% to 30% of all lung cancers, and adenocarcinomas account for 40% of all lung cancers.4
As with other cancers, the earlier that lung cancer is detected and treated, the better the prognosis.2 Nevertheless, approximately 22% of lung cancers have already spread to regional lymph nodes at diagnosis, and 57% have metastasized to a distant site.1 The 5-year relative survival rate for patients with lung cancer that has spread regionally is 27.4%, and only 4.2% for patients with metastatic disease.1
The treatment of metastatic NSCLC typically includes chemotherapy, targeted therapy, immunotherapy, or a combination of these options.6 The majority of targeted therapies are either small-molecule compounds designed for targets inside the cell, or monoclonal antibodies designed for targets on the outer surface of cancer cells.6
Scientific advances in cell biology and gene expression have fostered the development of new targeted therapies and have changed the landscape of NSCLC treatment.7 Several predictive biomarkers have emerged.7 Some of these biomarkers include the sensitizing epidermal growth factor receptor (EGFR) mutations (eg, exon 19 deletion or exon 21 L858R mutation) and the anaplastic lymphoma kinase (ALK) fusion gene.7
Sensitizing EGFR mutations are harbored in an estimated 10% of Caucasian patients with NSCLC, and in up to 50% of Asian patients with NSCLC.7 ALK gene rearrangements are found in an estimated 2% to 7% of patients with NSCLC.7 Oncology practice guidelines recommend testing for EGFR mutations and ALK gene rearrangements for select patients with NSCLC.7,8 Other biomarkers have been identified and are being studied for their potential response to targeted therapies.7
Molecular profiling and the evolving research and development of targeted, patient-precise treatments are transforming the therapeutic options for patients with NSCLC. In 2015, several targeted therapies—including immunotherapies, angiogenesis inhibitors, tyrosine kinase inhibitors, and monoclonal antibodies—were approved for the treatment of patients with NSCLC. Several of those approvals were new indications for drugs already available on the market, and several approvals were for novel targeted therapies—altogether, the year 2015 was a banner year for the approval of new therapy for NSCLC, significantly increasing the treatment options available for patients with lung cancer.
Necitumumab Approval for First-Line Therapy in Metastatic Squamous NSCLC
On November 24, 2015, the US Food and Drug Administration (FDA) approved the EGFR antagonist necitumumab (Portrazza; Eli Lilly) injection for intravenous (IV) use, in combination with gemcitabine and cisplatin, for the first-line treatment of patients with metastatic squamous NSCLC.9,10 Necitumumab is not indicated for the treatment of patients with nonsquamous NSCLC.10
The EGFR protein is expressed in the majority of NSCLC tumors, particularly in squamous-cell disease.11 Necitumumab, a recombinant human immunoglobulin (Ig)G1 monoclonal antibody, is an EGFR antagonist with potential antineoplastic activities.10,12
Commenting on necitumumab’s approval, Richard Pazdur, MD, Director of the FDA’s Office of Hematology and Oncology Products, said, “Lung cancer tumors can be varied, so treatment options need to be tailored to the specific type of lung cancer in the patient. Today’s approval provides certain patients with squamous cell lung cancer a new option that may extend survival.”9
Mechanism of Action
Necitumumab is an IgG1 monoclonal antibody and an EGFR antagonist that acts by binding to EGFR and blocking the binding of EGFR to its ligands, ultimately preventing the activation and the dimerization of the receptor.12 This process may inhibit the EGFR-dependent downstream pathways, thereby blocking EGFR-dependent tumor-cell proliferation and metastasis.10,12 Based on in vitro studies, the binding activity of necitumumab also resulted in antibody-dependent cellular cytotoxicity in cells that express EGFR.10
Dosing and Administration
The recommended dose of necitumumab is 800 mg (absolute dose) administered as an IV infusion for 60 minutes on days 1 and 8 of each 3-week cycle before the infusion of gemcitabine and cisplatin. Treatment with necitumumab should be continued until the disease progresses or until unacceptable toxicity occurs.10
Necitumumab is available as an 800-mg/50-mL (16-mg/mL) solution in a single-dose vial.10
Clinical Trials
The SQUIRE Study: Patients with Squamous NSCLC
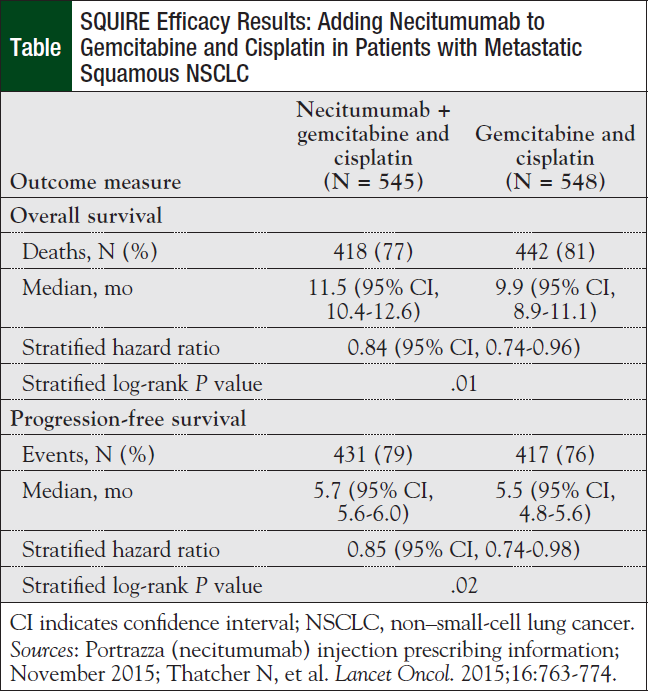
