Lung cancer is the leading cause of cancer-related mortality in the United States among men and women.1 The most common type of the disease, non–small-cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases.1 NSCLC comprises several disease histologies, including adenocarcinoma, squamous-cell carcinoma, nonsquamous carcinoma, large-cell anaplastic carcinoma, and adenosquamous carcinoma.1
Traditionally, patients with metastatic NSCLC have been managed with systemic (cytotoxic) chemotherapies.1 Although the use of platinum-based doublet chemotherapy has improved median overall survival for patients with newly diagnosed advanced NSCLC, these doublets are associated with significant toxicities, including myelosuppression, nausea and vomiting, kidney damage, and alopecia.1 For patients with relapsed metastatic NSCLC, single-agent chemotherapy has been the standard of care.1
With enhanced knowledge of lung cancer cell biology and gene expression, the development of novel agents has altered the treatment paradigm for NSCLC.2 Several driver oncogenes have been identified in NSCLC, including the epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), and KRAS.2,3 Approximately 10% of patients from North America and approximately 33% of patients from East Asia have EGFR mutations.2 The majority of EGFR mutations occur in women with adenocarcinomas who have never smoked.2 The 2 tyrosine kinase inhibitors that target EGFR, afatinib (Gilotrif) and erlotinib (Tarceva), are currently approved for patients with specific EGFR mutations.2
Overall, 2 other agents, crizotinib (Xalkori) and ceritinib (Zykadia) target NSCLC with ALK mutations.3 In unselected populations of patients with NSCLC, the frequency of ALK translocation ranges from 2% to 7%.3 In the majority of patients with NSCLC, EGFR and ALK mutations are not correlated with the presence of other oncogenic mutations, including KRAS.3
Once thought to be poorly immunogenic, lung cancer is now understood to be vulnerable to checkpoint inhibition.4 The immune system functions to recognize foreign proteins (antigens) as abnormal and destroy them, while simultaneously protecting against tissue damage.4 Immune checkpoints, including programmed death (PD)-1, are specific pathways that are hardwired into the immune system to allow this self-tolerance. By blocking these immune checkpoint pathways, immuno-oncology agents can strengthen the ability of tumor-infiltrating T-cells to recognize cancer cells as dangerous and subsequently remove them.5
Several clinical trials are evaluating the efficacy and safety of immune checkpoint inhibitors for patients with advanced lung cancer.6,7
Based on a demonstrated overall survival advantage compared with cytotoxic chemotherapy in patients with relapsed NSCLC, nivolumab (Opdivo) was approved on October 9, 2015, by the US Food and Drug Administration (FDA) for use in patients with metastatic squamous NSCLC that has progressed with or after platinum-based chemotherapy.6-8
Pembrolizumab Approval
On October 2, 2015, the FDA approved pembrolizumab (Keytruda Injection; Merck Sharp & Dohme Corp) for the treatment of patients with metastatic NSCLC whose tumors express PD ligand 1 (PD-L1) as determined by a genetic diagnostic test, and whose disease progressed with or after platinum-containing chemotherapy.9,10 The FDA approved the companion diagnostic test, PD-L1 IHC 22C3 pharmDx, which determines PD-L1 expression, on October 9, 2015.9
The labeling for pembrolizumab for metastatic NSCLC specifically notes that patients with EGFR or ALK mutations should have disease progression while receiving FDA-approved therapy for these mutations before receiving pembrolizumab.9,10
Pembrolizumab was the first human PD-1 inhibitor to receive approval for this specific indication in the United States.11
Pembrolizumab was initially approved by the FDA on September 4, 2014, for the treatment of patients with unresectable or metastatic melanoma whose disease progressed after ipilimumab therapy, and, if BRAF V600 mutation–positive, after a BRAF inhibitor.12
Pembrolizumab, which is administered via intravenous infusion, was approved under the FDA’s accelerated approval program based on the surrogate end points of overall response rate and duration of response.10 An improvement in survival or disease-related symptoms with pembrolizumab has not yet been established in metastatic melanoma or in metastatic NSCLC.10
