Chronic myelogenous leukemia (CML) is a hematopoietic stem-cell disorder resulting from the formation of the Philadelphia (Ph) chromosome, which is the reciprocal translocation of chromosomes 9 (Abelson murine leukemia; ABL) and 22 (breakpoint cluster region; BCR).1,2 The resulting BCR-ABL fusion gene leads to enhanced activity of tyrosine kinase enzymes and reduced apoptosis of granulocytes.2 CML accounts for nearly 15% of adult leukemias in the United States.1 In 2013, an estimated 5920 new cases of CML will be diagnosed, with 610 deaths expected.3 High-dose radiation is the only known risk factor for CML, and no genetic predisposition has been identified.2
The clinical course of CML has 3 distinct phases, with the majority of patients presenting in the chronic phase (CP).1 As cellular differentiation becomes more impaired, CML progresses into the accelerated phase (AP) and the blast crisis (BC) phase. In BC-CML, ≥20% of cells in the peripheral blood or bone marrow are blasts, and the disease is aggressive and acute. Untreated CP-CML progresses to advanced phases within 3 to 5 years.1
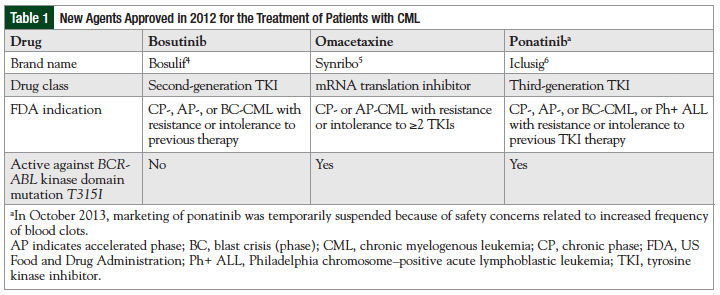
For most patients with CML, the initial treatment options can be one of the tyrosine kinase inhibitors (TKIs), including imatinib, nilotinib, or dasatinib; hematopoietic stem-cell transplantation; or enrollment in a clinical trial.1 Patients receiving a TKI who have partial to no cytogenetic response or cytogenetic relapse are candidates for therapy with an alternative TKI, other than imatinib.1 Resistance to first-line TKI treatment and a scarcity of therapeutic alternatives have led to the research and approval of 3 additional agents in 2012 for the treatment of CML, including bosutinib, omacetaxine mepesuccinate, and ponatinib (Table 1).4-6 However, in October 2013, the US Food and Drug Administration (FDA) temporarily suspended the marketing of ponatinib because of safety concerns related to increased frequency of blood clots.7
Omacetaxine, a first-in-class cephalotaxine, was approved by the FDA in 2012 for use in patients with CP-CML or AP-CML with resistance and/or intolerance to ≥2 TKIs.5 Omacetaxine mepesuccinate was approved according to the orphan drug process.
The safety and efficacy of omacetaxine and its place in therapy are the topics of this review, which involves a thorough discussion of the pharmacology, pharmacokinetics, clinical efficacy, indications for use, safety profile, and dosing and administration considerations of omacetaxine.
Pharmacology
Omacetaxine is an alkaloid compound derived from Cephalotaxus harringtonia, an evergreen tree that is native to Japan.8 Omacetaxine is an mRNA translation inhibitor. Specifically, omacetaxine inhibits the initial step in translation elongation by binding to the A site on the ribosome and preventing proper positioning of the incoming aminoacyl-tRNA, thereby preventing formation of the peptide bond by peptidyl transferase.9 Because omacetaxine does not target the ABL-kinase domain of BCR-ABL for its mechanism of action, it is an effective cytotoxic agent in wild-type BCR-ABL and in T315I-mutated BCR-ABL–expressing cells in vitro and in vivo.10
The rationale for using translation inhibitors as a therapeutic option stems from the reports of significantly elevated rates of translation in a variety of cancers, including CML, which has been found to contribute to the development and progression of the disease.11-13 The increased translation of short-lived mRNAs encode essential proteins involved in cell proliferation and survival.14 These short-lived mRNAs have guanine/cytosine–rich 5' untranslated regions with complex 3-dimensional structures compared with longer-lived mRNAs where the 5' untranslated regions are short and unstructured.14
In CML, the constitutively active BCR-ABL tyrosine kinase stimulates the phosphoinositide-3-kinase/Akt/mammalian target of rapamycin (mTOR)-signaling pathways. mTOR can stimulate translation by regulating the assembly and the activity of eukaryotic initiation factor 4F (eIF4F). eIF4F preferentially recognizes and translates short-lived mRNAs, including the proto-oncoproteins cyclin D1 and c-Myc, prosurvival proteins, as well as angiogenic proteins, such as vascular endothelial growth factor, fibroblast growth factor, and matrix metalloproteinase.13 C-Myc is a transcription factor that regulates the expression of even more genes involved in the cell cycle, transcription, translation, and protein folding.15
By inhibiting translation, omacetaxine has been shown to reduce the protein levels of BCR-ABL, as well as several of the short-lived proteins previously mentioned.10,16 The preferential reduction in translation of these short-lived mRNAs, resulting in apoptosis, is thought to be an important underlying mechanism in the therapeutic effect of omacetaxine on myeloid leukemia cells.