The FDA approval of necitumumab was based on findings from SQUIRE, an open-label, controlled phase 3 clinical trial conducted at 184 sites in 26 countries. Overall, 1093 patients (median age, 62 years) with squamous NSCLC were randomized to necitumumab (800 mg) plus gemcitabine and cisplatin or to gemcitabine and cisplatin alone for a maximum of 6 cycles or until evidence for disease progression or unacceptable toxicity.10,11
The main efficacy measure was overall survival. The secondary efficacy outcome measures included investigator-assessed progression-free survival and overall response rate. The results showed that patients in the necitumumab arm had a median overall survival of 11.5 months versus 9.9 months in patients receiving gemcitabine and cisplatin alone; this represents a significant difference of more than 2 months survival with necitumumab (Table).10,11
In addition, the median progression-free survival was 5.7 months in the necitumumab arm versus 5.5 months with gemcitabine plus cisplatin alone, representing a significant difference.10 There was no difference in overall response rate between the 2 study arms—31% (95% confidence interval [CI], 27-35) with necitumumab compared with 29% (95% CI, 25-33) with gemcitabine and cisplatin alone (P = .40).10
The INSPIRE Study: Patients with Nonsquamous NSCLC
INSPIRE was a randomized, open-label clinical trial of 633 patients with nonsquamous NSCLC. The combination of necitumumab, pemetrexed, and cisplatin did not provide a clinical benefit compared with pemetrexed and cisplatin alone.10,13 This study was terminated early because of an increased rate of any-cause mortality and thromboembolic events in the necitumumab-containing arm. Therefore, necitumumab is not indicated for the treatment of patients with nonsquamous NSCLC.10
Adverse Events
The most common adverse events (all grades) reported in patients receiving necitumumab at a rate of ≥15%, and ≥2% higher than gemcitabine and cisplatin alone included rash (44%), vomiting (29%), diarrhea (16%), and dermatitis acneiform (15%). The most common severe (grade ≥3) adverse reactions that occurred at a ≥2% higher rate in the necitumumab group versus those who received gemcitabine and cisplatin alone included thromboembolic events (5%), rash (4%), and vomiting (3%).10
Hypomagnesemia was the most common electrolyte abnormality in patients who received necitumumab plus gemcitabine and cisplatin (all grades, 83%; grade 3 or 4, 20%) compared with patients who received gemcitabine and cisplatin alone (all grades, 70%; grade 3 or 4, 7%).10
Overall, 12% of patients in the SQUIRE clinical trial discontinued treatment with necitumumab because of an adverse reaction. The most common toxicity leading to the discontinuation of necitumumab was a skin rash (1%).10
Necitumumab has no contraindications.10
Drug Interactions
When necitumumab was administered in combination with gemcitabine and cisplatin to 12 patients with advanced solid tumors, the gemcitabine geometric mean dose-normalized area under the curve increased by 22%, and the Cmax increased by 63% compared with the administration of gemcitabine and cisplatin alone; however, cisplatin exposure remained unchanged.10
Necitumumab exposure was not altered when it was administered concomitantly with gemcitabine and cisplatin.10
Warnings and Precautions
Boxed warnings. Necitumumab carries a boxed warning regarding the risk for cardiopulmonary arrest and/or sudden death, and the risk for hypomagnesemia.10
Cardiopulmonary arrest and/or sudden death occurred in 3% of patients who received necitumumab in combination with gemcitabine and cisplatin. The patient’s serum electrolyte levels should be closely monitored during and after treatment with necitumumab.10
Hypomagnesemia occurred in 83% (severe in 20%) of patients who received necitumumab in combination with gemcitabine and cisplatin. Patients should be monitored for hypomagnesemia, hypocalcemia, and hypokalemia before the administration of each dose of necitumumab, during treatment with necitumumab, and for at least 8 weeks after completing treatment with necitumumab. Necitumumab should be withheld in patients with grade 3 or 4 electrolyte abnormalities; electrolytes should be replaced based on medical necessity.10
Venous thromboembolic events (VTEs) and arterial thromboembolic events (ATEs). VTEs and ATEs, some fatal, have been reported with necitumumab. Based on an exploratory study analysis, the relative risk for these events was approximately 3-fold higher in patients with a history of a VTE or an ATE than in patients with no such history. Necitumumab should be discontinued in patients with a serious or life-threatening VTE or ATE.10
