Background: Medication therapy management (MTM) has been shown to improve patient outcomes. Patients with cancer are at high risk for adverse events caused by the toxicity and complexity of their medication regimens, and they may benefit from MTM services.
Objectives: The primary objective was to document drug therapy problems, using an MTM model in an ambulatory oncology setting. A second objective was to evaluate the potential revenue from these services.
Methods: MTM consults were conducted by 2 oncology pharmacists between January 1, 2010, and December 31, 2010, at Hennepin County Medical Center, Minneapolis, MN. A retrospective chart review of patient care notes was performed to collect data on drug therapy problems that were identified and resolved during the patient encounters.
Results: A total of 107 patients were seen during 121 encounters. The sample included 60 females and 47 males. Most MTM billing claims were submitted to Medicaid (32%) or Medicare (27%), resulting in approximately $101 of potential revenue per visit. During an average of 33 minutes spent per encounter, 322 drug therapy problems were recognized (averaging 2.7 drug therapy problems per patient), of which 281 drug therapy problems were resolved in the original encounter. Because most patients were initially seen for chemotherapy teaching, the most prevalent category of drug therapy problem identified was Compliance/ Complex Regimen.
Conclusion: It is feasible to incorporate the MTM model into current oncology pharmacy practice in the ambulatory setting. Pharmacists are able to submit billing claims for the service; as of yet, the revenue is undetermined.
Oncology pharmacists specialize in the use of chemotherapeutic agents. These pharmacists practice in acute and ambulatory care settings to review chemotherapy treatment plans for accuracy and safety, provide clinical services to patients with cancer, and coordinate orders and prescriptions for chemotherapy and supportive medications. Oncology pharmacists also play an integral role in the formulary review of new chemotherapeutic agents, as well as in medication safety and proper handling of chemotherapeutic agents.1 Recently, the provision of medication therapy management (MTM) visits has been incorporated into current practice at Hennepin County Medical Center (HCMC), Minneapolis, MN, allowing for additional patient interaction in the outpatient setting.
MTM is defined by the American Pharmacists Association as “a term used to describe a broad range of health care services provided by pharmacists, the medication experts on the health care team.”2 On average, patients with cancer have 3 comorbidities and are taking 9.1 medications for cancer treatment, supportive care, and comorbid conditions.3 The use of multiple drug therapies increases the risk of altered pharmacokinetics and adverse drug reactions. Furthermore, because of prolonged treatment periods and the increased toxicity of antineoplastic agents, patients with cancer are prone to complications and decreased adherence.4
Face-to-face interaction with oncology pharmacists allows for medication reconciliation and disease state management, as well as for patient and family education to prevent nonadherence. Drug therapy problems can be identified, helping to ensure that the patient’s medication regimen is safe, minimally toxic, and appropriately effective to treat the patient’s oncologic condition. There is a need for research on MTM outcomes in the ambulatory oncology setting. To advance the incorporation of the MTM model into oncology pharmacy practice, we must document the feasibility and potential added value of performing MTM in this setting.
The purpose of the current study is to describe an ambulatory oncology MTM practice and measure its impact, based on the identification and resolution of drug therapy problems. A secondary outcome is to report the amount billed per patient encounter, which represents potential revenue added by MTM services.
Methods
A retrospective chart review was completed on all patients who underwent an MTM visit with an oncology pharmacist at HCMC between January 1, 2010, and December 31, 2010. There were no exclusion criteria, and all patients who had been seen during the study period were included in the analysis. Patients were referred to the pharmacists by oncologists or nurses from HCMC for medication review or for new patient teaching of chemotherapy and supportive care. HCMC is a Level 1 Trauma Center in Minneapolis, MN, and is considered a “safety-net” hospital, which provides care for low-income, uninsured, and vulnerable populations. The infusion room within the Hennepin Comprehensive Cancer Center administers chemotherapy to between approximately 6 and 12 patients daily, Monday through Friday.