CML is a disease originating from pluripotent BCR-ABL–positive hematopoietic stem cells.17 This subset of stem cells harboring the Ph chromosome, referred to as leukemia-initiating cells (LICs), is responsible for driving tumorigenesis and maintenance of the disease, at least in CP-CML. The expansion of mature myeloid cells in the bone marrow and peripheral blood originates from the LIC population. Evidence suggests that as the disease progresses, granulocyte macrophage progenitors can become a second population of LICs.17 TKIs, such as imatinib, are effective at killing the more differentiated myeloid cells, but not the LICs.16,17 Therefore, in patients with CML, even in those with complete molecular remission, the disease most often relapses if the use of TKIs is discontinued.18 Unlike TKIs, omacetaxine is cytotoxic to the mature myeloid cells, as well as to the LICs and, therefore, is a likely contributing mechanism to its clinical effectiveness.10,16
The mechanisms explaining the differential cytotoxic effects of TKIs and omacetaxine on LICs have not been elucidated. However, in addition to a generalized difference in the gene-expression profile between imatinib and omacetaxine,19 several specific hypotheses exist.
First, the cytotoxic effects of omacetaxine on LICs may involve the downregulation of cytokine receptors.20 Various cytokines have been shown to protect LICs from the action of TKIs via the activation of prosurvival mitogenic-signaling pathways.20 Omacetaxine has been shown to decrease the expression of the common beta-subunit c of the cytokine receptors for interleukin-3, interleukin-5, and granulocyte-macrophage colony-stimulating factor, which renders LICs insensitive to the prosurvival effects of these cytokines.20
Second, the differential effects of these 2 drug classes (ie, omacetaxine and TKIs) may theoretically involve omacetaxine-induced downregulation of beta-catenin, a short-lived proto-oncoprotein that is central to the Wnt signaling pathway.21 The expression of beta-caten- in has been shown to be essential for the survival of LICs in mice with CML that are insensitive to TKIs.22
Furthermore, it has been shown that the self-renewal of LICs is dependent on beta-catenin expression; therefore, LIC self-renewal is impaired in beta-catenin–conditional null mice, mice that lack expression of beta-catenin predominantly in the hematopoietic system.23 In addition, mice exhibited increased resistance to developing CML when transplanted with beta-
catenin –/– BCR-ABL–expressing bone marrow cells.23
Therefore, beta-catenin has proved to be central to the survival and renewal of LICs. Omacetaxine has been reported to decrease the expression of beta-catenin in myeloma cell lines; however, it is not yet known if omacetaxine exerts the same effect on LICs.21 Clearly, further research is needed to identify mechanisms that explain the disparity between the cytotoxic effects of TKIs and omacetaxine’s effect on LICs.
Pharmacokinetics
Clinical pharmacokinetic data after 11 days of the administration of subcutaneous omacetaxine suggest that peak drug concentrations are evident at 30 to 35 minutes after administration.5,24 The absolute bioavailability of omacetaxine is unknown. Higher mean Cmax concentrations were observed on day 11 compared with day 1, which may suggest drug accumulation.25 Omacetaxine has a steady-state volume of distribution of 141 L ± 93.4 L.5 As much as 50% or less of omacetaxine binds to plasma proteins.5 Omacetaxine is hydrolyzed by esterases to 2 inactive metabolites: 4'-desmethylhomoharringtonine (4'-DMHHT) and cephalotaxine.