Mechanism of Action
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor to block interaction with its ligands, PD-L1 and PD-L2.10 This binding activates T-cell–mediated immune responses against tumor cells. Animal studies showed that blocking PD-1 activity resulted in decreased tumor growth in genetically identical mouse tumor models.10
Dosing and Administration
The recommended dosage of pembrolizumab is 2 mg/kg administered as an intravenous infusion for 30 minutes every 3 weeks.10 Pembrolizumab should be administered until disease progression or until unacceptable toxicity.10
No dose adjustment of pembrolizumab is needed for patients with renal impairment or for patients with mild hepatic impairment, defined as total bilirubin of upper limit of normal or less and aspartate aminotransferase (AST) more than upper limit of normal or total bilirubin >1 to 1.5 times upper limit of normal and any AST.10 Pembrolizumab has not been studied in patients with moderate or severe hepatic impairment.10
KEYNOTE-001 Clinical Trial
The approval of pembrolizumab for patients with PD-L1–expressing metastatic NSCLC was based on a subgroup analysis of 280 patients enrolled in the KEYNOTE-001 clinical trial, a multicenter, open-label, multicohort, activity-estimating study.9,10,13
This international clinical trial was designed to evaluate the efficacy and safety of pembrolizumab in patients with advanced NSCLC, as well as to define and validate a tumor PD-L1 expression level that is associated with the likelihood of clinical benefit with pembrolizumab.13
To be included in the efficacy analysis, patients with metastatic NSCLC had to demonstrate disease progression after platinum-containing chemotherapy, and, if appropriate, targeted therapy for ALK or EGFR mutations, as well as evidence of PD-L1 expression by a clinical trial immunohistochemistry assay.10 Patients with an autoimmune disease, with a medical condition that required immunosuppression, and those who had received more than 30 Gy of thoracic radiation within the previous 26 weeks were ineligible to participate.10
This prospectively defined subgroup was retrospectively analyzed using a validated test for PD-L1 expression tumor proportion score.10 This analysis identified a subgroup of 61 (22% of 280) patients who had a PD-L1 expression tumor proportion score of ≥50% tumor cells as determined by the PD-L1 IHC 22C3 pharmDx Kit.10 These patients received pembrolizumab 10 mg/kg every 2 or 3 weeks until unacceptable toxicity or disease progression. Assessments of tumor status were performed at 9-week intervals.10
In the retrospectively defined subgroup of 61 patients with previously treated metastatic NSCLC (median age, 60 years), 61% of patients were male and 79% were white.10 All patients had an Eastern Cooperative Oncology Group performance status of 0 or 1. The disease histology included nonsquamous (75%) and squamous (21%) NSCLC. Overall, 98% of patients had M1 disease, and 11% had brain metastases. Previous therapies included 1 (26%), 2 (30%), or ≥3 (44%) regimens.10 EGFR and ALK mutations were rare.10
Among the 61 patients whose tumors expressed PD-L1, the overall response rate associated with pembrolizumab was 41% (95% confidence interval, 29%-54%; Table 1).10 All responses were partial responses.10 Of the 25 responding patients, 21 patients had ongoing responses at the final analysis of the overall response rate, and 11 (44%) had ongoing responses of ≥6 months.10
A separate subgroup of KEYNOTE-001 had enrolled 25 patients with a PD-L1 expression tumor proportion score ≥50%; these patients received pembrolizumab at a dose of 2 mg/kg every 3 weeks. Although follow-up was limited, activity was also observed.10
Adverse Events
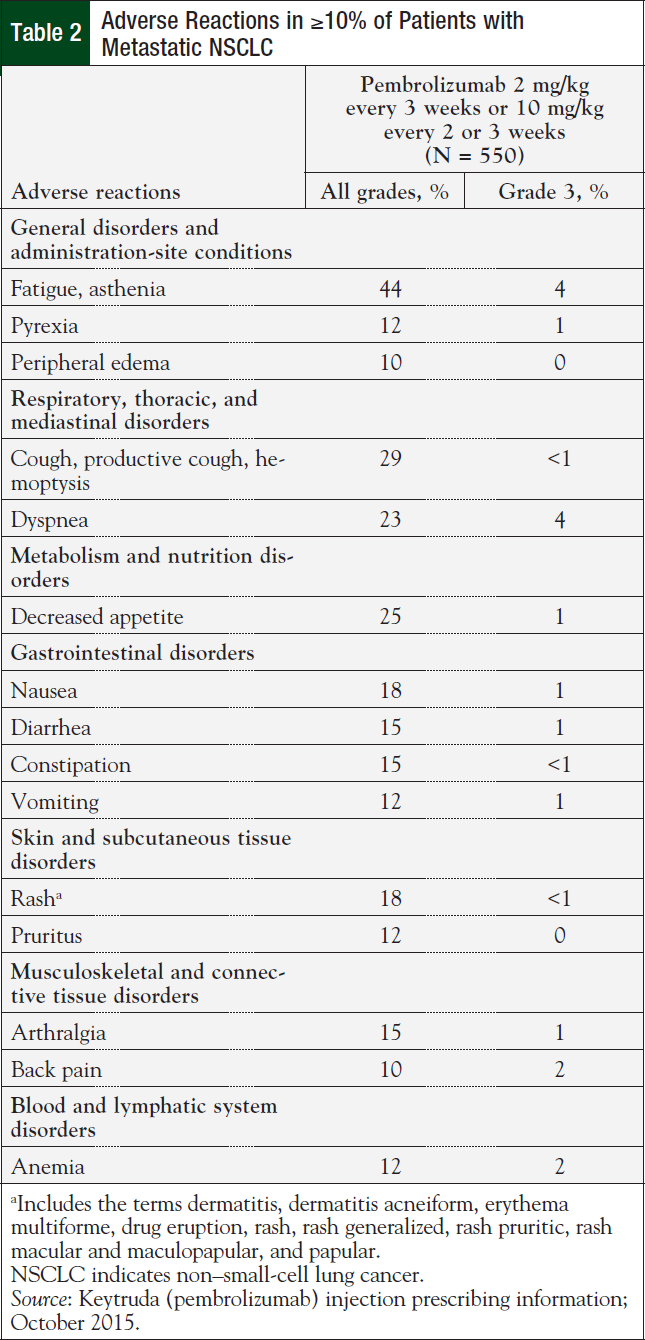
Among the 550 patients with previously treated metastatic NSCLC in KEYNOTE-001 who received pembrolizumab, the median duration of therapy was 2.8 months (range, 1 day to 25.6 months).10 Adverse reactions reported in at least 20% of patients included fatigue, decreased appetite, dyspnea, and cough (Table 2).10
Pembrolizumab was discontinued in 14% of patients because of adverse reactions.10