Each patient encounter was documented and billed in the electronic health record, using the EpicCare EMR (Epic software; Verona, WI), and was also documented in an Excel spreadsheet for internal quality improvement purposes by 1 of the 2 oncology pharmacists. Data gathered in the spreadsheet included billing level, encounter time, patient insurance, drug therapy problems noted, oncologic disease, and reason for the encounter. The pharmacists also noted the patient name, medical record number, doctor, visit date, pharmacist name, and follow-up instructions. In addition to the spreadsheet data, the investigator reviewed all pharmacist notes for details on drug therapy problems and interventions.
The primary outcome was measured by the number of drug therapy problems identified and recorded per patient encounter. Because of the many drug therapy problems available in the EpicCare EMR electronic health record oncology-specific drug therapy problem categories were created to ease the documentation process and highlight the most common drug therapy problems found in this patient population. The drug therapy problems were categorized as Indication/Prevention, Compliance/Complex Regimen, Drug Interaction, and Drug Safety Concern. Categories used to document drug therapy problems were used interchangeably at times because of similarities within the 4 of them and the complexity of drug therapy problems identified.
Indication/Prevention focused on the need for additional drug therapy or the identification of unnecessary drug therapy. Additional drug therapy was suggested if a condition, such as nausea, was not addressed in the medication regimen, if there were preventive or prophylactic medications available, if a synergistic medication could be added, or if the treatment goal was not being achieved. Unnecessary drug therapy included the identification and discontinuation of medications with no medical indication and drug (eg, marijuana) use for recreation. Duplicate therapy, as well as medications that were being used for the treatment of avoidable adverse drug reactions, were also considered unnecessary.
Compliance/Complex Regimen identified drug therapy problems within complex medication regimens, which can represent patient barriers to medication adherence. Drug therapy problems in this category pertained to insurance coverage, patient ability to afford the drug, and patient willingness to take the medication. In addition, pharmacists looked at the ability of the patient to take the medication by its specified route and to read and understand administration directions, and the pharmacists identified the need for adherence tools.
Drug Interaction and Drug Safety Concern identified any drug therapy problems that involved the safety of the patient. Drug safety focused on the prevention of serious side effects and included making dose adjustments when needed, modifying dosing schedules, identifying possible drug–drug interactions, and making laboratory or drug monitoring recommendations. Drug therapy problems in the Drug Safety Concern category were identified and included allergic reactions, incorrect administration, complex regimens requiring stepwise increases or taper, and medications with undesirable effects.
A secondary outcome was potential revenue gained from each MTM encounter and was measured by compiling the amount charged to the patient’s insurance. Billing levels were based on a predetermined amount dependent on the time spent and complexity of the encounter—taking into consideration the number of medications the patient is currently taking, the number of medical conditions present, the number of drug therapy problems that are identified, and the face-to-face time of the encounter—as specified by the Minnesota Health Care Programs (MHCPs). Billing level and insurance, if applicable, were documented in the EpicCare EMR patient care note, as well as in the Excel spreadsheet.
Data collected by the 2 pharmacists were reviewed by the investigator, a pharmacist intern at HCMC, and analyzed using descriptive analyses (including numbers, percentages, and averages). This study was approved by the University of Minnesota Institutional Review Board, as well as by the Minneapolis Medical Research Foundation Institutional Review Board.
Results
Over the course of 1 year, 107 patients were seen by a pharmacist in 121 patient encounters. The demographic characteristics of the patient population are displayed in Table 1. A total of 60 females and 47 males were included (average age, 58.6 years). Nearly 80% of patients were aged between 46 and 79 years. Breast cancer (16%) was the most common oncologic disease represented, followed by lymphoma (12%).