The primary elimination route of omacetaxine is unknown, with excretion of omacetaxine, 4'-DMHHT, and cephalotaxine comprising 12% to 15%, 4% to 5%, and 0.07% to 0.14%, respectively, of the administered dose.24 The half-lives of omacetaxine and of 4'-DMHHT are 7 and 16 hours, respectively.24
The pharmacokinetics of omacetaxine have not been determined in patients with renal or hepatic impairment.5
With respect to potential drug interactions, omacetaxine is not a substrate or inhibitor of CYP450 enzymes; its potential for enzyme induction is not known.5 Omacetaxine is a P-glycoprotein substrate. Experience with omacetaxine so far has not identified any clinically relevant drug interactions.5
Clinical Trials
The Cephalotaxus harringtonia (homoharringtonine) compound has been studied in leukemias since the 1970s.26 In several clinical trials of patients with CP-CML between 1997 and 2003, participants who received homoharringtonine with interferon-alpha and/or cytarabine achieved complete hematologic responses of more than 80% and major cytogenetic responses (MCyRs) ranging from 17% to 84%.26 Around the time of these clinical trials of patients with CP-CML, imatinib was approved (ie, in 2001) and was quickly established as first-line therapy for patients with CML.
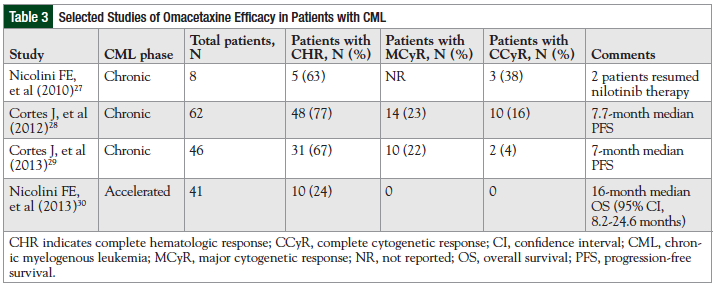
Further development of homoharringtonine had been delayed until recent interest in its potential role for CML in the setting of TKI failures. At this time, to our knowledge, clinical data on omacetaxine in the posthomoharringtonine era are primarily available as 1 case series,27 2 published phase 2 studies,28,29 1 letter to the editor,30 and study updates in abstract form. The disease response criteria used in these studies were consistent with published guidelines on CML (Table 2).1

A report of 8 patients with CP-CML resistant to imatinib describes the use of omacetaxine in patients with CP-CML and the T315I mutation.27 Of these 8 patients, 4 had received nilotinib or dasatinib, and 5 were in hematologic relapse at baseline. The patients had been withdrawn from previous therapy for a median of 2 months (range, 1-9 months). Omacetaxine was administered with an induction course of 1.25 mg/m2 subcutaneously twice daily for 14 days of every 28 days until hematologic response was achieved. Maintenance therapy included the same regimen but reduced to 5 to 7 days every 28 days.
A median of 4 cycles was necessary to reduce the T315I/ABL ratio to below 1%. Complete hematologic response was achieved or maintained in 5 patients, and complete cytogenetic response (CCyR) was achieved in 3 patients. In 2 patients, the T315I mutation became undetectable, but BCR-ABL transcripts were still elevated. The 2 patients were rechallenged with nilotinib for a median of 12 months, and 1 patient obtained CCyR.27
A multicenter phase 2 study was performed to evaluate the use of omacetaxine in patients with CP-CML and the T315I mutation who had failed 1 or more TKIs.28 Of the patients, 62 had discontinued other CML therapies for at least 2 weeks before starting treatment with omacetaxine. Omacetaxine was administered with an induction course of 1.25 mg/m2 subcutaneously twice daily for 14 days of every 28 days until hematologic response was achieved. Maintenance therapy with the same regimen was decreased to 7 days of every 28 days. The primary efficacy end points were the proportion of patients achieving complete hematologic response and MCyR. All patients (median age, 56 years) had previously received imatinib, with 18 (29%) patients having achieved MCyR. A total of 46 (74%) patients had failed treatment with 2 or more TKIs.28 At baseline, 15 (24%) patients were in complete hematologic response. A median of 7 treatment cycles (range, 1-41 cycles) were administered, and 28 (45%) patients discontinued therapy as a result of disease progression or lack of response.