Overall, 38% of the 550 patients who received pembrolizumab experienced serious adverse reactions.10 The most frequent serious adverse reactions reported in ≥2% of patients were pleural effusion, pneumonia, dyspnea, pulmonary embolism, and pneumonitis; none of these reactions was reported as grade 4 or 5.10
Pembrolizumab has no contraindications.10
Warnings and Precautions
Immune-mediated pneumonitis. Pneumonitis occurred in 19 of 550 patients who received pembrolizumab. The median time to development of pneumonitis was 1.7 months, and the median duration was 1.2 months. Pneumonitis led to the discontinuation of pembrolizumab in 12 patients and completely resolved in 9 patients. In addition, 1 of 9 patients died of pneumonitis within 30 days of their last dose of pembrolizumab.10
Patients taking pembrolizumab should be monitored for pneumonitis. Corticosteroids should be administered if grade ≥2 pneumonitis is detected. Pembrolizumab should be withheld for moderate pneumonitis, and permanently discontinued for severe, life-threatening, or recurrent moderate pneumonitis.10
Immune-mediated colitis. Colitis occurred in 4 patients in KEYNOTE-001. The median time to onset of colitis was 1.6 months, and the median duration was 16 days. Overall, 1 patient permanently discontinued pembrolizumab because of colitis, and 3 of the 4 patients with colitis had complete resolution. Patients receiving pembrolizumab should be monitored for colitis. Corticosteroids should be administered for grade ≥2 colitis. Pembrolizumab should be withheld for moderate or severe colitis, and permanently discontinued for life-threatening colitis.10
Immune-mediated hepatitis. Patients receiving pembrolizumab should be monitored for changes in liver function. Corticosteroids should be administered for grade ≥2 hepatitis. Pembrolizumab should be withheld or discontinued based on the severity of liver enzyme elevations.10
Immune-mediated hypophysitis. Grade 3 inflammation of the pituitary gland occurred in 1 patient in KEYNOTE-001. The time to onset was 3.7 months, and the patient received systemic corticosteroids and physiologic hormone replacement to resolve hypophysitis. Patients receiving pembrolizumab should be monitored for hypophysitis. Pembrolizumab should be withheld for moderate hypophysitis, withheld or discontinued for severe hypophysitis, and permanently discontinued for life-threatening hypophysitis.10
Immune-mediated thyroid disorders. Hyperthyroidism was observed in 10 patients.10 The median time to onset was 1.8 months, and the median duration was 4.5 months.10 None of the 10 patients permanently discontinued pembrolizumab because of hyperthyroidism.10
Hypothyroidism occurred in 38 patients. The median time to onset was 4.2 months. No patient discontinued pembrolizumab because of hypothyroidism. Because thyroid disorders can occur at any time during treatment with pembrolizumab, patients should be monitored for changes in thyroid function at the start of treatment and periodically during treatment. Pembrolizumab should be withheld for severe hyperthyroidism, and permanently discontinued for life-threatening hyperthyroidism.10
Type 1 diabetes mellitus. Type 1 diabetes mellitus, including diabetic ketoacidosis, has been reported after the administration of pembrolizumab. Patients should be assessed for hyperglycemia or for other signs and symptoms of diabetes. Insulin should be administered for type 1 diabetes. Pembrolizumab should be withheld in patients with severe hyperglycemia.10
Renal dysfunction and immune-mediated nephritis. Patients receiving pembrolizumab should be monitored for changes in renal function. Corticosteroids should be administered for grade ≥2 nephritis. Pembrolizumab should be withheld for moderate nephritis, and permanently discontinued for severe or life-threatening nephritis.10
Other immune-mediated reactions. Other clinically important immune-mediated adverse reactions can occur while receiving pembrolizumab. Clinically significant, immune-mediated adverse reactions in <1% with pembrolizumab in the KEYNOTE-001 trial included rash, vasculitis, hemolytic anemia, serum sickness, and myasthenia gravis. Pembrolizumab should be permanently discontinued for any severe or grade 3 immune-mediated adverse reaction and for any life-threatening immune-mediated reaction.10