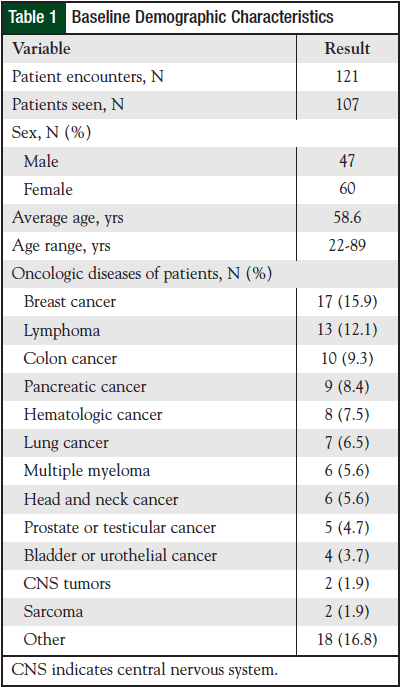
A range of insurance plans, displayed in Table 2, were charged for MTM services. In the Other category, 4 encounters (3.3%) were documented, which were billed to Metropolitan Health Plan or MinnesotaCare.
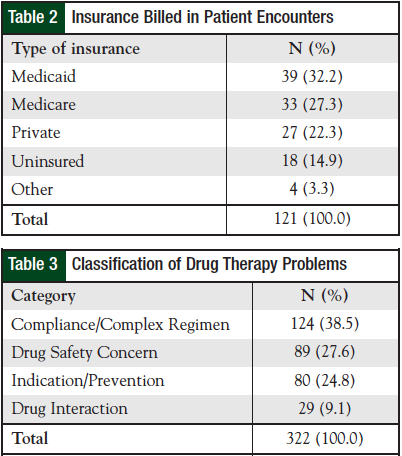
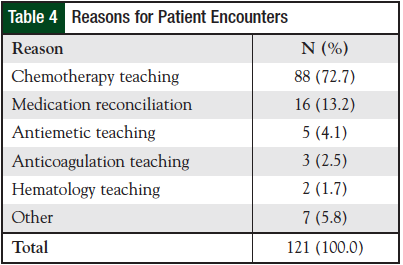
Of the 121 patient encounters during this time, 322 drug therapy problems were recognized. The number of drug therapy problems identified in an encounter ranged from 1 to 6, with an average of 2.7 drug therapy problems per encounter. Of the drug therapy problems identified, 281 were resolved by the pharmacist and documented in the patient care note. More than 85% of the drug therapy problems identified by pharmacists during a patient encounter were resolved within the same encounter. As noted in Table 3, drug therapy problems were identified most frequently (N = 124) in the Compliance/Complex Regimen class. Patients were referred to the pharmacist by oncologists and nurses at the initiation of chemotherapy or for management of side effects during chemotherapy treatment. The primary reasons for the encounter, as highlighted in Table 4, were for chemotherapy teaching and medication reconciliation, in approximately 73% and 13% of encounters, respectively. In addition, pharmacists were consulted to educate patients on the use of anticoagulants (2.5% of encounters) and for antiemetic instructions not incorporated into previous chemotherapy teaching (4%).
Patients seen in the cancer center within HCMC spent an average of 33 minutes with a pharmacist (range, 15-105 minutes). On average, the pharmacists saw between 2 and 3 patients weekly, which accounted for approximately 5% to 10% of infusion room visits for chemotherapy. This number excluded infusion room visits for nonchemotherapy medications.
The amount charged to third-party payers ranged from $34 to $148 and averaged $101 per encounter for the 121 encounters. A total of $12,224 in potential revenue was created through approximately 66.5 hours of face-to-face time with patients. This demonstrates the potential earnings for each patient encounter and not the actual returned revenue, which is as of yet unknown.
Discussion
In this retrospective analysis, oncology clinical pharmacists conducted MTM visits while maintaining a clinical practice, illustrating the feasibility of implementing MTM in this setting.
MTM visits are a recent addition to the clinical responsibilities of the 2 part-time residency-trained, board-certified oncology pharmacists (equal to 1 full-time pharmacist). Their current duties include patient-specific verification of chemotherapy treatment plans; drug information for nursing, medical staff, and patients; collaborative practice management of infusion reactions, management of nausea and vomiting, and other chemotherapy adverse effect management; coverage of code blue calls in a specified area of the hospital; pharmacy student and resident teaching; quality improvement project development; and committee membership. Duties also include coordination for access to medications (referral for patient assistance programs and grants), development of pharmacy policies and procedures, and coordination of care for inpatient chemotherapy.
Current duties do not include dispensing of medications.