Efficacy response rates are shown in Table 3. Complete hematologic response was achieved with a median of 1 cycle (range, 1-5) and a duration of 9.1 months (range, 2-46+ months). The median progression-free survival was 7.7 months (95% confidence interval [CI], 5.8-11 months). Of 46 evaluable patients, 17 (37%) had a 50% to 100% reduction from baseline of the T315I-mutated clone.28 The first-generation TKIs have not demonstrated activity against the T315I mutation.
Although studies of omacetaxine in patients with CML and the T315I mutation show a potential promising treatment option in patients whose disease is resistant to other therapies, omacetaxine has not been approved by the FDA specifically for patients with the mutation. A validated assay for the evaluation of T315I mutation status is necessary.
The safety and efficacy of omacetaxine in 46 patients with CP-CML that is resistant or intolerant to 2 or more TKIs were evaluated in a phase 2 clinical trial.29 The investigators defined resistance as progressive leukocytosis or lack of complete hematologic response, CCyR, or MCyR at 12, 24, or 52 weeks, respectively. Omacetaxine was administered with the identical induction and maintenance regimen used in the previously described phase 2 trial.28 The primary end point for this trial was the number of patients achieving complete hematologic response for 8 weeks or MCyR.29 The patients’ median time since CML diagnosis was 6.2 years.
All patients had received imatinib, and 85% of the patients had received 2 or more TKIs. At baseline, 17% of patients were in complete hematologic response. Patients received a median of 4.5 treatment cycles (range, 1-36 cycles). Among patients not in complete hematologic response at baseline, 61% achieved complete hematologic response. A total of 10 patients achieved MCyR. The median follow-up time was 19.1 months (range, 0.3-35.3 months); the investigators estimated the median overall survival (OS) to be 30.1 months (95% CI, 20.3 months-not reached).
Of note, mutational analyses of the BCR-ABL kinase domain were performed in 33 patients with the most common mutations, F395V (N = 4) and V299L (N = 3).29
Omacetaxine was also evaluated in 41 patients with AP-CML who had been enrolled in the CML-202 and CML-203 studies.30 The investigators defined AP-CML in consistency with the National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines® Version 2.2014.1 Omacetaxine was administered using the identical regimen described in the phase 2 clinical trials.28,29 All patients had failed 2 TKIs, and 59% of the patients failed 3 TKIs. The reasons for TKI failure included disease resistance, intolerance, or resistance to one TKI and intolerance to another TKI in 88%, 7%, and 5% of patients, respectively. Two patients had received stem-cell transplantation. In the AP-CML setting, patients received a median of 2 treatment cycles (range, 1-29 cycles). In total, 11 (27%) patients achieved major hematologic response with a median duration of 9 months (95% CI, 3.6-14.1 months). Minor cytogenetic response was observed in 15% of patients, but MCyR and CCyR were not achieved.30
Various analyses of clinical data that led to the FDA approval of omacetaxine for patients with CML are available in abstract form, and additional data are on file with the manufacturer. A subset of the phase 2 study data evaluating the use of omacetaxine in patients with CP-CML categorized them by TKI resistance, which included 69 patients who never achieved or who lost response to 2 or more TKIs, 7 patients with disease intolerant to the drugs, and 5 patients with disease resistant and intolerant of them.31 After treatment with omacetaxine, 19%, 29%, and 20% of the patients with resistance, intolerance, and resistance plus intolerance, respectively, achieved MCyR. Of note, the median OS times were 33.9 months and 25 months in the resistance patients and resistance and intolerance patients, respectively. OS had not yet been reached in the intolerance subset.30
Safety and Tolerability
The most common adverse events associated with omacetaxine were primarily hematologic abnormalities, infection, diarrhea, nausea, fatigue, and asthenia. Grade 3 or 4 adverse reactions most frequently reported in the phase 2 studies with omacetaxine are summarized in Table 4.