Infusion-related reactions. Severe infusion-related reactions, including life-threatening reactions, have occurred with pembrolizumab across studies and at different doses and schedules in patients with various solid tumors. Patients receiving pembrolizumab should be monitored for infusion reactions, including rigors, chills, wheezing, pruritus, flushing, rash, hypotension, hypoxemia, and fever. If severe or life-threatening reactions occur, the infusion should be stopped, and pembrolizumab should be permanently discontinued.10
Embryofetal toxicity. Pembrolizumab may cause fetal harm.10 If pembrolizumab is used during pregnancy, or if a patient becomes pregnant while taking it, she should be told of the potential hazard to the fetus.10 Women of childbearing age should use effective contraception during treatment with pembrolizumab and for at least 4 months after the last dose.10
Specific Populations
Pediatric patients. The safety and efficacy of pembrolizumab in pediatric patients have not been established.10
Geriatric use. Of the 550 patients with metastatic NSCLC who received pembrolizumab, 47% were aged ≥65 years.10 No meaningful differences in the efficacy or safety of pembrolizumab were observed between elderly patients and younger patients.10
Pregnancy. Pembrolizumab can cause fetal harm.10
Nursing mothers. Nursing should be discontinued during treatment with pembrolizumab and for 4 months after the last dose.10
Conclusion
Pembrolizumab, a PD-1 inhibitor, is an effective and safe alternative for patients with metastatic NSCLC whose disease has progressed after platinum-based chemotherapy and whose tumor expresses PD-L1. PD-L1 expression may represent a new biomarker for the treatment of patients with NSCLC.
Pembrolizumab was approved under the FDA accelerated approval process based on the overall patient response rate data. A multicenter, randomized clinical trial is under way to compare pembrolizumab (2 mg/kg) and standard therapy in patients with relapsed NSCLC as a condition of the drug’s accelerated approval. This clinical trial, known as P010, has 2 coprimary end points, progression-free survival and overall survival.9
The efficacy and safety of pembrolizumab are also being evaluated in other solid and liquid tumors, including small-cell lung cancer, ovarian cancer, renal-cell carcinoma, recurrent head and neck cancer, urothelial cancer, glioblastoma, and follicular lymphoma.14
References
- Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc. 2008;83:584-594.
- Uramoto H, Mitsudomi T. Which biomarker predicts benefit from EGFR-TKI treatment for patients with lung cancer? Br J Cancer. 2007;96:857-863.
- Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. 2010;363:1693-1703. Erratum in N Engl J Med. 2011;364:588.
- Brahmer JR, Pardoll DM. Immune checkpoint inhibitors: making immunotherapy a reality for the treatment of lung cancer. Cancer Immunol Res. 2013;1:85-91.
- Tseng WW, Malu S, Zhang M, et al. Analysis of the intratumoral adaptive immune response in well differentiated and dedifferentiated retroperitoneal liposarcoma. Sarcoma. 2015 Jan 29. Epub ahead of print.
- US FDA New and Events. FDA expands approved use of Opdivo to treat lung cancer. March 4, 2015. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm436534.htm.
- US FDA New and Events. FDA expands approved use of Opdivo in advanced lung cancer. October 9, 2015. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm466413.htm. Accessed October 23, 2015.
- Opdivo (nivolumab) [prescribing information]. Princeton, NJ: Bristol-Myers Squibb Company; October 2015.
- US FDA Drug Approvals and Databases. Pembrolizumab (NSCLC). http://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm465650.htm. Accessed October 23, 2015.
- Keytruda (pembrolizumab) [prescribing information]. Whitehouse Station, NJ: Merck Sharp & Dohme Corp; October 2015.
- Loftus P. FDA Approves Merck’s New-Wave Cancer Drug. The Wall Street Journal. September 4, 2014. http://www.online.wsj.com/articles/fda-approves-mercks-cancer-drug-1409856320. Accessed October 13, 2015.
- US FDA Drug Approvals and Databases. Pembrolizumab (Melanoma). www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm412861.htm. Accessed October 13, 2015.
- Garon EB, Rizvi NA, Hui R, et al. Pembrolizumab for the treatment of non–small-cell lung cancer. N Engl J Med. 2015;372:2018-2028.
- ClinicalTrials.gov. Pembrolizumab. http://clinicaltrials.gov/ct2/results?term=pembrolizumab&Search=Search. Accessed October 20, 2015.