The primary outcome for this study was to describe the average number of drug therapy problems identified and recorded per patient encounter at an ambulatory oncology practice. Drug therapy problems were categorized as Indication/Prevention, Compliance/Complex Regimen, Drug Interaction, and Drug Safety Concern.
The early identification and resolution of drug therapy problems provide patients with a more preventive and less reactive approach to oncology care. However, there are barriers to the development of MTM services, such as the additional work for clinical pharmacists as well as the unknown actual revenue for the services provided.
At HCMC, there was no additional cost to staff clinical pharmacists, because the MTM practice model was incorporated into the current workflow and the volume of visits was manageable without adding staff. Before the incorporation of the MTM model into this clinical practice, pharmacists provided patient education. With the creation of the MTM model, pharmacists are now able to charge for the patient education services that they have traditionally provided. To separate this type of patient encounter from purely counseling a patient, pharmacists must document that they reviewed the patient’s medication history, document the patient’s chief complaint, and supply a list of the patient’s prescription and nonprescription medications, with their respective indications. Based on the information gathered, pharmacists must give recommendations for improving the patient’s medication use.5
The MTM program is successful in part because pharmacists currently document, as needed, for reimbursement. Documentation requirements for reimbursement set by Minnesota include date of encounter/documentation, time spent with the patient, list of prescription and nonprescription drugs (ie, drug doses, directions, and intended use), relevant medical devices, alcohol and tobacco history, environmental factors that impact the patient, assessment of identified drug problems, a written plan including goals and actions needed to resolve the drug therapy problems, and information and instructions for the patient. Pharmacists have the ability to assign billing levels to each encounter and bill third-party payers based on the amount of time spent with the patient and the complexity of drug therapy problems, allowing for payment of the services provided. Billing allows for the addition of potential revenue for work that was being completed by pharmacists on a daily basis. Acceptance of MTM services by patients, physicians, pharmacists, and insurers is crucial for the success of these services.6
Ruder and colleagues looked at clinical interventions and consultations through the addition of a clinical oncology pharmacist in an ambulatory care oncology setting.7 The initial goal of adding a pharmacist was to help manage therapies and control cost. The researchers divided interventions into drug-related and consultative, which accounted for 35% and 65% of the interventions, respectively.7 Drug-related interventions consisted of documenting drug history, drug information, and drug calculations. Consultation interventions comprised patient education sessions and follow-ups. After approximately 2 years of data collection, 583 clinical interventions were documented on 199 patients; the average time spent per intervention was 10 minutes.7
Ruder and colleagues found that of 378 consultation interventions, 143 were for patient teaching.7 In the current study, chemotherapy teaching was documented as the primary reason for the encounter 73% of the time but was mentioned in almost all of the patient care notes. Face-to-face visits often occurred before day 1 of chemotherapy, allowing patients to ask questions about treatment. Pharmacists took this opportunity to review the management of side effects produced by chemotherapy medications and made arrangements for prophylaxis if necessary. Arrangements included sending prescriptions to pharmacies, assembling pillboxes, obtaining prior authorizations, and referral to patient assistance programs.
Ruder and colleagues reported that an average of 10 minutes was spent per intervention (patient consultation lasted 9-12 minutes),7 whereas in the current study we found that an average of 33 minutes was spent per face-to-face patient encounter. The initial goal of Ruder and colleagues was not to provide clinical services to patients; rather, this was a role that the pharmacist took over time.7 In the current study we specifically looked at the drug therapy problems identified through face-to-face interactions, which likely contribute to the differences in intervention times between the studies. Although Ruder and colleagues included interventions not directly associated with patient interaction, there was added value with the addition of a clinical oncology pharmacist and a positive impact on patient care.7
Reimbursement from insurance companies has been a limiting factor in the advancement of MTM services. Medicaid is currently one form of insurance that accepts MTM billing claims and accounted for 32.2% of billed encounters in this study. Although there is a billing method in place for pharmacists to bill for encounters, insurance companies want proof that MTM improves patient outcomes while saving money, and they will continue to drive the demand for MTM services through their willingness to pay for the service.