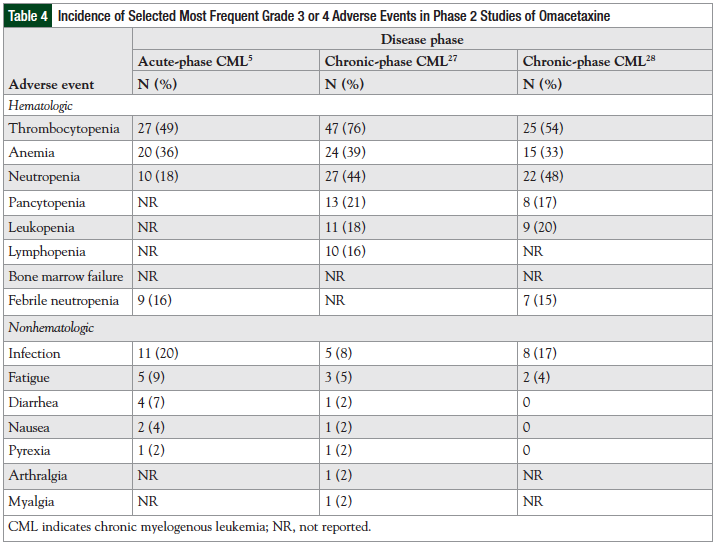
In the phase 2 study of omacetaxine in patients with CP-CML and T315I mutations, therapy was generally well tolerated.28 Blood count nadir occurred within 2 to 3 weeks after the first dose of the cycle, with recovery at 1 to 3 weeks later. Thrombocytopenia and neutropenia were the most common reasons for treatment delay, with dose delays occurring most frequently after cycles 2 and 3. Filgrastim and erythropoietin-stimulating agents were administered to 13% and 21% of patients, respectively.
Hematologic adverse events were managed by reducing the number of days that omacetaxine was administered in subsequent cycles, which decreased the incidence of grade 3 or 4 toxicities by 10% to 20%.28 Erythema from injection-site reactions was reported in 21%28 and 17%29 of patients in these phase 2 trials. In 1 of the phase 2 trials, 6 deaths occurred during the study or 30 days after the study’s completion.29 The investigators rated 1 of these deaths as probably related to omacetaxine, because the patient had pancytopenia that never resolved.29
The safety of omacetaxine was evaluated in 21 patients with advanced hematologic malignancies or solid tumors.24 A total of 13 (62%) patients received 1 cycle of omacetaxine 1.25 mg/m2 subcutaneously twice daily for 14 days, and 2 patients completed at least 2 cycles. No patients discontinued therapy as a result of toxicity. The most common adverse events were hematologically related, with 17 patients experiencing at least 1 grade 3 or 4 event. Of the serious adverse events, the most common were thrombocytopenia (48%) and neutropenia (33%).24
When omacetaxine was evaluated in patients with AP-CML, hematologic toxicities were reported in 78% of patients, necessitating the administration of granulocyte-stimulating factors, erythropoietin, red blood cells, and platelet transfusions in 5%, 17%, 76%, and 59% of the patients, respectively.30 The most common nonhematologic toxicities in this setting included infection, diarrhea, fatigue, and asthenia.30
In addition to these toxicities, omacetaxine may induce alterations in glycemic control. One patient developed hyperosmolar nonketotic hyperglycemia.5 It is recommended to closely monitor for grade 3 or 4 hyperglycemia, especially in patients with diabetes or those with risk factors for diabetes.1 Patients with uncontrolled diabetes should have their blood glucose optimized before starting omacetaxine therapy.5
Dosing Recommendations
Omacetaxine is indicated for induction and maintenance therapy in the management of patients with CP-CML or AP-CML.5 For induction, omacetaxine is administered as 1.25 mg/m2 subcutaneously twice daily for 14 consecutive days every 28 days. The 28-day cycle is repeated every 28 days until patients achieve a hematologic response. After induction, a maintenance dose (1.25 mg/m2) is administered subcutaneously twice daily for 7 consecutive days every 28 days of the 28-day cycle, continuing until patients no longer experience clinical benefit from the treatment. While short intravenous infusions were administered in phase 1 clinical trials with omacetaxine, dose-limiting and life-threatening cases of hypotension and tachycardia were observed.24,26 Subcutaneous dosing of omacetaxine has been shown to decrease these adverse effects.24
Weekly monitoring of complete blood count is recommended during induction and during initial maintenance cycles of omacetaxine, and then every 2 weeks or as otherwise indicated.5 Hematologic toxicities, such as neutropenia (absolute neutrophil count <0.5 × 109/L) or thrombocytopenia (platelet count <50 × 109/L), warrant the delay of a treatment cycle with omacetaxine or the reduction in the number of doses during the cycle.1,5
The studies of omacetaxine therapy did not include patients with renal or hepatic impairment; therefore, dosing recommendations in these patient populations are not available. Although the primary elimination route of omacetaxine is not known, metabolites comprise <15% of the administered dose.5 Therefore, dose adjustments in renal impairment may not be necessary.