Dermatologic toxicities. Patients should be monitored for dermatologic toxicities, which were reported in 79% of patients who received necitumumab (with 8% severe cases) in SQUIRE. These toxicities include rash, dermatitis acneiform, acne, dry skin, pruritus, generalized rash, skin fissures, maculopapular rash, and erythema.10
Infusion-related reactions. Patients should be monitored for symptoms of infusion-related reactions, the majority of which occur after the first or the second necitumumab infusion. Necitumumab should be discontinued if serious or life-threatening infusion-related reactions occur.10
Nonsquamous NSCLC. There is an increased risk for toxicity and mortality in patients with nonsquamous NSCLC who receive necitumumab plus pemetrexed and cisplatin.10
Embryo-fetal toxicity. Necitumumab can cause fetal harm when administered to a pregnant woman.10
Use in Specific Populations
Pregnancy. Pregnant women should be advised about the potential risk to the fetus, and the risk to postnatal development. Women of reproductive potential should be advised to use effective contraception during treatment with necitumumab, and for 3 months after the final dose of necitumumab.10
Geriatric use. In an exploratory subgroup analysis of SQUIRE, the hazard ratio for overall survival in patients aged ≥70 years was 1.03. There was an increased incidence of VTE, including pulmonary embolism, in patients aged ≥70 years.10
Patients with renal or hepatic impairment. No correlation was identified between necitumumab exposure and renal or hepatic function.10
Conclusion
A new first-line treatment option for patients with metastatic squamous NSCLC became available with the FDA approval of necitumumab, an EGFR antagonist and an IgG1 monoclonal antibody, for use in combination with gemcitabine and cisplatin. Patients with advanced squamous NSCLC who received necitumumab plus gemcitabine and cisplatin had a significant improvement in overall survival and progression-free survival compared with patients who received gemcitabine and cisplatin alone.
References
- National Cancer Institute. SEER cancer stat fact sheets: lung and bronchus cancer. http://seer.cancer.gov/statfacts/html/lungb.html. Accessed February 2, 2016.
- Mayo Clinic staff. Diseases and conditions: lung cancer. September 25, 2015. www.mayoclinic.org/diseases-conditions/lung-cancer/basics/causes/con-20025531. Accessed February 3, 2016.
- American Cancer Society. Cancer facts and figures 2015. 2015. www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-044552.pdf. Accessed February 3, 2016.
- American Cancer Society. Lung cancer (non-small cell). Revised February 8, 2016. www.cancer.org/cancer/lungcancer-non-smallcell/detailedguide/non-small-cell-lung-cancer-what-is-non-small-cell-lung-cancer. Accessed February 23, 2016.
- National Cancer Institute. Non-small cell lung cancer treatment (PDQ®). Updated May 12, 2015. www.cancer.gov/types/lung/patient/non-small-cell-lung-treatment-pdq. Accessed February 4, 2016.
- National Cancer Institute. Targeted cancer therapies. www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet. Accessed February 4, 2016.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): non-small cell lung cancer. Version 4.2016. January 12, 2016. www.nccn.org/professionals/physician_gls/pdf/nscl.pdf. Accessed February 3, 2016.
- Leighl NB, Rekhtman N, Biermann WA, et al. Molecular testing for selection of patients with lung cancer for epidermal growth factor receptor and anaplastic lymphoma kinase tyrosine kinase inhibitors: American Society of Clinical Oncology endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Guideline. J Clin Oncol. 2014;32:3673-3679.
- US Food and Drug Administration. FDA approves Portrazza to treat advanced squamous non-small cell lung cancer. Press release. November 24, 2015. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm474131.htm. Accessed January 18, 2016.
- Portrazza (necitumumab) injection [prescribing information]. Indianapolis, IN: Eli Lilly and Company; November 2015.
- Thatcher N, Hirsch FR, Luft AV, et al; for the SQUIRE Investigators. Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2015; 16:763-774.
- National Cancer Institute. NCI Drug Dictionary. Necitumumab. www.cancer.gov/publications/dictionaries/cancer-drug?cdrid=656651. Accessed January 22, 2016.
- Paz-Ares L, Mezger J, Ciuleanu TE, et al; for the INSPIRE Investigators. Necitumumab plus pemetrexed and cisplatin as first-line therapy in patients with stage IV non-squamous non-small-cell lung cancer (INSPIRE): an open-label, randomised, controlled phase 3 study. Lancet Oncol. 2015;16:328-337.