Minnesota has a fee-for-service system, allowing providers to submit for and receive reimbursement electronically for their services, based on a predetermined rate. MHCP providers must follow billing policies, including billing after completion of a covered service in a timely manner, using billing codes that are compliant with the HIPAA (Health Insurance Portability and Accountability Act of 1996), billing electronically, and billing the customary charge.8 Current billing codes for MTM recognize an initial face-to-face encounter and follow-up visits, both lasting more than 15 minutes. The approval of the Medicare Modernization Act in January 2006 granted pharmacists the ability to bill for MTM services.9 In July 2005, three CPT® (Current Procedural Terminology) codes were approved for pharmacists to use when billing for MTM services.9
Although this review did not have the capabilities to look into the amounts that the hospital was actually paid for the MTM services, other research has documented that oncology pharmacists help ensure the best use of medications, which can help save health systems money through decreased numbers and lengths of hospital stays.1 In addition, dollars are saved by decreasing the number of adverse events expected from chemotherapy regimens.1 Beyond face-to-face interactions, Ruder and colleagues documented a savings of $210,000 in patient charges by reducing chemotherapy doses, preventing drug waste, and rounding to the nearest vial size when appropriate.7
Limitations
Because this study describes the current practice at HCMC, it has a single-center research design. In addition, this retrospective review was not blinded; there was no control population with which to compare the study participants; and all patients receiving treatment at the cancer center were eligible for pharmacist referral, without a formal outline of inclusion and exclusion criteria. Patients were referred by doctors and nurses, as needed, for medication reconciliation and teachings on chemotherapy, hematology, antiemetics, and anticoagulation.
Moreover, variability in documentation existed because of the free text nature of the notes, even though the procedure for documentation was the same for the 2 participating pharmacists. Another limitation is that drug therapy problems are a surrogate end point for actual patient outcomes, and actual patient outcomes would be preferred.
Conclusions
The purpose of the current study was to retrospectively review the outcomes of the HCMC oncology MTM service in 2010. It is feasible to integrate an MTM service into an ambulatory oncology clinical practice. This service is a valuable tool for educating patients regarding chemotherapy regimens, completing medication reconciliation, and addressing supportive care issues to prevent drug therapy problems in patients with cancer.
Author Disclosure Statement
Dr Moss, Dr Won, and Dr Weber have reported no conflicts of interest.
References
- Stull DM, Iannucci A, Bertin RJ. Board-certified oncology pharmacists: partners in the multidisciplinary care of cancer patients. Community Oncol. 2006;3:284-286.
- American Pharmacists Association. APhA MTM Central. www.pharmacist.com/ mtm. Accessed August 15, 2011.
- Hwang JP, Holmes HM, Kallen MA, et al. Accuracy of reporting current medications by cancer patients presenting to an emergency center. Support Care Cancer. 2010;18:1347-1354.
- Liekweg A, Westfeld M, Jaehde U. From oncology pharmacy to pharmaceutical care: new contributions to multidisciplinary cancer care. Support Care Cancer. 2004;12:73-79.
- Thompson CA. National billing codes announced for pharmacists’ clinical services. Am J Health Syst Pharm. 2005;62:1640-1642.
- Pellegrino AN, Martin MT, Tilton JJ, Touchette DR. Medication therapy management services. Drugs. 2009;69:393-406.
- Ruder AD, Smith DL, Madsen MT, Kass FH. Is there a benefit to having a clinical oncology pharmacist on staff at a community oncology clinic? J Oncol Pharm Pract. 2011;17:425-432.
- Minnesota Department of Human Services. Billing policy overview. MHCP Provider Manual Website. http://www.dhs.state.mn.us/main/idcplg?IdcService=GET_DYNAMIC_CONVERSION&RevisionSelectionMethod=LatestReleased&dDocName=id_008924. Revised May 24, 2011. Accessed November 25, 2011.
- Zingone MM, Malcolm KE, McCormick SW, Bledsoe KR. Analysis of pharmacist charges for medication therapy management services in an outpatient setting. Am J Health Syst Pharm. 2007;64:1827-1831.