Omacetaxine should be prepared and administered by a healthcare provider, which would entail patient visits to a clinic or a hospital.5 Delay in therapy can occur if infusion centers are not open for weekends or extended hours. There is a short time period of stability once omacetaxine has been prepared: 12 hours at room temperature and 24 hours if refrigerated.5
Place in Therapy
Omacetaxine offers an alternative treatment for patients with CML. The NCCN Clinical Practice Guidelines® for the treatment of CML suggest omace- taxine as a treatment option for patients who cannot tolerate TKI therapy or who have experienced disease progression secondary to resistance with the treatment of at least 2 TKIs.1 Omacetaxine is acknowledged in these guidelines as a treatment option for patients with several mutations, including T315I, T315A, F317L/V/I/C, Y253H, E255K/V, F359V/C/I, and V299L.1 Any other mutation is also an indication for treatment with omacetaxine.1
In the case of T315I mutations, ponatinib had been designated as preferred over omacetaxine.1 The safety and efficacy of ponatinib were evaluated in the Ponatinib Ph+ ALL and CML Evaluation (PACE) trial, which included patients resistant to or intolerant of other TKIs, or those with T315I mutation.1,32 In 64 patients with CP-CML and T315I mutation, 70% achieved MCyR.
Major hematologic responses were also observed in patients with BC-CML or AP-CML and T315I mutation.32 Ponatinib’s prescribing information contains a black box warning about the potential for the development of arterial thrombosis or hepatotoxicity with this drug.6 Omacetaxine may provide an alternative for patients susceptible to those conditions, especially given the recent suspension of ponatinib marketing by the FDA,7 as noted earlier.
Bosutinib, a second-generation TKI, was approved by the FDA for all phases of patients with CML resistant or intolerant to previous TKI therapy. Its approval was based on an analysis of 118 patients with CP-CML who had received imatinib and had developed resistance or intolerance to it, and had also received dasatinib and/or nilotinib.33 In this heavily pretreated population, treatment with bosutinib for a median of 28.5 months led to rates of 32% MCyR and 73% complete hematologic response. Adverse effects were primarily grades 1 and 2 diarrhea, vomiting, and rash.33
These results suggest a very modest improvement in efficacy and a more tolerable safety profile compared with omacetaxine; however, these 2 agents have not been compared directly. Omacetaxine may provide a non-TKI alternative therapy to bosutinib.
National guideline recommendations for omacetaxine are based on data in very limited numbers of patients, as a result of the relative rarity of CML and the urgency to provide therapeutic alternatives for a disease with limited cure potential. Omacetaxine is indicated for CP-CML and AP-CML; however, full data on patients with AP-CML are not available. Published peer-reviewed data supporting omacetaxine efficacy in patients with specific BCR-ABL kinase domain mutations, or comparative data with other therapeutic alternatives are also lacking. In addition, pharmacoeconomic and quality-of-life analyses are lacking.
Although treatment with omacetaxine extends complete hematologic response in more than 60% of patients, it is still not clear whether its risks outweigh its benefits. There may be an OS benefit with omace- taxine, but the overall quality-of-life benefit has not been established. In the majority of patients, adverse events have been of mild-to-moderate severity; more clinical experience with omacetaxine will help to better elucidate its safety profile and will help to target which patients will benefit most from this therapy, with minimal risk.
Another consideration for providers and payers is the cost of omacetaxine. Experts in CML drugs, including the investigators of omacetaxine studies, published an open editorial to drug manufacturers discussing the cost of cancer drugs and the debate about healthcare costs.34 The cost of omacetaxine is specifically cited in the editorial as $28,000 for an induction cycle and $14,000 for the course of maintenance. If a patient requires only 1 induction cycle, the annual cost becomes $182,000 ($28,000 induction plus $154,000 maintenance); most patients in the clinical trials reported required multiple induction cycles, which would be even more than $182,000 annually. In the open editorial, bosutinib is quoted at an annual cost of $118,000.34
Additional costs that are not considered in this calculation include costs for administration time, hospital or clinic charges, and costs that are involved with supportive care measures, such as transfusions or colony-stimulating factors. Clearly, more research is necessary to determine the clinical and cost benefits of omacetaxine.
Conclusion
Omacetaxine, the first-in-class mRNA translation inhibitor, offers a therapeutic alternative to TKIs, with a novel mechanism of action, for the treatment of patients with drug-resistant CML. The majority of adverse reactions reported in clinical trials with this drug include hematologic abnormalities, infection, diarrhea, and nausea, and they were mild to moderate in severity in patients with CP-CML. Omacetaxine provides another treatment option for patients who are resistant to or intolerant to first-line TKIs, but its place in therapy compared with bosutinib or ponatinib is not known. Many questions remain concerning its niches in therapy, adverse effect profile, and overall benefit.
Author Disclosure Statement
Dr Olin, Dr Cole, and Dr Dirks-Naylor reported no conflicts of interest.
References
- National Comprehensive Cancer Network. National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN Guidelines®): chronic myelogenous leukemia. Version 2.2014. November 1, 2013. www.nccn.org/profes sionals/physician_gls/pdf/cml.pdf. Accessed November 25, 2013.
- Druker BJ, Lee SJ. Chronic myelogenous leukemia. In: DeVita VT, Lawrence TS, Rosenberg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer: Principles & Practice of Oncology. 9th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2011:1962-1972.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11-30.
- Bosulif (bosutinib) tablets [prescribing information]. New York, NY: Pfizer Laboratories; September 2013.
- Synribo (omacetaxine mepesuccinate) for injection [prescribing information]. North Wales, PA: Teva Pharmaceuticals USA, Inc; October 2012.
- Iclusig (ponatinib) tablets [prescribing information]. Cambridge, MA: ARIAD Pharmaceuticals, Inc; December 2012.
- US Food and Drug Administration. FDA drug safety communication: FDA asks manufacturer of the leukemia drug Iclusig (ponatinib) to suspend marketing and sales. November 5, 2013. Updated November 12, 2013. www.fda.gov/Drugs/DrugSafety/ucm373040.htm. Accessed November 25, 2013.
- Chen Y, Peng C, Sullivan C, et al. Novel therapeutic agents against cancer stem cells of chronic myeloid leukemia. Anticancer Agents Med Chem. 2010;10:111-115.
- Gürel G, Blaha G, Moore PB, Steitz TA. U2504 determines the species specificity of the A-site cleft antibiotics: the structures of tiamulin, homoharringtonine, and bruceantin bound to the ribosome. J Mol Biol. 2009;389:146-156.
- Chen Y, Hu Y, Michaels S, et al. Inhibitory effects of omacetaxine on leukemic stem cells and BCR-ABL-induced chronic myeloid leukemia and acute lymphoblastic leukemia in mice. Leukemia. 2009;23:1446-1454.
- Dong Z, Zhang JT. Initiation factor eIF3 and regulation of mRNA translation, cell growth, and cancer. Crit Rev Oncol Hematol. 2006;59:169-180.
- De Benedetti A, Graff JR. eIF-4E expression and its role in malignancies and metastases. Oncogene. 2004;23:3189-3199.
- Yin JY, Dong Z, Liu ZQ, Zhang JT. Translational control gone awry: a new mechanism of tumorigenesis and novel targets of cancer treatments. Biosci Rep. 2011;31:1-15.
- Robert F, Carrier M, Rawe S, et al. Altering chemosensitivity by modulating translation elongation. PLoS One. 2009;4:e5428.
- Menssen A, Hermeking H. Characterization of the c-MYC-regulated transcriptome by SAGE: identification and analysis of c-MYC target genes. Proc Natl Acad Sci U S A. 2002;99:6274-6279.
- Wetzler M, Segal D. Omacetaxine as an anticancer therapeutic: what is old is new again. Curr Pharm Des. 2011;17:59-64.
- Stuart SA, Minami Y, Wang JY. The CML stem cell: evolution of the progenitor. Cell Cycle. 2009;8:1338-1343.
- Rousselot P, Huguet F, Rea D, et al. Imatinib mesylate discontinuation in patients with chronic myelogenous leukemia in complete molecular remission for more than 2 years. Blood. 2007;109:58-60.
- Kulkarni H, Göring HH, Diego V, et al. Association of differential gene expression with imatinib mesylate and omacetaxine mepesuccinate toxicity in lymphoblastoid cell lines. BMC Med Genomics. 2012;5:37.
- Klag T, Härtel N, Erben P, et al. Omacetaxine mepesuccinate prevents cytokine-dependent resistance to nilotinib in vitro: potential role of the common β-subunit c of cytokine receptors. Leukemia. 2012;26:1321-1328.
- Kuroda J, Kamitsuji Y, Kimura S, et al. Anti-myeloma effect of homoharringtonine with concomitant targeting of the myeloma-promoting molecules, Mcl-1, XIAP, and beta-catenin. Int J Hematol. 2008;87:507-515.
- Hu Y, Chen Y, Douglas L, Li S. Beta-catenin is essential for survival of leukemic stem cells insensitive to kinase inhibition in mice with BCR-ABL-induced chronic myeloid leukemia. Leukemia. 2009;23:109-116.
- Zhao C, Blum J, Chen A, et al. Loss of beta-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell. 2007;12:528-541.
- Nemunaitis J, Mita A, Stephenson J, et al. Pharmacokinetic study of omacetaxine mepesuccinate administered subcutaneously to patients with advanced solid and hematologic tumors. Cancer Chemother Pharmacol. 2013;71:35-41.
- ChemGenex Pharmaceuticals, Inc. Briefing book. Oncology Drugs Advisory Committee Meeting NDA 22-374: Omapro (omacetaxine mepesuccinate). www.fda.gov/downloads/advisorycommittees/committeesmeetingmaterials/drugs/oncologicdrugsadvisorycommittee/ucm199562.pdf. Accessed August 28, 2013.
- Quintás-Cardama A, Kantarjian H, Cortes J. Homoharringtonine, omace- taxine mepesuccinate, and chronic myeloid leukemia circa 2009. Cancer. 2009;115:5382-5393.
- Nicolini FE, Chomel JC, Roy L, et al. The durable clearance of the T315I BCR-ABL mutated clone in chronic phase chronic myelogenous leukemia patients on omacetaxine allows tyrosine kinase inhibitor rechallenge. Clin Lymphoma Myeloma Leuk. 2010;10:394-399.
- Cortes J, Lipton JH, Rea D, et al; for the Omacetaxine 202 Study Group. Phase 2 study of subcutaneous omacetaxine mepesuccinate after TKI failure in patients with chronic-phase CML with T315I mutation. Blood. 2012;120:2573-2580.
- Cortes J, Digumarti R, Parikh PM, et al; for the Omacetaxine 203 Study Group. Phase 2 study of subcutaneous omacetaxine mepesuccinate for chronic- phase chronic myeloid leukemia patients resistant to or intolerant of tyrosine kinase inhibitors. Am J Hematol. 2013;88:350-354.
- Nicolini FE, Khoury HJ, Akard L, et al. Omacetaxine mepesuccinate for patients with accelerated phase chronic myeloid leukemia with resistance or intolerance to two or more tyrosine kinase inhibitors. Haematologica. 2013;98:e78-e79.
- Akard LP, Kantarjian H, Nicolini FE, et al. Omacetaxine mepesuccinate in chronic-phase chronic myeloid leukemia (CML) in patients resistant, intolerant, or both to two or more tyrosine-kinase inhibitors (TKIs). J Clin Oncol. 2012;30(15 suppl). Abstract 6596.
- Cortes JE, Kim DW, Pinilla-Ibarz J, et al; for the PACE Study Group. A pivotal phase 2 trial of ponatinib in patients with chronic myeloid leukemia (CML) and Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL) resistant or intolerant to dasatinib or nilotinib, or with the T315I BCR-ABL mutation: 12-month follow-up of the PACE trial. Blood (ASH Annual Meeting Abstracts). 2012;120. Abstract 163.
- Khoury HJ, Cortes JE, Kantarjian HM, et al. Bosutinib is active in chronic phase chronic myeloid leukemia after imatinib and dasatinib and/or nilotinib therapy failure. Blood. 2012;119:3403-3412.
- Experts in Chronic Myeloid Leukemia. The price of drugs for chronic myeloid leukemia (CML) is a reflection of the unsustainable prices of cancer drugs: from the perspective of a large group of CML experts. Blood. 2013;121:4439-4442.